Clear Sky Science · en
Single-molecule dynamics of the TRiC chaperonin system in vivo
How Cells Keep New Proteins Out of Trouble
Every second, your cells crank out thousands of new proteins—tiny molecular tools that must fold into just the right shape to work. Misfolded proteins can clog the cell’s machinery and contribute to diseases ranging from neurodegeneration to developmental disorders. This study peeks inside living human cells at single molecules to watch, in real time, how a major cellular folding team, the TRiC chaperonin and its helper prefoldin, shepherds new proteins into the correct shape and decides what to do when folding goes wrong.
A Cellular Quality-Control Team at Work
Inside our cells, special helper proteins called chaperones guide other proteins as they are made and folded. TRiC is a large barrel-shaped complex that can temporarily enclose an unruly protein chain, while prefoldin is a smaller tentacle-like complex that delivers clients to TRiC. Earlier work, mostly in test tubes, showed that TRiC can fold important proteins such as actin and tubulin. But what happens in the crowded, bustling interior of a real cell—how often TRiC and prefoldin meet their clients, how long they hold them, and how they respond to difficult cases—has remained murky. The authors developed advanced single-particle tracking microscopy in living human cells to follow individual TRiC and prefoldin molecules and their encounters with newly made proteins.
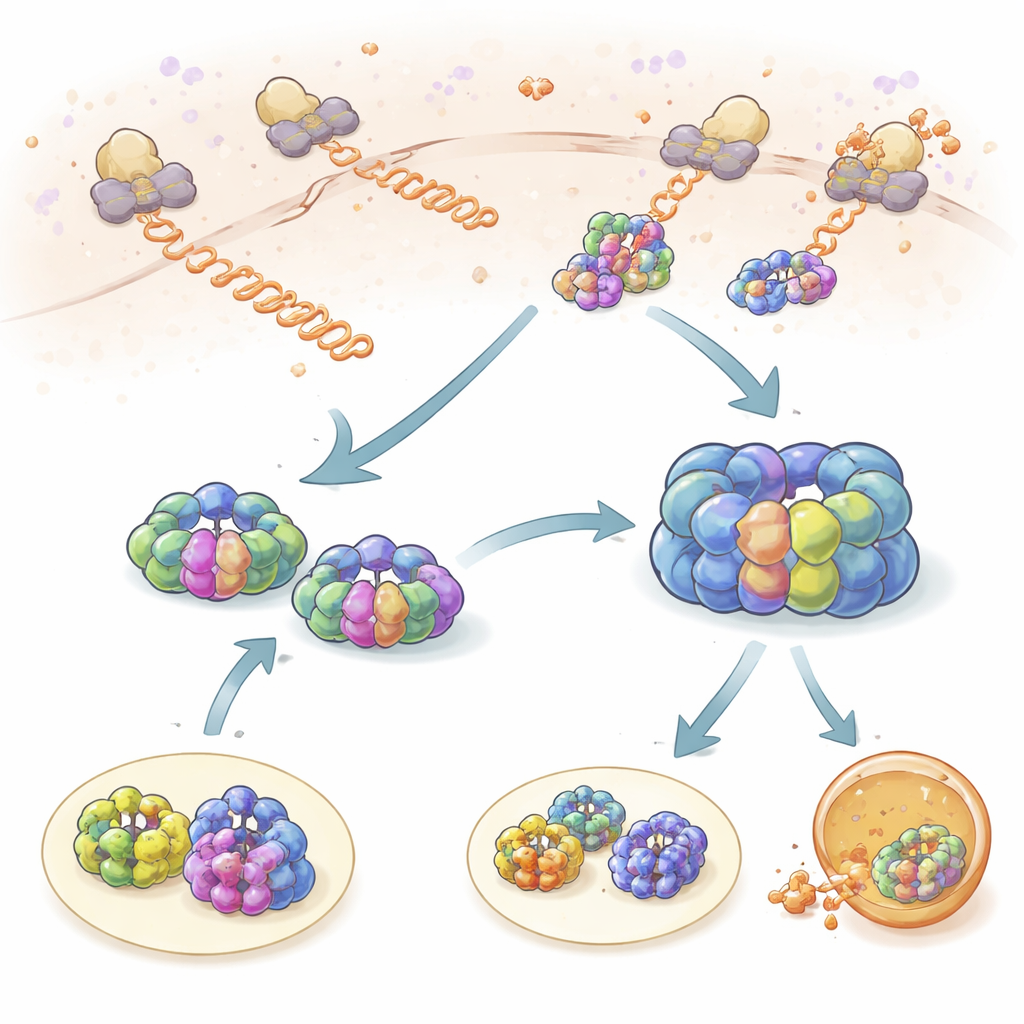
Brief Checkups While Proteins Are Being Made
The team first looked at how TRiC and prefoldin interact with freshly synthesized protein chains as they emerge from ribosomes, the cell’s protein factories. By tagging ribosomes, TRiC, and prefoldin with different fluorescent dyes, they could watch when their movements became linked in space and time. Both TRiC and prefoldin repeatedly touched nascent chains in short “probing” events lasting about one second. Prefoldin contacted new chains more often than TRiC, and reducing prefoldin levels sharply