Clear Sky Science · en
Bacterial cell fate under stress: lipid remodeling and antimicrobial peptide attack
Why this matters for fighting infections
As more bacteria become resistant to our best antibiotics, scientists are turning to natural defense molecules called antimicrobial peptides, found in organisms from bees to humans. But bacteria are not passive targets: they can remodel the oily membrane that surrounds their cells, potentially dodging these new drugs. This study asks a practical question with big implications: when bacteria change their membrane lipids under stress, can two well-known antimicrobial peptides—melittin from honey bee venom and human LL-37—still kill them, and how exactly do they do it?
Bacteria under pressure
Bacteria constantly face harsh conditions: shifts in temperature or salt, immune attacks, and exposure to antibiotics. To cope, they reconfigure the fatty molecules, or lipids, that make up their outer boundary, tuning properties like thickness, fluidity, and surface charge. A key lipid building block, phosphatidic acid, normally present only in tiny amounts, can build up when other negatively charged lipids are depleted. Those charged lipids are usually prime landing spots for positively charged antimicrobial peptides. The authors used the model bacterium Escherichia coli and a mutant strain that largely lacks two major anionic (negatively charged) lipids, replacing them with more phosphatidic acid, to mimic the kind of membrane remodeling that might occur under drug pressure.
Two peptides, two styles of attack
The team focused on melittin and LL-37, classic membrane-active peptides that share a helical shape but differ in how their water-loving and fat-loving regions are arranged. Using structure-prediction tools and model membranes, they showed that both peptides can bind not only to the usual anionic lipids but also to phosphatidic acid, inserting into these altered membranes and disturbing their organization. Melittin proved especially powerful at making model E. coli membranes leaky, while LL-37 showed weaker direct leakage under the same conditions, hinting that it may rely on a broader set of actions than simple hole punching in the membrane.
Watching cells die in real time
To connect membrane damage with actual bacterial death, the researchers ran six complementary assays side by side. These tracked how well peptides bound to the cell surface, whether the outer and inner membranes became permeable, whether the electrical potential across the inner membrane collapsed, and how the cells’ energy (ATP) and metabolic enzyme activity changed. Crucially, they measured these effects in both normal E. coli and the lipid-remodeled mutant while also counting how many cells could still form colonies. This produced detailed “profiles” showing which events rose and saturated at which peptide concentrations and times, effectively mapping the road from first contact to cell death.
