Clear Sky Science · en
Peritumoural adipose tissue promotes ferroptosis resistance by 3-hydroxykynurenine-mediated suppression of ferritinophagy
Fat Around Tumors: A Hidden Protector
Many cancers grow in close contact with soft, cushiony fat. Far from being a passive bystander, this nearby fat can secretly help tumors survive both chemotherapy and modern immunotherapies. This study uncovers a surprising way in which fat surrounding tumors blocks a type of iron‑driven cell death inside cancer cells, and shows how disrupting this shield could make existing treatments work better, especially in abdominal cancers that spread across fatty surfaces.
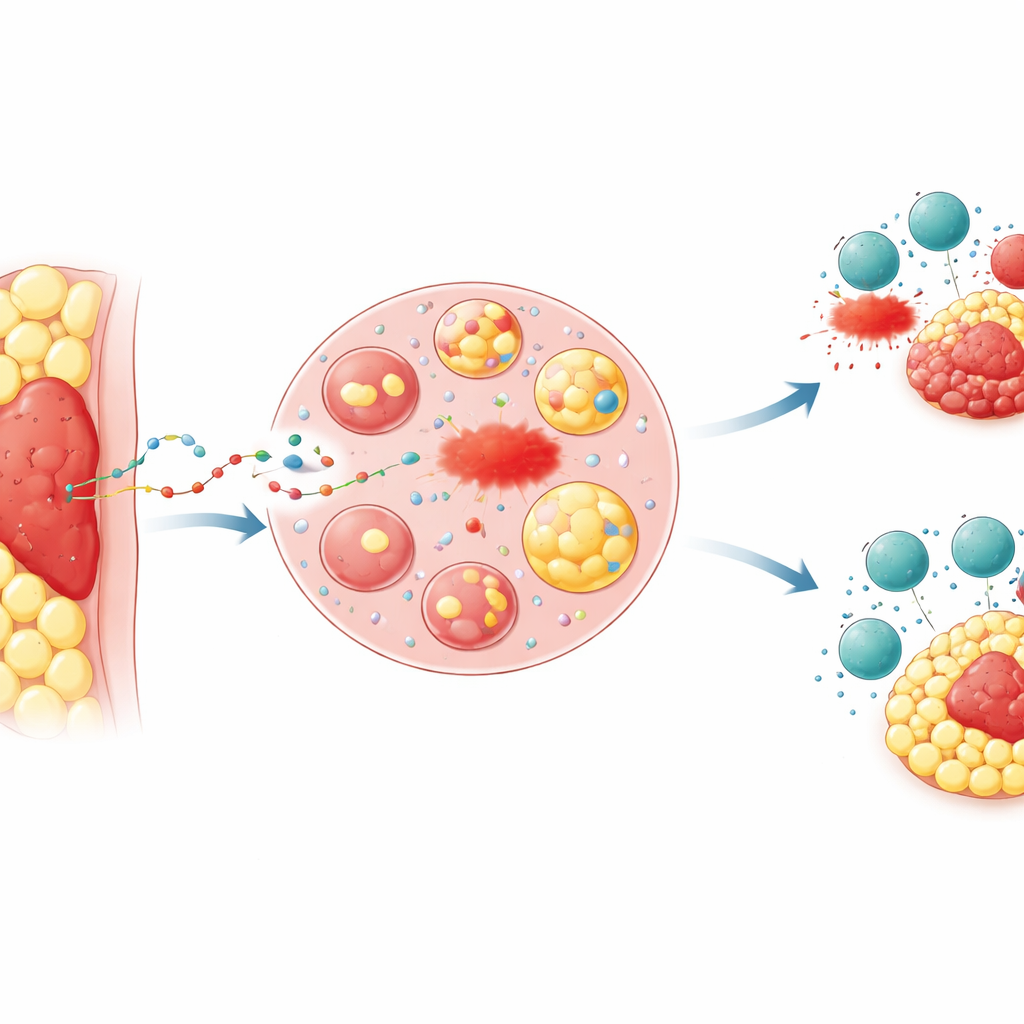
When Location Gives Tumors an Edge
Doctors have long noticed that tumors buried in fat, such as advanced colon and stomach cancers that invade the abdominal lining, often respond poorly to treatment and lead to worse outcomes. The researchers focused on the “peritumoural adipose tissue”—the ring of fat immediately surrounding a tumor. In mouse models, they implanted the same cancer cells either under the skin, where there is little fat, or directly into a fat pad. Tumors nestled in fat grew faster, were harder to kill with drugs that trigger a form of cell death called ferroptosis, and were less sensitive to immune attack from killer T cells or to anti‑PD‑1 immunotherapy. Chemical markers of lipid damage, which signal that ferroptosis is occurring, were much lower in fat‑embedded tumors, mirroring patterns seen in tissue samples from patients with advanced colorectal and gastric cancers.
