Clear Sky Science · en
Aging clocks delineate neuron types vulnerable or resilient to neurodegeneration and identify neuroprotective interventions
Why some brain cells age faster than others
Why do some brain cells wither early in life while others stay robust for decades? This question lies at the heart of many disorders of aging, from Alzheimer’s to Parkinson’s disease. In this study, researchers used a tiny roundworm, Caenorhabditis elegans, whose entire nervous system is mapped cell by cell, to ask whether different neuron types have their own “biological ages” and whether those ages predict which cells will degenerate. Their work not only pinpoints vulnerable neuron types and a molecular process that speeds their decline, but also reveals small molecules that can slow or accelerate neuronal aging—offering clues that may extend to the human brain.
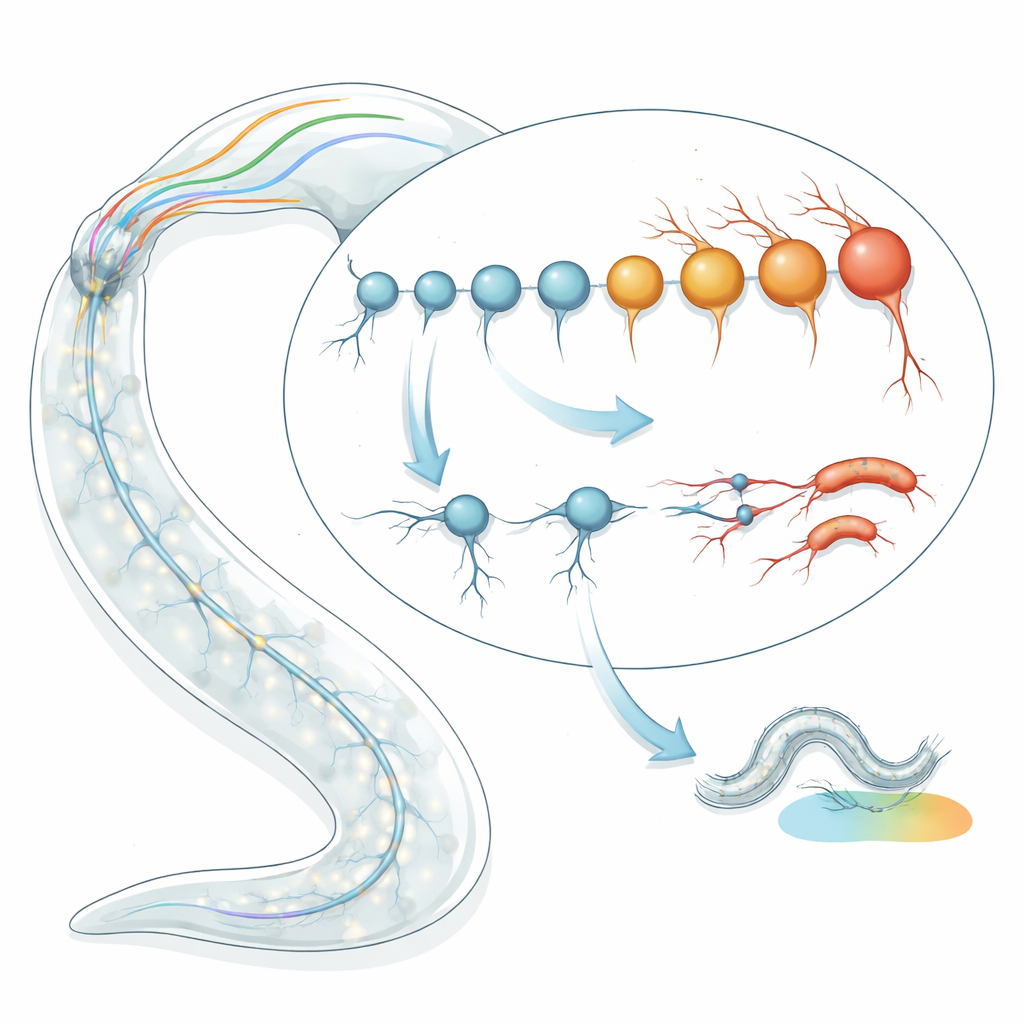
Timing the age of single nerve cells
The team began by applying transcriptomic “aging clocks” to individual neuron types in C. elegans. Instead of looking at the worm’s age in days, they examined patterns of gene activity to estimate each cell type’s biological age. Using two independent clock models—one based on characteristic aging gene signatures and another that captures how random changes accumulate over time—they analyzed RNA sequencing data for 128 well-defined neuron types in late larval and young adult worms. Surprisingly, they found nearly a two- to four-fold spread in predicted biological age among neurons that were all the same chronological age. Some neurons appeared much “younger” than expected, while others seemed prematurely “old,” and these estimates were consistent across multiple datasets and sequencing methods.
