Clear Sky Science · en
Pyra-metho-carnil disrupts cancer cell proteostasis and induces apoptosis by binding to KDEL receptors
Turning Cancer’s Protein Factories Against Themselves
Cancer cells grow fast and churn out huge amounts of proteins, which must be carefully folded and shipped inside the cell. This study explores a small molecule that throws a wrench into that internal shipping system, overloading cancer cells with damaged proteins until they self‑destruct, while pointing to a new way to target tumors.
A Small Molecule with a Big Target
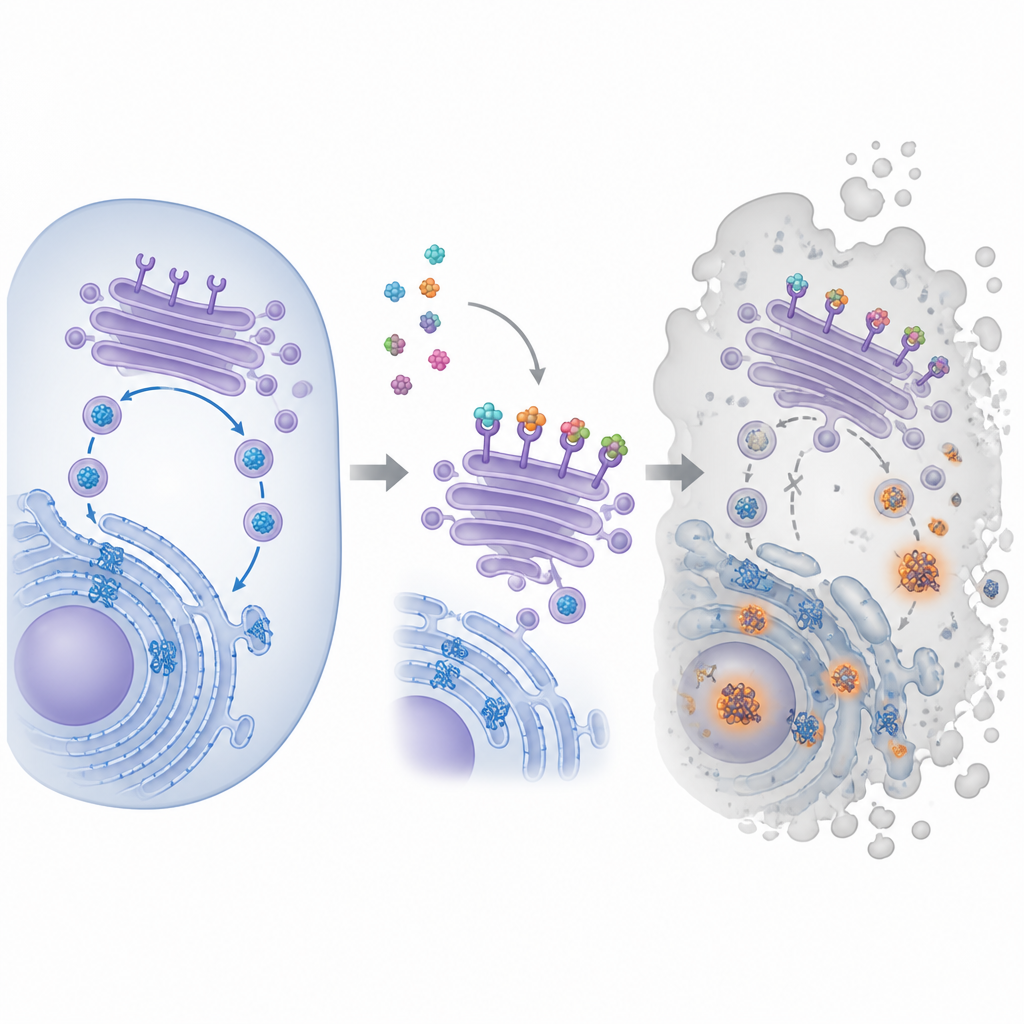
The compound at the center of this work is called pyra‑metho‑carnil, or PMC, originally found in a natural product library. Earlier studies showed that PMC slows the growth of many types of cancer cells, even when they carry different genetic mutations or resist other drugs. Yet its main target inside cells was unknown. To track that down, the researchers attached PMC to magnetic beads, let it fish for binding partners in cancer cell extracts, and then identified the captured proteins. This approach highlighted a family of proteins known as KDEL receptors, which sit in the cell’s internal shipping hub and help retrieve certain helper proteins back to the folding compartment.
Hijacking the Cell’s Recycling Route
Inside cells, new proteins are folded in a maze‑like structure called the endoplasmic reticulum, then pass through the Golgi, a stack of membrane pockets that sorts and sends them onward. KDEL receptors patrol the Golgi and grab helper proteins that carry a short tag at their tail, sending them back for reuse so the folding machinery can keep up. The team showed that PMC binds all three known human KDEL receptor types and changes where they sit inside cells. Using fluorescent tags, they found that receptors normally clustered at the front side of the Golgi moved out of place after PMC treatment. At the same time, a special dye revealed growing clumps of misfolded proteins, signaling that the protein folding and recycling loop was starting to fail.