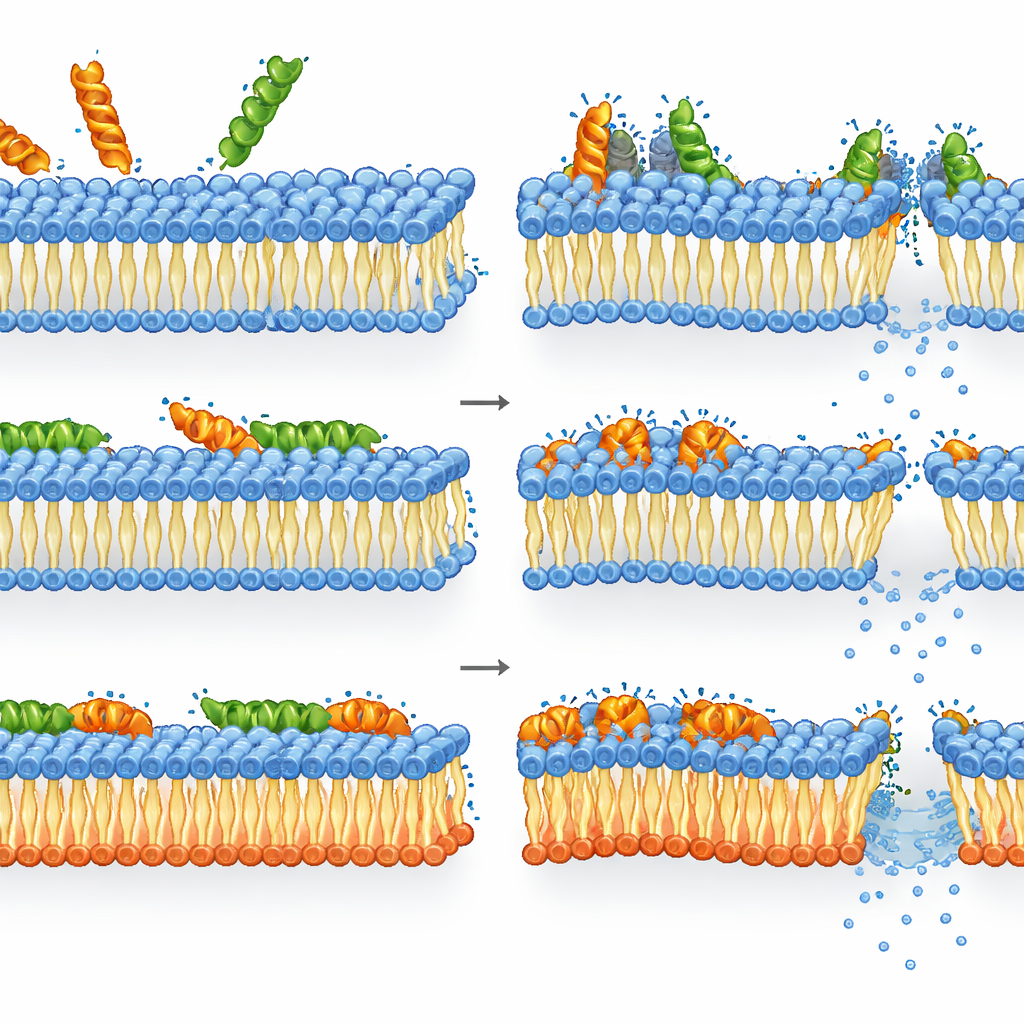
How melittin and LL-37 handle remodeled membranes
Melittin turned out to be even more lethal against bacteria lacking the usual anionic lipids than against the wild type. In the mutant, loss of membrane integrity, declining energy levels, and loss of viability were tightly linked: once melittin disrupted the remodeled membrane, the cells rapidly died. In the wild type, however, the picture was more nuanced. Membrane permeabilization and depolarization were already saturated at concentrations where ATP and enzyme activity remained relatively high. In other words, many cells had badly damaged membranes yet were still metabolically active but unable to form colonies—suggesting melittin’s killing power comes from severe, possibly transient, membrane damage that the cells cannot fully recover from, even if their internal machinery briefly keeps running.
A tougher, more versatile human peptide
LL-37 behaved differently. It neutralized surface charge and damaged both membranes in wild-type and mutant cells to nearly the same extent, indicating that its action was largely independent of the precise mix of anionic lipids. At concentrations where inner and outer membranes were already heavily compromised, many cells still showed measurable metabolic activity, and ATP levels did not always drop to zero. This timing gap implies that LL-37’s final killing blow likely involves targets beyond the membrane itself—such as disrupting energy generation, triggering oxidative stress, or stiffening the cytoplasm by binding to DNA and ribosomes—once it has gained access to the cell interior.
What this means for future antimicrobials
Overall, the study shows that membrane remodeling—even drastic shifts away from the usual anionic lipids toward phosphatidic acid—does not rescue bacteria from melittin or LL-37. Both peptides adapt to the new lipid landscape, still binding, inserting, and ultimately killing. The detailed assay profiles reveal that bacterial death is not a simple on–off switch at the moment the membrane is pierced, but a web of overlapping events that can leave cells damaged and non-growing long before all metabolic signals vanish. By linking specific membrane and viability markers to actual killing, this work provides a blueprint for how to evaluate and compare new antimicrobial peptides, and it suggests that simply changing membrane lipid composition is unlikely to be an easy escape route for bacteria facing these potent, multi-pronged molecules.
Citation: Vejzovic, D., Schwaiger, T., Topciu, A. et al. Bacterial cell fate under stress: lipid remodeling and antimicrobial peptide attack. npj Antimicrob Resist 4, 22 (2026). https://doi.org/10.1038/s44259-026-00195-7
Keywords: antimicrobial peptides, bacterial membranes, lipid remodeling, antibiotic resistance, LL-37 and melittin