From molecular age to visible damage and lost behavior
To test whether these age predictions mattered in real worms, the researchers tracked the physical condition of selected neurons over time using fluorescent markers. Neurons predicted to be biologically young (such as I2, OLL and PHC) showed only mild structural damage in a minority of animals as the worms aged. In stark contrast, neurons predicted to be old (including ASI, ASJ and ASK) already showed signs of deterioration in nearly half of late larvae, rising to about 90% by a week of adulthood. The damage appeared as tiny swellings and outgrowths along the nerve fibers and increased roughly randomly along each neurite, consistent with a stochastic aging process rather than a fixed developmental program. Early structural decline also translated into real behavioral deficits: in circuits where a single neuron type controls a specific task, degeneration of that neuron closely matched failures in pathogen avoidance or salt-based learning.
Why exposed sensory neurons are at special risk
Mapping biological age estimates onto the worm’s full nervous system connectome revealed a striking pattern. The oldest neurons clustered among ciliated sensory cells in the head, especially amphid neurons whose cilia are directly exposed to the environment. These cells detect chemicals, odors and other cues and are richly endowed with neuropeptides and receptors. Across neuron types, a higher number of expressed neuropeptides and receptors correlated with older biological age, whereas the sheer number of synapses did not. Ciliated neurons whose sensory endings protrude to the outside world were the most aged, suggesting that both environmental exposure and heavy signaling workloads may predispose them to early decline. This echoes human neurology, where environmentally exposed, ciliated neurons in the olfactory system are among the first to degenerate in Alzheimer’s disease.
Protein production as a driver—and a handle—on aging
To uncover mechanisms behind these age differences, the team grouped neurons by biological age and examined gene expression trends. They found that processes related to ribosome biogenesis, protein synthesis and nucleolar function were enriched in older neurons, along with changes in synaptic and neuropeptide signaling genes. This pointed to heavy protein production as a possible burden that accelerates neuronal wear and tear. The researchers then tested a modest dose of cycloheximide, a compound that partially slows translation. In fast-aging neurons, this gentle inhibition significantly reduced structural damage and preserved performance in a salt-learning task, with protective effects that persisted even after treatment stopped. Slower-aging neurons, by contrast, were largely unaffected. These results link high translational load to neuron-specific vulnerability and show that dialing down protein synthesis can restore structural and functional integrity in at-risk cells.