Iron Lockboxes Inside Cancer Cells
Ferroptosis depends on free iron; when enough iron is available, it helps drive runaway oxidation of cell membranes, killing the cell. Cancer cells can buffer this threat by storing iron inside protein shells called ferritin. The team asked whether the nearby fat somehow rewired tumor iron handling. They grew cancer cells in liquid from human and mouse fat tissue and found that this “conditioned” medium made cells markedly more resistant to ferroptosis. Proteomic profiling revealed a strong increase in both main ferritin subunits, without major changes in other known ferroptosis regulators. At the same time, the pool of free iron inside cells actually fell. When the researchers knocked down ferritin, the protective effect of fat‑derived medium disappeared, and tumors in fat‑rich sites became smaller and more sensitive to iron‑driven damage.
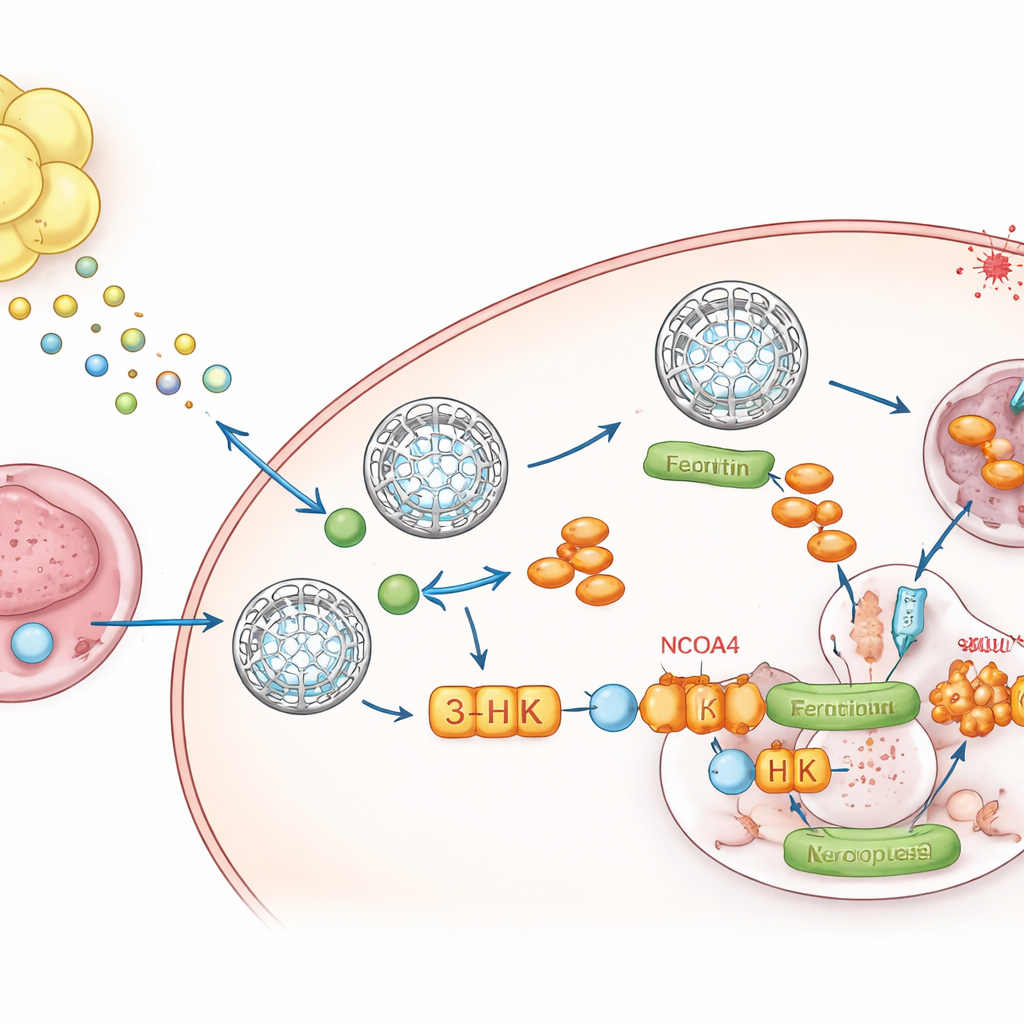
A Metabolic Messenger From Fat to Tumor
To pinpoint what the fat was sending, the team turned to metabolomics, a survey of small molecules. One compound, kynurenine, a breakdown product of the amino acid tryptophan, stood out as consistently elevated in fat‑conditioned media and in tumors growing within fat. The fluid bathing these tumors contained high kynurenine and low tryptophan, showing that the local environment is rewired. The surrounding fat expressed high levels of the enzymes IDO1 and TDO2 that make kynurenine, and single‑nucleus RNA sequencing showed that adipocytes and their precursor cells were the major sources. When cancer cells were exposed to kynurenine, they boosted ferritin levels, lowered free iron, and resisted ferroptosis. Blocking IDO1 and TDO2 in fat abolished this ferritin increase unless kynurenine was added back.
A Single Molecule That Blocks the Iron Release Valve
Kynurenine does not act alone. Inside tumor cells it is converted along a pathway into several metabolites. By selectively blocking individual steps, the authors discovered that one intermediate, 3‑hydroxykynurenine (3‑HK), is the key player. 3‑HK, even at low doses, stabilized ferritin, reduced iron release, and strongly protected cells from ferroptosis. Mechanistically, 3‑HK bound directly to a protein called NCOA4, which normally escorts ferritin to cellular “recycling centers” where it is broken down and iron is released. Structural and biophysical experiments showed that 3‑HK latches onto NCOA4 and prevents it from docking with ferritin, effectively freezing these iron lockboxes in place and starving ferroptosis of the metal it needs to ignite. When NCOA4 was removed, 3‑HK could no longer raise ferritin or defend cells against ferroptotic death.
