Clear Sky Science · en
Reprogramming of bacterial virulence by lysine acetylation
How Bacteria Tune Their Attack
Many harmful bacteria live inside our cells and must avoid killing their host too quickly. This study uncovers how one such microbe fine‑tunes its aggression using a tiny chemical tag called acetylation. By decorating a key virulence protein with this tag, the bacterium can switch the protein’s shape, location, and activity, effectively dialing its offensive tools up or down to match the host cell’s energy state.
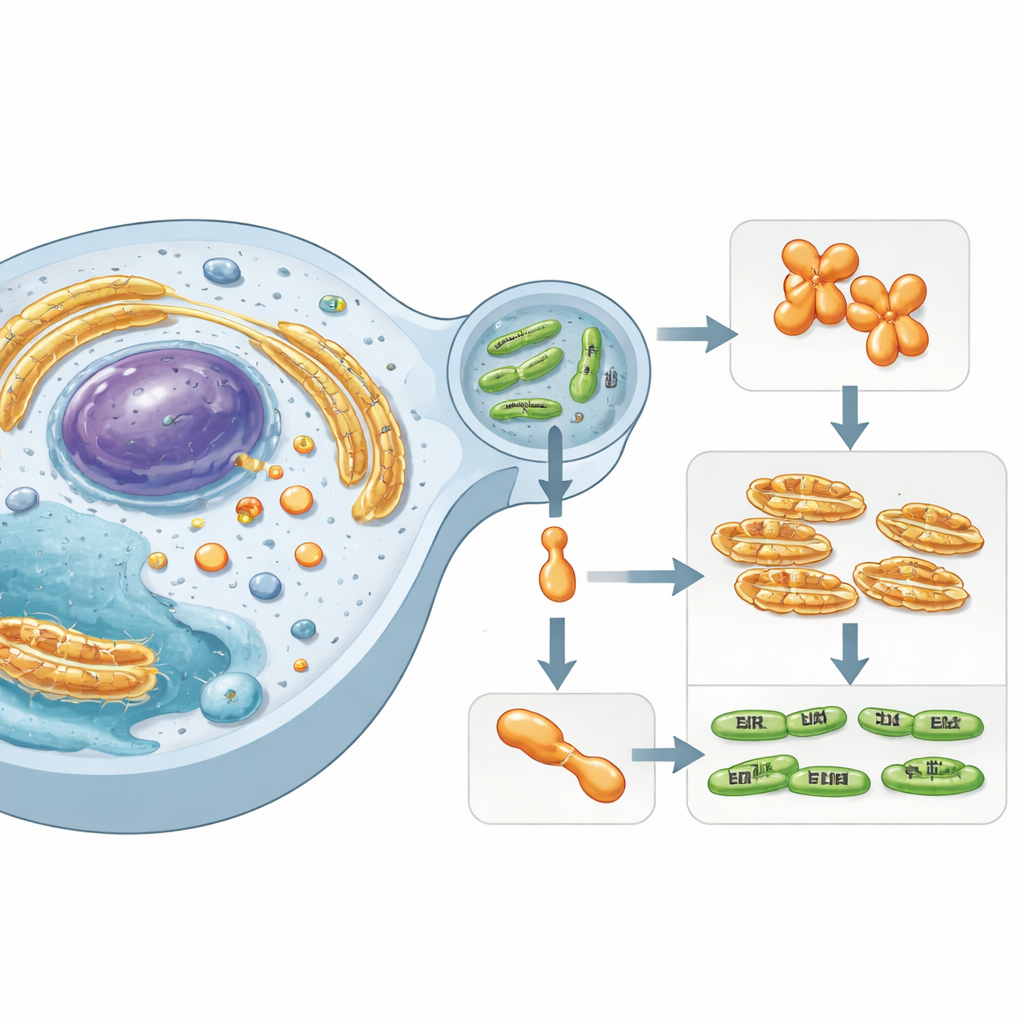
A Hidden Player Inside Human Cells
The work focuses on Simkania negevensis, a Chlamydia‑like bacterium linked to long‑lasting respiratory infections. This microbe survives inside a membrane‑bound bubble within human cells and injects specialized “effector” proteins into the surrounding cytoplasm. One of these effectors, called SnCE1, belongs to a family of enzymes known to interfere with the host’s protein‑tagging systems. These tags—small proteins such as ubiquitin and SUMO—act as cellular traffic signals, controlling immunity, stress responses, and cell death. Disrupting SUMO and ubiquitin chains allows bacteria to sabotage host defenses and create a more hospitable niche.
