Clear Sky Science · en
Cuproptosis-associated PDHA1 promotes sarcoma progression and immunotherapy responsiveness via the E2F1–PD-L1 axis: a multi-omics and clinical validation study
Why this research matters
Sarcomas are rare but aggressive cancers that often resist modern immune-based treatments. Many patients with advanced disease exhaust standard options like surgery, chemotherapy, and radiation, yet still face poor survival. This study explores a hidden metabolic switch inside sarcoma cells that appears to both fuel tumor growth and shape how the immune system responds. Understanding this switch could open new ways to predict which patients benefit from immunotherapy and to design smarter combination treatments.
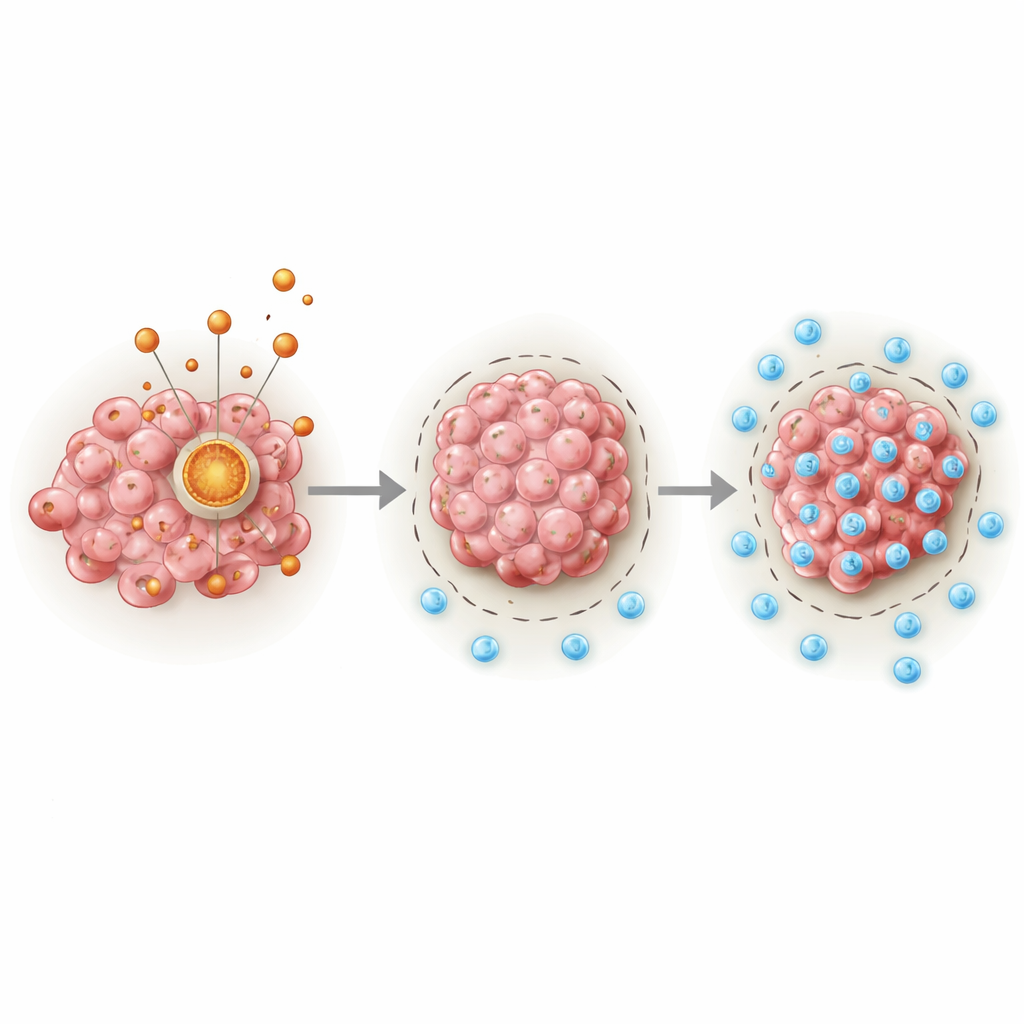
A hidden engine inside cancer cells
At the center of the story is PDHA1, a protein that acts as a key gateway between sugar breakdown and the cell’s main energy cycle in the mitochondria. The authors analyzed large cancer databases and clinical samples and found that sarcomas consistently carry higher levels of PDHA1 than normal tissues. Patients whose tumors had more PDHA1 tended to live for a shorter time, regardless of age, sex, or several other clinical factors. By combining PDHA1 levels with standard clinical information, the team built a prediction tool that estimated one-, three-, and five-year survival more accurately than traditional measures alone.
