Clear Sky Science · en
ZFTA–RELA ependymomas make itaconate to epigenetically drive fusion expression
Why this brain cancer story matters
Ependymomas are rare but aggressive brain tumors that often strike children and young adults. For many patients, surgery and radiation can hold the cancer at bay for a time but rarely cure it. This study uncovers a surprising weakness in one common subtype of these tumors: they depend on a little-known molecule, itaconate, to keep their main cancer-causing gene switched on. By tracing how the tumor makes and uses this molecule, the researchers reveal several new ways future drugs might shut these cancers down.
A dangerous fusion in the brain
More than half of ependymomas that arise in the upper part of the brain carry a genetic mash‑up called ZFTA–RELA. Alone, the two original genes are harmless, but when fused they create a potent cancer driver that moves into the cell nucleus and rewires how DNA is read. Until now, most work on ZFTA–RELA focused on its role in gene control. The new study asked a different question: does this fusion also reprogram the tumor’s metabolism, the network of chemical reactions that provide energy and building blocks, in ways that could be exploited for therapy?
A macrophage molecule turned to the dark side
Using mouse brain stem cells engineered to carry the fusion, patient‑derived tumor cells, and mouse models, the team profiled hundreds of metabolites. One stood out sharply: itaconate, a molecule normally produced by immune cells called macrophages during infection. The tumor cells made large amounts of itaconate through an enzyme named ACOD1, whose levels rose and fell in step with ZFTA–RELA. Knocking down ACOD1 or blocking it with small molecules was toxic to fusion‑positive cells and shrank tumors in mice, while adding extra itaconate rescued tumor growth. These results show that the cancer has co‑opted an immune‑cell pathway for its own survival. 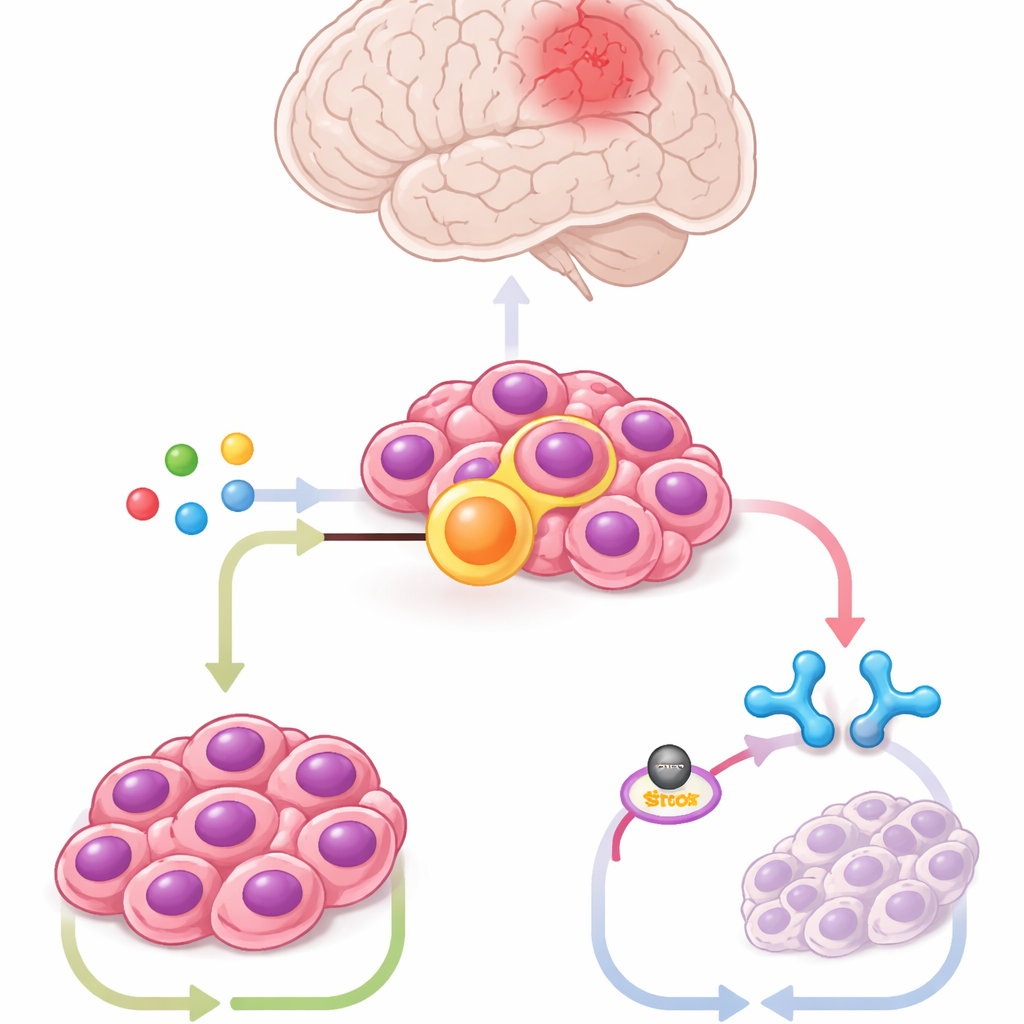