A Dual‑Function Molecular Switch
The researchers show that SnCE1 is a true multitasker. On one hand, it is an efficient “deSUMOylase”: it cuts SUMO chains into single units much more readily than it snips ubiquitin chains. High‑resolution crystal structures reveal how SnCE1 grips a SUMO molecule in a surface pocket, using precisely placed charged and hydrophobic regions to favor SUMO over ubiquitin. On the other hand, SnCE1 also behaves as an auto‑acetyltransferase: it takes acetyl groups from the cellular fuel molecule acetyl‑CoA and attaches them to several of its own lysine residues. Surprisingly, both activities use the same catalytic center, so the enzyme must choose between cutting SUMO and modifying itself.
Shaping, Clustering, and Cutting the Effector
Acetylation turns out to remodel how SnCE1 is assembled. When unmodified, a catalytically inactive version of the protein tends to form tetramers—clusters of four units. Structural analysis shows that the interfaces between subunits block the site where SUMO would normally bind, making this clustered form poorly suited for deSUMOylation. When specific lysines, especially one at position 231, are acetylated, this salt bridge‑based interface can no longer form, and SnCE1 exists as separate monomers. These single units have an open SUMO‑binding groove and show strong deSUMOylase activity. Importantly, acetylation at Lys231 appears irreversible under all tested human and bacterial deacetylases, locking SnCE1 into its active, monomeric state once this modification occurs.
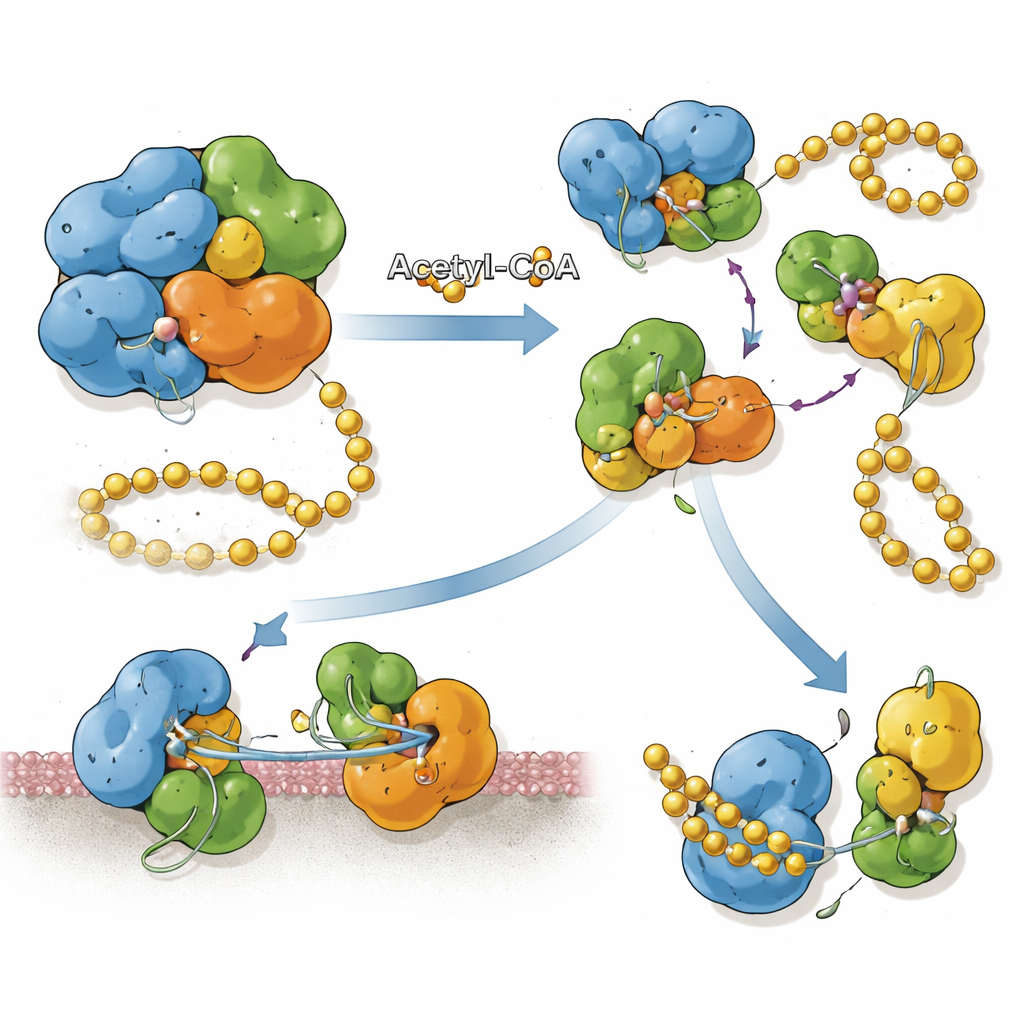
Linking Chemical Tags to Protein Position
SnCE1 is produced with an N‑terminal segment that behaves like a membrane anchor, helping it attach to the membrane of the bacterial vacuole and the nearby endoplasmic reticulum. The team found that when SnCE1 is heavily acetylated on several lysines in this N‑terminal region, the protein remains full‑length and membrane‑bound. When host enzymes called sirtuins remove some of these acetyl marks, SnCE1 undergoes a self‑cleavage event that trims off roughly 10 kilodaltons from its N‑terminus, including the membrane anchor. This autoproteolytic processing releases SnCE1 into the surrounding cytosol and toward mitochondria. Site‑specific acetylation experiments show that no single lysine controls this step; instead, multiple acetylated sites collectively slow or prevent the cut.