Turning the Shield Into a Therapeutic Target
Because ferroptotic death of cancer cells can enhance antitumor immunity, the fat‑driven 3‑HK–ferritin pathway offers an explanation for why fat‑rich metastases, such as peritoneal implants from colon or stomach cancers, often resist immunotherapy. In multiple mouse models, dual inhibition of IDO1 and TDO2 lowered kynurenine and 3‑HK in the tumor–fat niche, reduced ferritin buildup, and made tumors much more vulnerable to ferroptosis inducers. Most strikingly, combining IDO1/TDO2 inhibitors with anti‑PD‑1 antibodies produced far stronger tumor control, including in peritoneal metastasis models, and this benefit was lost when ferroptosis was chemically blocked. Patient data supported these findings: peritoneal metastases showed higher ferritin and lower lipid damage than primary tumors, and gene signatures predicting abundant 3‑HK correlated with more advanced disease and poorer survival.
What This Means for Future Cancer Care
This work reveals a previously hidden conversation between fat and tumor in which the surrounding adipose tissue manufactures kynurenine, feeds it to cancer cells, and thereby raises 3‑HK levels that lock iron inside ferritin and block ferroptosis. In doing so, fat not only helps tumors withstand ferroptosis‑targeting drugs, but also blunts the ability of the immune system and checkpoint inhibitors to destroy them. By interrupting this kynurenine–3‑HK–ferritin axis, for example with combined IDO1/TDO2 inhibitors, clinicians may be able to strip away this protective shield and render fat‑embedded cancers far more responsive to immunotherapy and other treatments.
Citation: Zhang, YY., Han, Y., Tan, YT. et al. Peritumoural adipose tissue promotes ferroptosis resistance by 3-hydroxykynurenine-mediated suppression of ferritinophagy. Nat Cell Biol 28, 783–796 (2026). https://doi.org/10.1038/s41556-026-01907-x
Keywords: tumor microenvironment, ferroptosis, kynurenine pathway, peritumoural adipose tissue, cancer immunotherapy