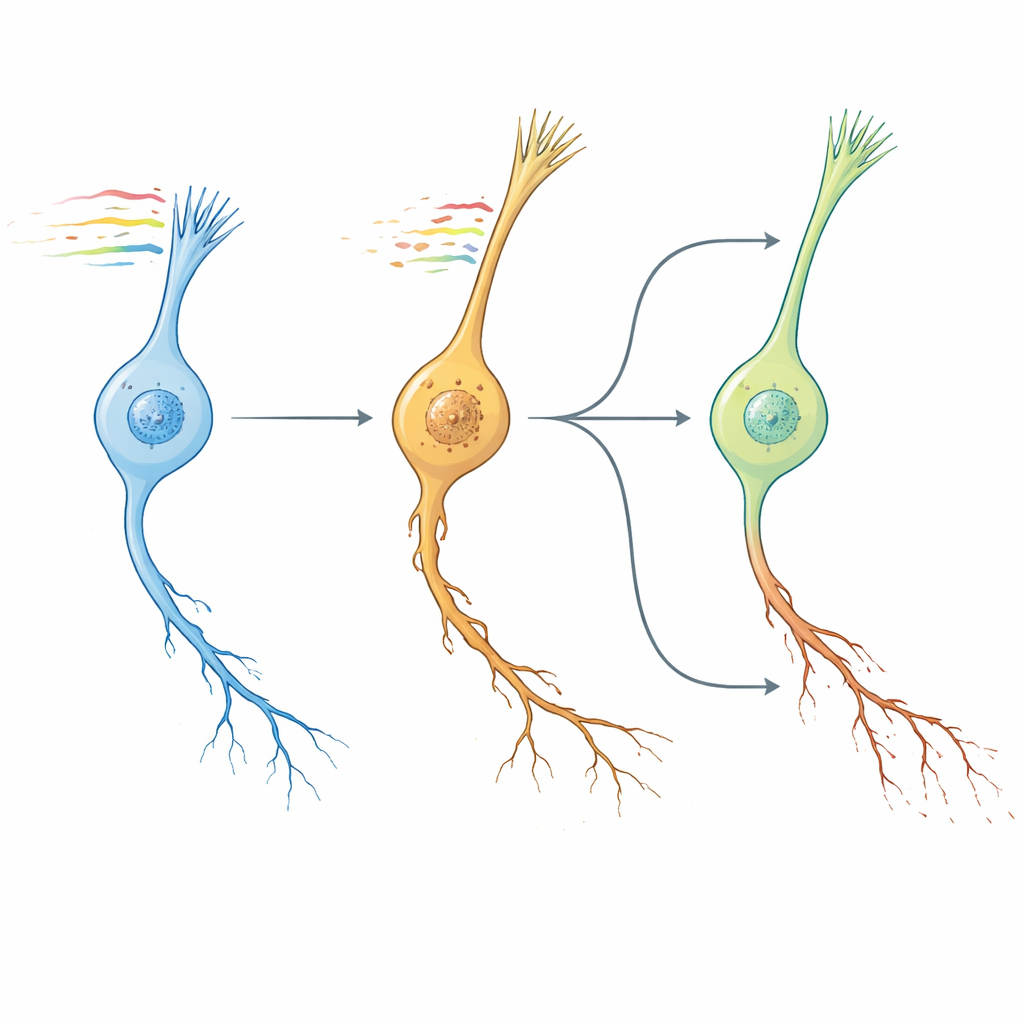
Worm clocks, human brains, and drug leads
Next, the authors asked whether their worm neuron aging pattern resembled brain aging in mammals. By comparing pathway-level changes across conserved genes, they found that the transcriptomic “NeuronAge” signature in C. elegans clustered tightly with aging trajectories from multiple mouse and human brain regions, while known geroprotective interventions—such as young blood factors, exercise and dietary compounds—showed opposing patterns. Leveraging this conservation, they turned to a large human cell-line resource to search in silico for small molecules whose effects counteract or mimic NeuronAge. Compounds whose profiles opposed NeuronAge were flagged as potential neuroprotectors; those that reinforced it as potential neurotoxins. Experimental tests in worms validated several predictions. The plant-derived metabolite syringic acid and the drug vanoxerine both reduced degeneration in fast-aging neurons without harming young ones, while agents such as Bay K8644 and WAY-100635 exacerbated neurite damage and impaired behavior. Even resveratrol, often touted as protective, emerged as pro-aging in this system and increased neuronal pathology.
What this means for brain health
In essence, this work shows that even within a single nervous system, different neuron types march through aging at different speeds, and that these internal clocks predict which cells will degenerate and lose function first. Sensory neurons that face the outside world and run high levels of protein production appear especially fragile. By tying neuron-specific aging to conserved gene expression pathways, the study offers a way to translate insights from a simple worm to the human brain. Just as importantly, it demonstrates that transcriptomic aging signatures can be used as a powerful filter to identify molecules that slow or hasten neurodegeneration. While much remains to be done before these candidates can be considered for human therapy, the approach points toward a future in which we can not only measure the biological age of individual neuron types, but also rationally design interventions to keep them younger for longer.
Citation: Gallrein, C., Meyer, D.H., Woitzat, Y. et al. Aging clocks delineate neuron types vulnerable or resilient to neurodegeneration and identify neuroprotective interventions. Nat Aging 6, 849–868 (2026). https://doi.org/10.1038/s43587-026-01067-5
Keywords: neuronal aging, neurodegeneration, protein synthesis, Caenorhabditis elegans, neuroprotection