Clear Sky Science · en
CRISPR-Cas9 screening identifies ATOX1-driven cisplatin resistance mechanisms in liver cancer and evaluates targeted inhibitor efficacy
Why this research matters
Liver cancer is one of the deadliest cancers worldwide, and many patients receive a common chemotherapy drug called cisplatin. Over time, however, tumors often learn to shrug off the drug, allowing the disease to return and spread. This study uncovers a surprising molecular culprit behind that resistance and introduces a designer compound that may help cisplatin work better and more safely. For readers, it offers a glimpse of how modern genetics, computer modeling, and chemistry can be combined to outsmart cancer’s defenses.
How liver tumors dodge chemotherapy
Cisplatin kills cancer cells by damaging their DNA, but tumors can become resistant through many tricks: pumping the drug out, repairing DNA faster, or rewiring survival signals. The authors used a powerful gene-editing tool, a genome-wide CRISPR-Cas9 screen, to systematically switch off nearly every gene in two liver cancer cell lines and then expose them to cisplatin. They asked a simple question: when a gene is knocked out, do cells become more or less sensitive to the drug? Among dozens of genes linked to resistance, one stood out—ATOX1, a small protein that normally helps cells handle the essential metal copper. Liver cancer cells had more ATOX1 than healthy liver cells, and patients whose tumors showed high ATOX1 levels tended to have worse survival and more cisplatin resistance.
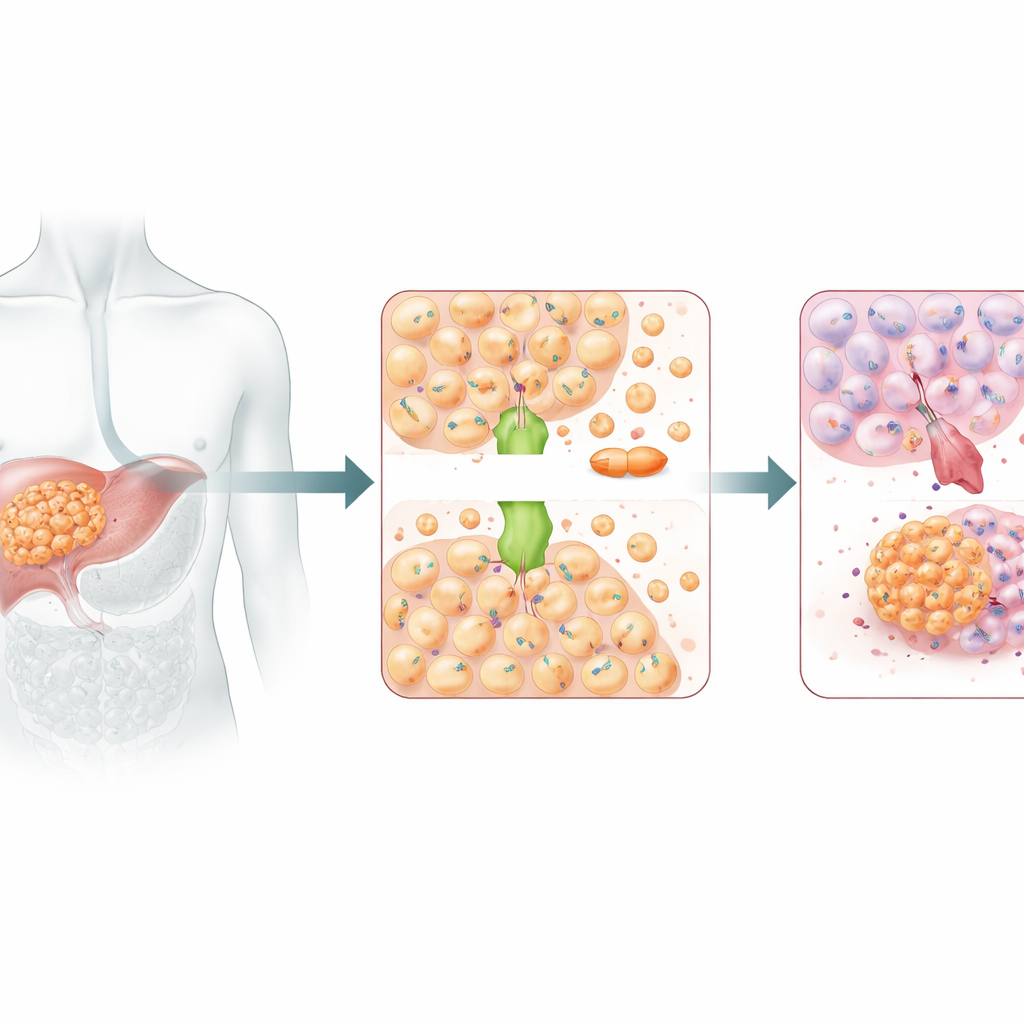
A copper courier with a dark side
ATOX1 usually acts as a copper courier, shuttling copper ions inside cells to keep this vital metal in balance. But copper and cisplatin look similar enough that they may ride some of the same transport routes. Earlier structural work had hinted that ATOX1 can bind cisplatin directly. In this study, silencing ATOX1 in liver cancer cells made them far more vulnerable to cisplatin: their growth slowed, they formed fewer colonies, and they underwent more programmed cell death. In mice carrying human liver tumors, knocking down ATOX1 also made cisplatin shrink tumors more effectively. Together, these results pointed to ATOX1 as a key helper that allows liver cancer cells to withstand platinum-based chemotherapy.
