Clear Sky Science · en
Multi-omic phenotyping of MAPT V337M neurons reveals early changes in axonogenesis and tau phosphorylation
Why this research matters for brain health
Many forms of dementia, including Alzheimer’s disease and frontotemporal dementia, involve clumps of a protein called tau inside brain cells. Doctors can see that abnormal tau strongly tracks with memory and thinking problems, but exactly how tau goes wrong—and what it does to young, still-developing nerve cells—has remained unclear. This study zooms in on an inherited tau mutation linked to frontotemporal dementia and asks a simple but crucial question: what happens to neurons long before symptoms appear?
A closer look at a risky version of tau
The researchers focused on a single genetic change, called V337M, in the MAPT gene, which provides the blueprint for tau. People who carry this mutation are almost certain to develop frontotemporal dementia, often in midlife. To study its early effects, the team used human induced pluripotent stem cells—adult cells reprogrammed to behave like embryonic cells—and turned them into neurons in the lab. They compared neurons with normal tau, neurons carrying one or two copies of the V337M mutation, and neurons in which tau levels were deliberately reduced.
Following many molecular clues at once
Instead of tracking just one signal, the scientists took a “multi-omic” view: they measured gene activity (RNA sequencing), the openness of DNA regions that control genes (ATAC-seq), and thousands of proteins and their chemical tags (proteomics and phosphoproteomics). Across these different layers, a consistent message emerged. In neurons with the V337M mutation, and in neurons where tau was lowered, the strongest changes involved genes and proteins that build and shape axons—the long, cable-like projections that carry signals to other cells. These changes appeared early, within the first weeks of neuronal development, before any obvious signs of cell death or protein clumping.
Young neurons grow shorter, not stronger
To see whether these molecular changes actually affected cell shape, the team watched young neurons grow in culture dishes. They labeled cell membranes with a fluorescent marker and traced axons over several days as they extended across the plate. Neurons with the V337M mutation and neurons with reduced tau both produced noticeably shorter main axons and less total outgrowth, even though the number of small side branches was not dramatically altered. In other words, the central “wire” of the neuron failed to reach as far as it should. This points to a loss of tau’s normal support role in axon extension, rather than an obvious toxic effect that kills cells outright.
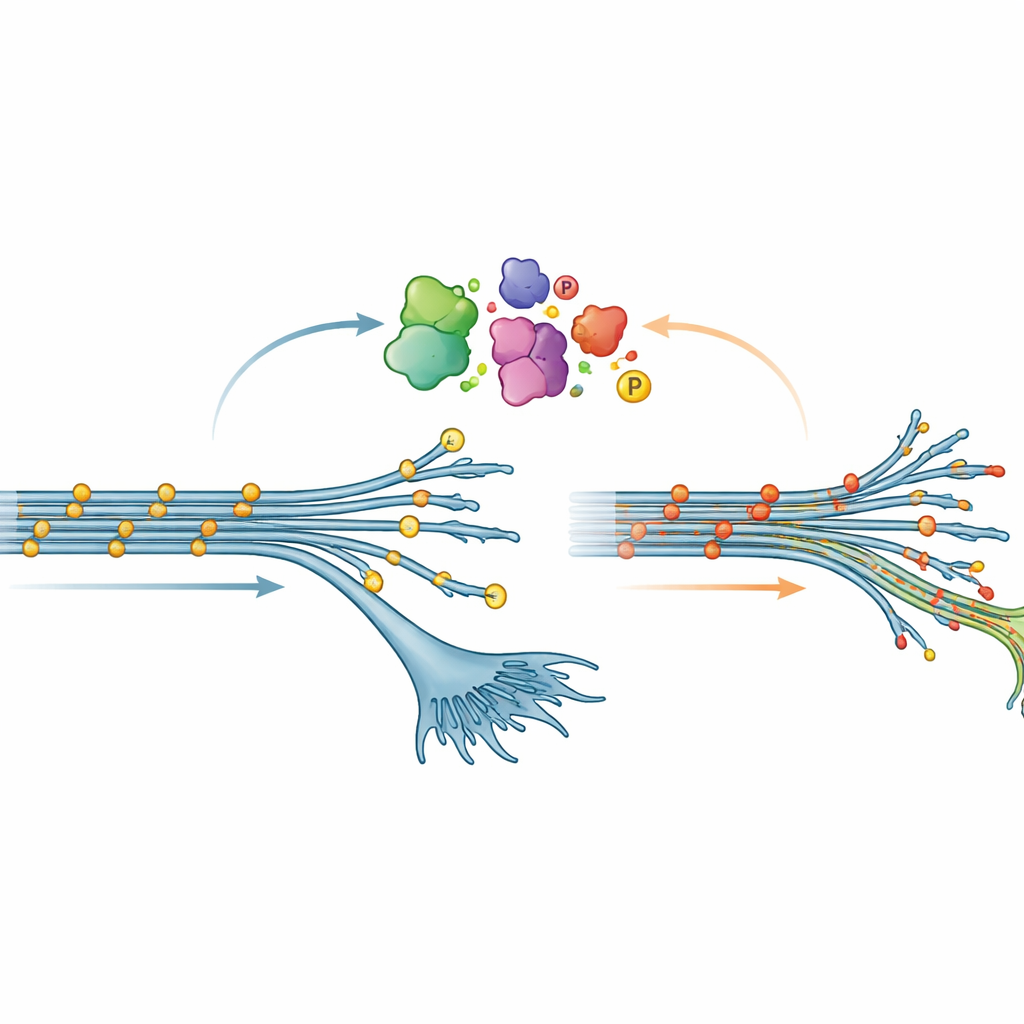
An unexpected pattern of tau chemistry