Impact on Mitochondria and Host Metabolism
Inside human cells, full‑length SnCE1 concentrates at the endoplasmic reticulum and at contact points with mitochondria, where it coincides with striking changes in mitochondrial shape. Whether catalytically active or not, SnCE1 expression promotes fragmentation of the normally tubular mitochondrial network, with the strongest effect seen in a non‑acetylated, monomeric mutant that is readily processed. Because mitochondrial dynamics are tightly linked to energy production and cell survival, this remodeling likely influences how hospitable the cell is for bacterial growth. The authors propose that the balance of acetyl‑CoA and NAD+ in the host cell—chemical readouts of metabolic state—governs SnCE1 acetylation, deacetylation, and processing, thereby tuning its activity and localization in space and time.
A New Layer of Control in Bacterial Virulence
In everyday terms, this study shows that Simkania uses acetylation as a dimmer switch on one of its key weapons. When acetyl groups are abundant, SnCE1 becomes a stable, active monomer that efficiently erases SUMO tags, helping the bacterium rewire host signaling while remaining attached to membranes. When host deacetylases are engaged, SnCE1 is trimmed and released, redistributing toward mitochondria and further reshaping the cell’s internal landscape. By tying virulence to small chemical tags that track cellular metabolism, the bacterium can adjust how aggressively it manipulates its host, revealing a sophisticated strategy that may be shared by other intracellular pathogens.
Citation: Schmöker, O., Girbardt, B., Schulze, S. et al. Reprogramming of bacterial virulence by lysine acetylation. Nat Commun 17, 3859 (2026). https://doi.org/10.1038/s41467-026-72244-8
Keywords: bacterial virulence regulation, protein acetylation, SUMO deconjugation, host–pathogen interactions, mitochondrial dynamics