Designing a precision blocker
Rather than targeting copper in a broad way, which can be toxic, the team set out to block ATOX1 itself. They simulated the motion of the ATOX1 protein on a computer to find stable pockets where a drug might bind, then virtually tested 1.5 million small molecules for their ability to fit. After several rounds of filtering and lab validation, one candidate—dubbed compound 8—emerged as a strong and specific binder. Biophysical assays showed that compound 8 attached tightly to ATOX1 and stabilized its structure, while having little effect on a related copper protein that older inhibitors also hit. In cell tests, compound 8 was less damaging to normal liver cells than a previous ATOX1 drug, suggesting a better safety margin even though its single-agent anticancer activity was modest.
Turning copper against the tumor
The most striking effects appeared when compound 8 was paired with cisplatin. In liver cancer cells grown in dishes, combining the two drugs cut growth more than either alone, reduced colony formation, and triggered higher rates of cell death. In mice, the combination produced the smallest tumors and the lowest levels of cell division markers, without obvious harm to major organs. To understand why, the researchers analyzed gene activity and found that blocking ATOX1 caused copper to build up inside cancer cells. That copper buildup was linked to increased chemical tags—DNA methylation—on the control region of a gene called NOTCH1, part of a pathway that helps tumor cells survive stress and repair DNA. Heavier methylation meant the NOTCH1 switch was turned down, and its partner gene HES1 also quieted. Importantly, using a copper-chelating compound reversed these changes, showing that copper itself was central to the effect.
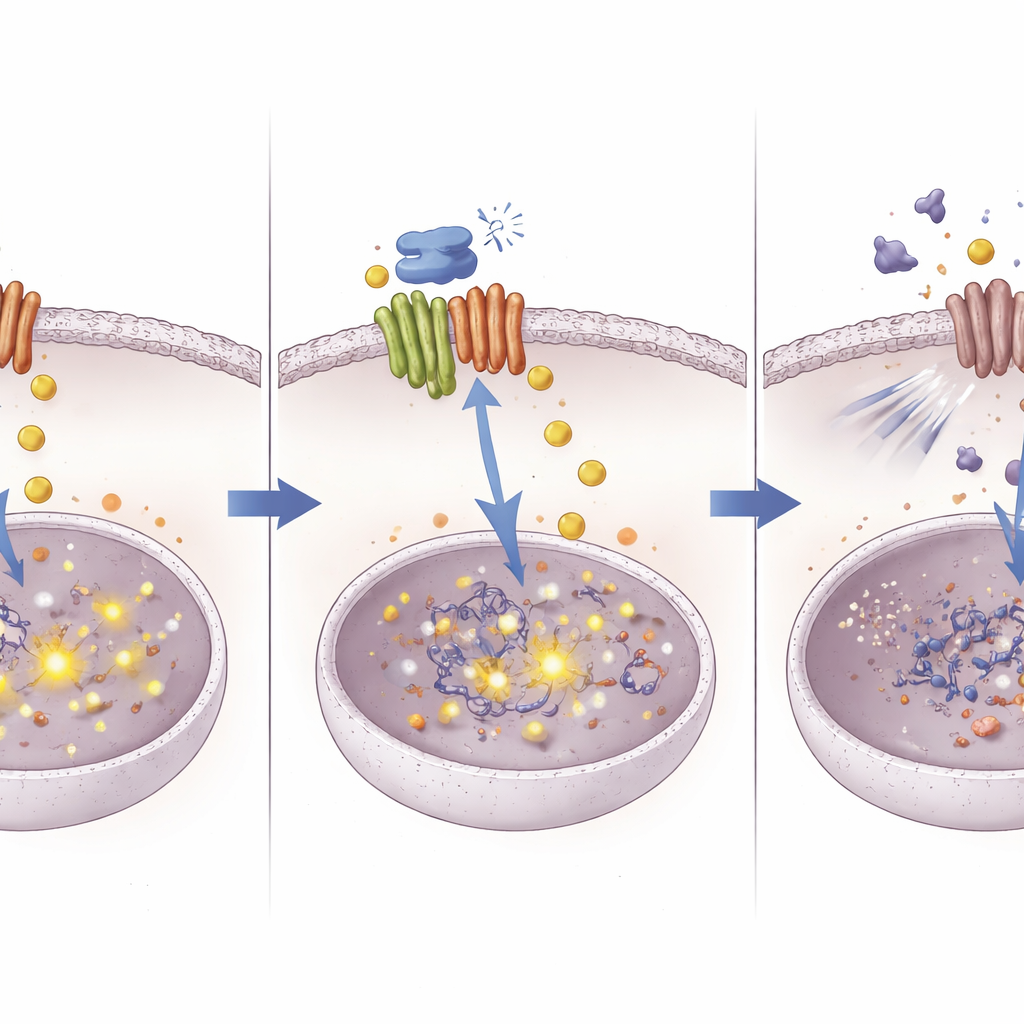
A new route to make old drugs work better
By tracing the path from a copper courier protein to DNA-level changes in a survival pathway, this study outlines a fresh way to tackle chemotherapy resistance in liver cancer. The work suggests that ATOX1 allows tumors to manage copper and signaling in a way that blunts cisplatin’s punch. Blocking ATOX1 with compound 8 causes copper to accumulate, which in turn silences NOTCH1 through DNA methylation and removes a key shield against cisplatin. Although compound 8 is still an experimental molecule, the concept it embodies—precisely targeting metal-handling proteins to rewire cancer cell behavior—could help revive the effectiveness of existing chemotherapies and inspire new combination treatments.
Citation: Hu, C., Tai, H., Zhu, R. et al. CRISPR-Cas9 screening identifies ATOX1-driven cisplatin resistance mechanisms in liver cancer and evaluates targeted inhibitor efficacy. Commun Biol 9, 439 (2026). https://doi.org/10.1038/s42003-026-09722-8
Keywords: liver cancer, cisplatin resistance, copper metabolism, ATOX1 inhibitor, Notch signaling