A self‑reinforcing loop in the tumor’s DNA control
Why would itaconate be so important? The answer lies in epigenetics—chemical marks on DNA‑packaging proteins that determine which genes are active. The authors discovered that itaconate inhibits a family of enzymes (KDM5) that normally remove an activating mark called H3K4me3 from histones. When KDM5 is blocked, H3K4me3 builds up, especially at key control regions of the ZFTA gene that are part of the ZFTA–RELA fusion. This increased marking boosts transcription of the fusion itself, creating a feed‑forward loop: ZFTA–RELA turns on ACOD1, ACOD1 makes itaconate, and itaconate in turn enhances expression of ZFTA–RELA. Disrupting ACOD1 or mimicking KDM5 activity broke this loop, lowered the fusion protein, and slowed tumor growth in multiple models.
Glutamine: the fuel behind the fuse
The study then traced where the tumor gets the raw material to make itaconate. By feeding cells forms of glutamine and glucose labeled with heavy carbon, the researchers showed that most of the carbon skeleton of itaconate comes from glutamine. ZFTA–RELA tumors overexpress glutamine transporters and the enzyme glutaminase, and they display activated PI3K–AKT–mTOR signaling, a pathway known to drive glutamine use. At the same time, the fusion epigenetically silences PTEN, a critical tumor suppressor that normally restrains this signaling. The result is a rewired metabolism that pulls in glutamine, channels it through the cell’s energy cycle, and diverts it into itaconate production, all to sustain high levels of the fusion driver. 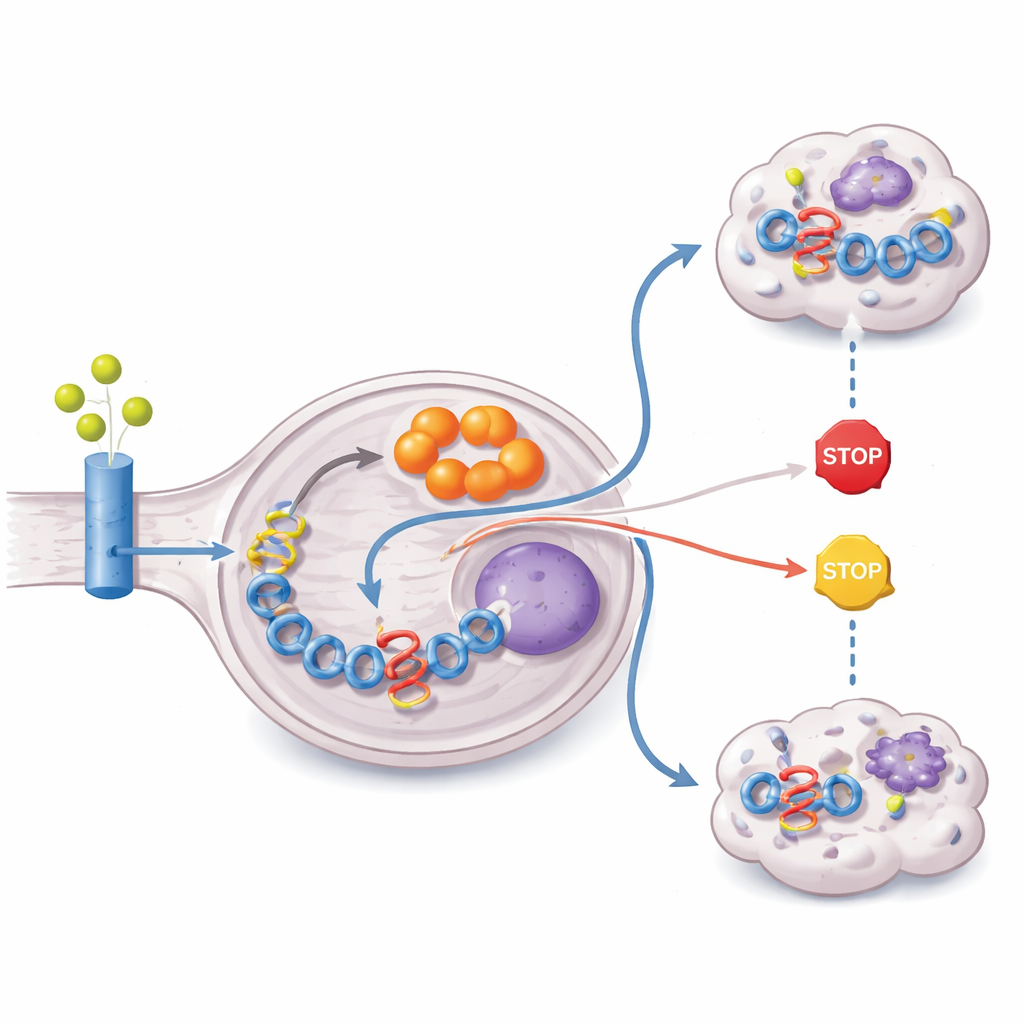