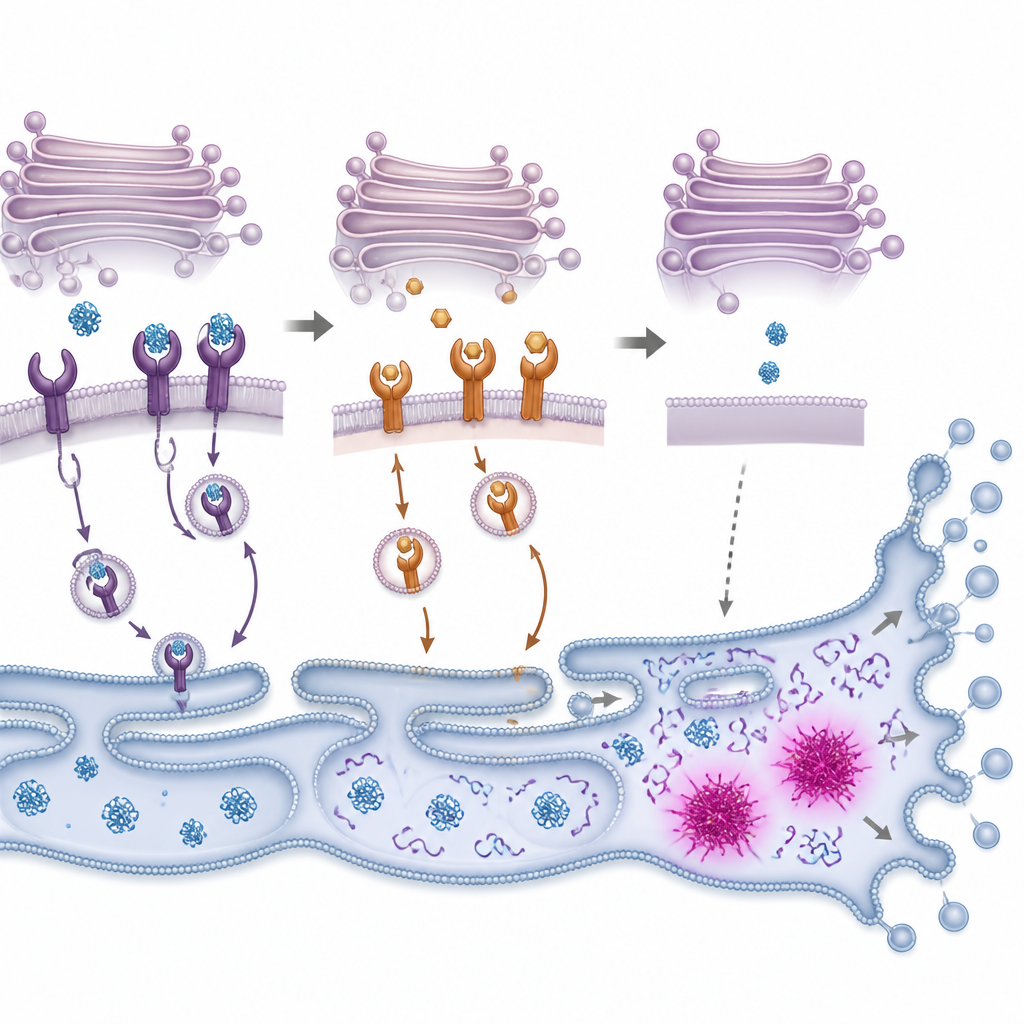
From Protein Overload to Cell Suicide
When misfolded proteins pile up, cells switch on an internal alarm system often called the unfolded protein response. This system first tries to restore balance by slowing new protein production and boosting the capacity to fold and clear damaged proteins. The researchers observed that PMC rapidly turned on key parts of this response in colon cancer cells, followed by activation of enzymes that chop up cellular components during programmed cell death. Blocking these enzymes, or reducing levels of a stress‑responsive switch called CHOP, made cells more likely to survive PMC exposure, although not completely. This suggests that PMC pushes cells past a tipping point where stress relief fails and self‑destruction pathways take over.
Making Cancer Cells More Vulnerable to Stress
The team also asked whether PMC could make cancer cells more sensitive to other forms of internal stress. They combined a low, otherwise harmless dose of PMC with thapsigargin, a compound that disturbs calcium handling and is widely used to provoke stress in the protein folding compartment. Together, the two drugs suppressed cell growth more strongly than thapsigargin alone, and stress markers rose higher, indicating that PMC lowers the threshold for damage. To show that KDEL receptors themselves are central to this effect, the scientists used small RNA molecules to reduce these receptors directly. That manipulation alone triggered the stress response and slowed cancer cell growth in a similar way.
What This Could Mean for Future Cancer Treatments
Overall, the study suggests that PMC works by binding to KDEL receptors, disturbing their location and function, and thereby sabotaging the cell’s ability to keep its protein inventory in order. Cancer cells, which already operate near the limit of their protein handling capacity, appear especially vulnerable to this kind of disruption and can be pushed into programmed death. While further work in animals and safety testing are needed, the results point to KDEL receptors and related protein stress pathways as promising entry points for new cancer drugs and for combinations that make existing stress‑based treatments more effective.
Citation: Yoshida, K., Nishi, K., Kitaguchi, T. et al. Pyra-metho-carnil disrupts cancer cell proteostasis and induces apoptosis by binding to KDEL receptors. Sci Rep 16, 15145 (2026). https://doi.org/10.1038/s41598-026-45604-z
Keywords: cancer cells, protein folding, endoplasmic reticulum stress, KDEL receptors, apoptosis