How metabolism shapes the tumor neighborhood
Beyond simply driving growth, PDHA1 appeared to reshape the tumor’s neighborhood—the mix of immune and support cells surrounding the cancer. Using computational methods that infer immune cell types from bulk tumor RNA, as well as single-cell sequencing of individual immune cells, the authors showed that PDHA1 is active not only in tumor cells but also across many immune populations, especially T cells and myeloid cells. Sarcomas with high PDHA1 tended to have signs of an “immune-excluded” state: fewer helpful killer T cells deep inside the tumor, more signals of stiff, fibrous stroma, and patterns suggesting that immune cells were kept at the margins rather than allowed to attack.
A molecular chain that blocks immune attack
Laboratory experiments revealed how PDHA1 connects metabolism to immune escape. When the researchers reduced PDHA1 levels in sarcoma cell lines, the cells divided less, moved less, formed fewer colonies, and underwent more programmed cell death. At the same time, levels of E2F1—a nuclear regulator of genes involved in cell division—and PD-L1—an immune “brake” on the surface of tumor cells—both dropped. By examining public DNA-binding data and performing targeted experiments, the team showed that E2F1 directly binds to the control region of the PD-L1 gene and switches it on. Restoring E2F1 in PDHA1-deficient cells brought PD-L1 back up and partially rescued cell survival, placing PDHA1 upstream of an E2F1–PD-L1 chain that helps tumors hide from T cells.
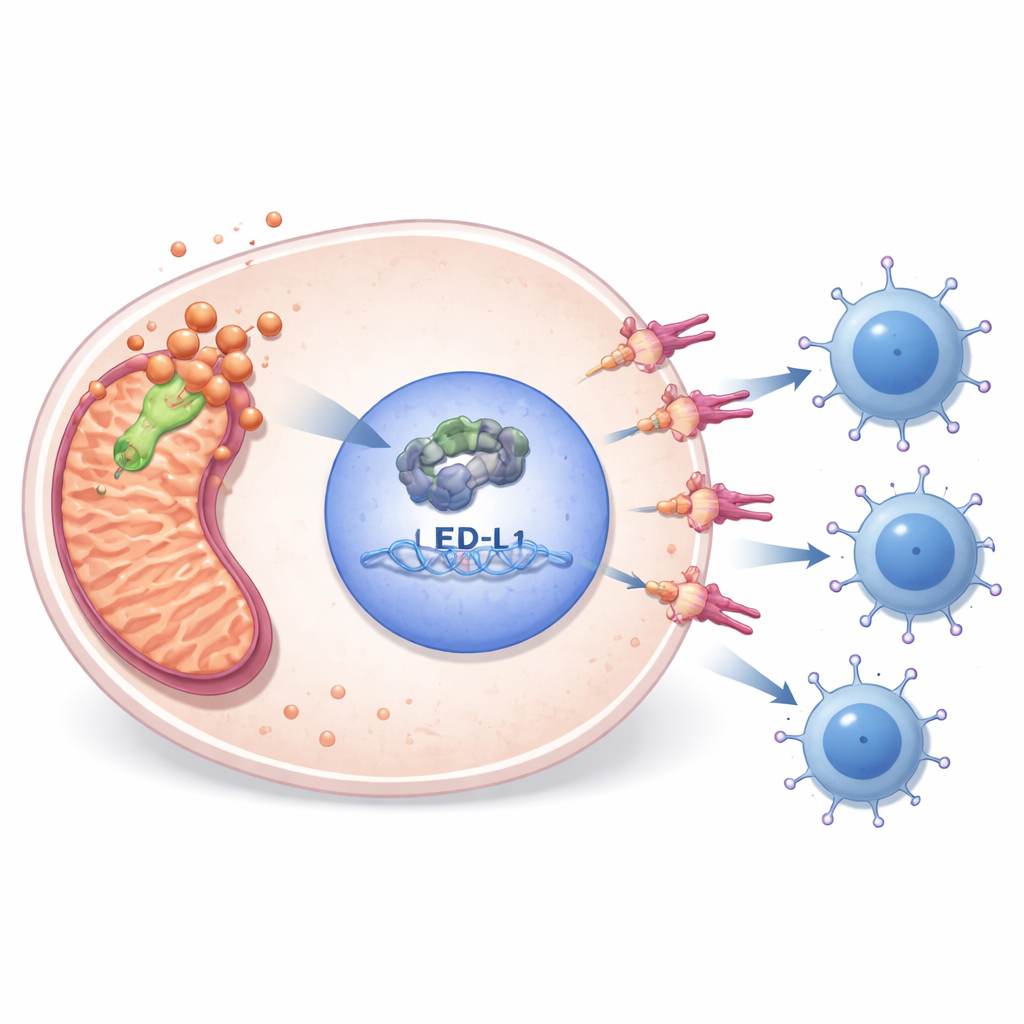
Copper-linked cell death and drug opportunities