One of the most surprising discoveries concerned phosphorylation, a common chemical modification where tiny phosphate groups are attached to proteins. In many brain diseases, tau becomes heavily phosphorylated; this is often viewed as a harmful change that encourages tau to form tangles. Here, however, early-stage neurons carrying the V337M mutation showed the opposite pattern: tau was less phosphorylated at many sites than normal. This “hypophosphorylation” gradually faded as the neurons matured, but it was robust enough to be confirmed by both mass spectrometry and standard protein tests. When the team artificially overproduced V337M tau in neurons whose own tau had been dialed down, the same low-phosphorylation state reappeared, suggesting that the mutation itself reshapes how tau is modified inside cells.
Signaling pathways that reshape axons and tau
To understand what drives these changes, the researchers searched for signaling pathways—chains of enzymes that add or remove phosphate groups—that were altered in V337M neurons. Their phosphoproteomics data pointed to proteins involved in neuron projection growth and to specific kinase families, including the p38 MAP kinase pathway, which is known to influence neurite outgrowth. They then performed large-scale CRISPR screens, turning thousands of genes up or down to see which ones changed the level of a key phosphorylated form of tau. Many hits were genes that organize the cell’s internal skeleton and axon structure, and several p38 pathway components affected tau phosphorylation specifically in V337M neurons. This suggests an intimate link between the machinery that builds axons and the machinery that tunes tau’s chemical state.
What this means for understanding dementia risk
Put together, the findings suggest that the V337M tau mutation does not simply make tau more toxic in the classic sense. Instead, in very early life it seems to blunt tau’s usual support for axon growth and alters how tau is phosphorylated, in ways that overlap with what happens when tau is reduced or lost. Subtle wiring defects during development or early adulthood—combined with ongoing changes in tau chemistry—could help explain why people carrying MAPT mutations show cognitive differences decades before clear dementia emerges. This work highlights that therapies aimed at broadly lowering tau might need to consider tau’s helpful roles in young and healthy neurons, and that carefully tuning, rather than simply blocking, tau-related pathways could be key to protecting the brain over a lifetime.
Citation: Mohl, G.A., Dixon, G., Marzette, E. et al. Multi-omic phenotyping of MAPT V337M neurons reveals early changes in axonogenesis and tau phosphorylation. npj Dement. 2, 24 (2026). https://doi.org/10.1038/s44400-026-00076-w
Keywords: tau protein, frontotemporal dementia, axon growth, neuronal development, protein phosphorylation