Hitting the tumor where it feeds
Because this circuit has several distinct steps, it offers multiple drug targets. The authors tested compounds that block glutamine import, glutaminase, ACOD1, or the PI3K–mTOR pathway, as well as a brain‑penetrant glutamine antagonist called JHU‑083. Fusion‑positive cells were far more sensitive to these agents than other ependymoma subtypes, and treated mice lived longer with smaller tumors. Particularly striking, blocking ACOD1 or combining glutamine antagonism with PI3K–mTOR inhibition not only reduced primary brain tumors but also prevented or eliminated spinal cord metastases in mouse models—lesions that are especially difficult to treat in patients.
What this means for patients and families
To a non‑specialist, the key message is that these ependymomas are addicted to a specific metabolic loop of their own making. They hijack an immune‑cell metabolite, itaconate, and a common nutrient, glutamine, to keep their chief cancer gene switched on. By mapping this loop from start to finish, the study identifies several existing or emerging drug types—targeting glutamine use, ACOD1, or PI3K–mTOR signaling—that could be combined to selectively weaken ZFTA–RELA tumors while sparing normal brain. Although these results are preclinical and not yet ready for routine treatment, they chart a clear path toward targeted therapies for a childhood brain cancer that currently lacks effective options.
Citation: Natarajan, S.K., Lum, J., Skeans, J.H. et al. ZFTA–RELA ependymomas make itaconate to epigenetically drive fusion expression. Nature 652, 1004–1015 (2026). https://doi.org/10.1038/s41586-025-10005-1
Keywords: ependymoma, brain tumor metabolism, itaconate, glutamine addiction, epigenetic therapy