PDHA1 is also tied to a recently described form of cell death triggered by copper, called cuproptosis. The study showed that blocking copper availability with a drug reduced a lipoylated mitochondrial protein linked to this pathway and dampened the PDHA1–E2F1–PD-L1 chain, especially when combined with PDHA1 knockdown. Genomic and epigenetic analyses suggested that high PDHA1 often coincides with DNA changes, low methylation of the PDHA1 gene, and RNA modification patterns associated with more aggressive tumors. Drug-screening databases and cell-based assays pointed to several medicines—such as the metabolic agent phenformin and an E2F1-pathway inhibitor—that sarcomas with active PDHA1 might be particularly sensitive to, hinting at rational combinations with immune checkpoint inhibitors.
Turning a bad sign into a treatment guide
In animal models and patient biopsies, tumors with high PDHA1 were larger, more proliferative, richer in PD-L1, and poorer in CD8⁺ killer T-cell infiltration. Yet these same high-PDHA1 tumors also showed features—like higher mutation burden and stronger checkpoint signals—that are often associated with better responses to drugs blocking PD-1 or PD-L1. In a small clinical cohort of advanced sarcoma patients receiving such immunotherapy, those with high PDHA1 were more likely to benefit. To a lay reader, the conclusion is that PDHA1 acts as both a gas pedal for sarcoma growth and a shield against immune attack, but this very behavior may make these tumors more vulnerable once the right brakes are applied. Measuring and targeting PDHA1 could therefore help identify high-risk patients, guide the use of immunotherapy, and inspire new metabolic–immune drug combinations for stubborn sarcomas.
Citation: Qin, H., Qi, T., Yao, N. et al. Cuproptosis-associated PDHA1 promotes sarcoma progression and immunotherapy responsiveness via the E2F1–PD-L1 axis: a multi-omics and clinical validation study. npj Precis. Onc. 10, 156 (2026). https://doi.org/10.1038/s41698-026-01298-0
Keywords: sarcoma immunotherapy, cancer metabolism, PDHA1, immune checkpoint blockade, tumor microenvironment