lowered TRiC’s access to these clients, showing that prefoldin acts as a recruiter. Using ribosome profiling, the authors found that hundreds of different proteins are monitored this way, with a strong overlap between TRiC and prefoldin clients, emphasizing that this partnership is central to maintaining a healthy protein population.
Lingering Engagement Near the Finish Line
To zoom in on a single obligate client, the researchers tracked actin, a key building block of the cell’s skeleton. Instead of tagging actin protein directly, they labeled its messenger RNA, allowing them to follow the sites where actin was being synthesized. As the actin chain grew longer, both prefoldin and TRiC visited more frequently and stayed longer. Near the end of translation, prefoldin often bound for several seconds, holding nearly complete actin chains and helping recruit TRiC for the next phase of folding. When the researchers artificially tethered full-length actin near the ribosome, they could see TRiC engage it in repeated cycles lasting about 2.5 seconds each, consistent with a multistep folding process. Prefoldin was crucial for the longer, more productive TRiC engagements, suggesting that it not only delivers clients but also tunes how TRiC works.
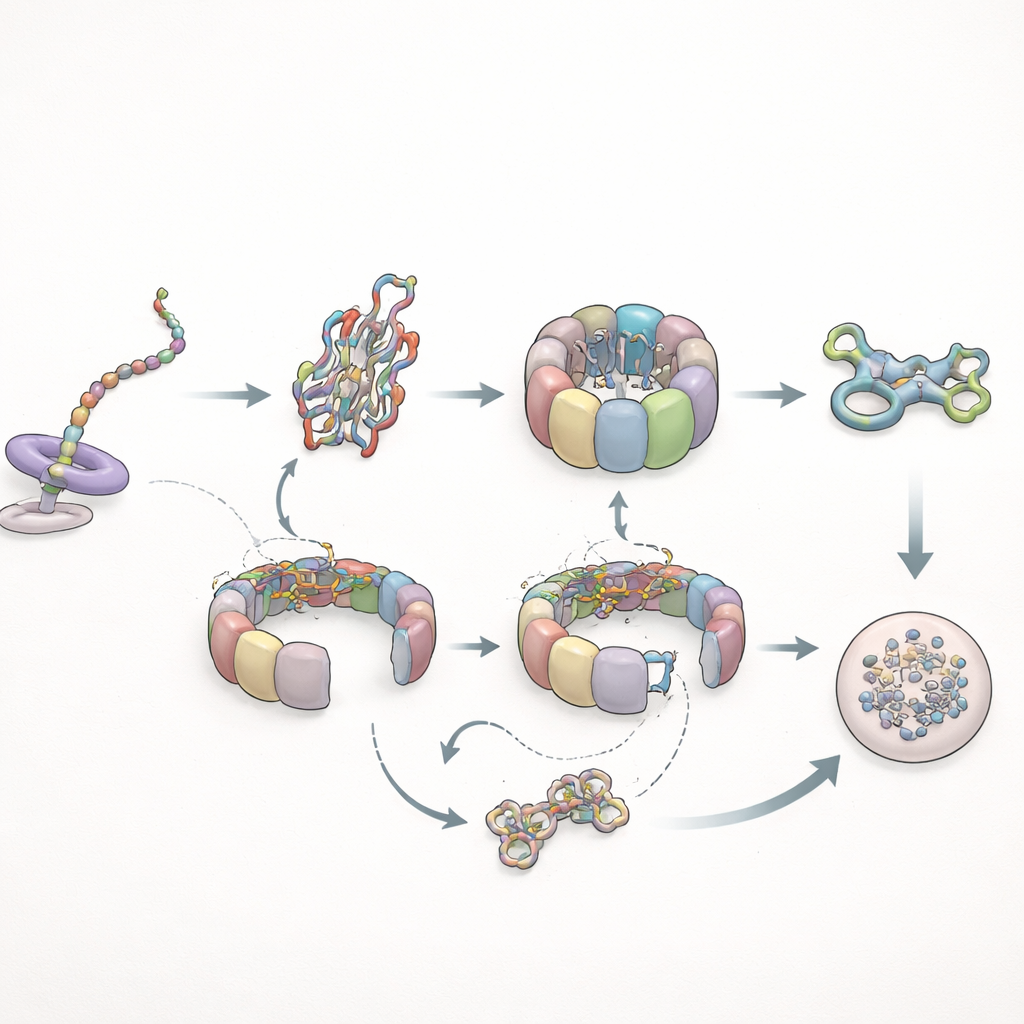
When Folding Fails, Cycling and Cleanup
The authors then tested how this system handles a problematic client: a mutant form of actin that is known to fold poorly. During protein synthesis, this mutant behaved almost normally with TRiC but failed to develop the prolonged prefoldin contacts seen for the healthy version, indicating that subtle conformational changes in the growing chain control how long prefoldin can hold on. After synthesis, the story changed dramatically. The mutant actin remained on TRiC much longer—up to several times the normal residence time—and cycled repeatedly through the chaperonin without reaching a stable folded state. Eventually, instead of joining the cell’s structural network, the mutant was passed on to the cell’s degradation machinery. In parallel, prefoldin’s interactions became more frequent but shorter, and its direct encounters with TRiC decreased, consistent with TRiC spending more time in a closed, encapsulating state around the misfolded client.
A Local Protective Zone for Folding
A striking observation was that TRiC and prefoldin often lingered near their client between binding rounds instead of diffusing away into the bulk cytoplasm. After releasing a protein, TRiC typically remained confined within a small region around the translation site for up to two seconds, then re-bound the same or a neighboring chain. The authors also detected transient contacts between different TRiC complexes that depended on ongoing protein synthesis. Together, these behaviors support the idea of a “protected folding zone”—a loose, dynamic cluster of chaperones and translation machinery in which newly made proteins are kept close to their helpers and shielded from harmful interactions until their fate is decided.
What This Means for Healthy Cells and Disease
In simple terms, this work shows that TRiC and prefoldin act as an agile quality-control duo: they briefly inspect many new protein chains, focus their attention as the chain nears completion, and then run rapid folding cycles within a small local bubble around the ribosome. Well-behaved proteins like normal actin generally reach their correct shape after multiple quick passes through TRiC. Faulty variants, by contrast, become trapped in extended cycles and are ultimately routed to degradation, tying up chaperone capacity along the way. These single-molecule insights help explain how cells manage the constant risk of misfolding in a crowded environment, and why mutations in either clients or chaperones can tip the balance toward disease.
Citation: Li, R., Dalheimer, N., Müller, M.B.D. et al. Single-molecule dynamics of the TRiC chaperonin system in vivo. Nature 652, 481–489 (2026). https://doi.org/10.1038/s41586-025-10073-3
Keywords: protein folding, molecular chaperones, TRiC chaperonin, actin dynamics, cellular proteostasis