Clear Sky Science · en
An in vitro approach for simulating divergent Golgi O-glycosylation of tumor-associated MUC1 from normal MUC1
Why sugar coats on proteins matter in cancer
Our cells constantly decorate their proteins with small chains of sugars, forming a kind of molecular "coat" that affects how cells grow, communicate, and interact with the immune system. In many cancers, this sugar coating changes in distinctive ways, especially on a protein called MUC1 that is abundant on epithelial cells and tumor surfaces. This study builds a laboratory model that mimics how cells add these sugars, allowing scientists to see in detail how normal sugar patterns on MUC1 diverge into tumor-associated forms that help cancers grow and evade detection.
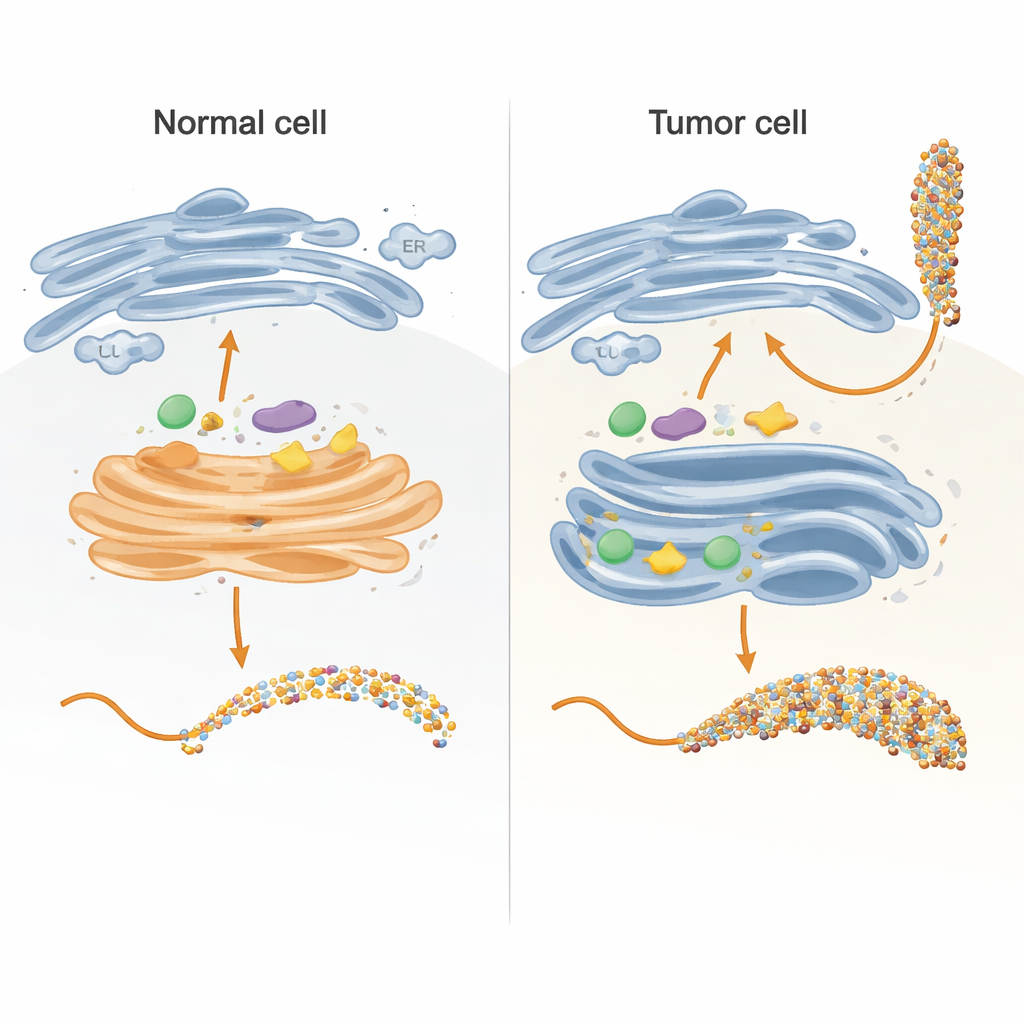
Building a controllable model of the cell’s sugar factory
Inside cells, sugar chains are built mainly in two compartments: the endoplasmic reticulum (ER) and the Golgi apparatus, which act like a multi-step assembly line. The authors recreated key parts of this line in a test tube using a carefully engineered version of the MUC1 protein attached to a carrier tag. They expressed this fusion protein in bacteria, purified it, and then exposed it to human enzymes that normally add sugars inside cells. By designing the MUC1 fragment so that each of its five sugar-attachment sites appears only once and in a natural sequence, they could track exactly which site each enzyme modified, and in what order, using sensitive kinetic measurements and mass spectrometry.
How normal and tumor pathways split apart
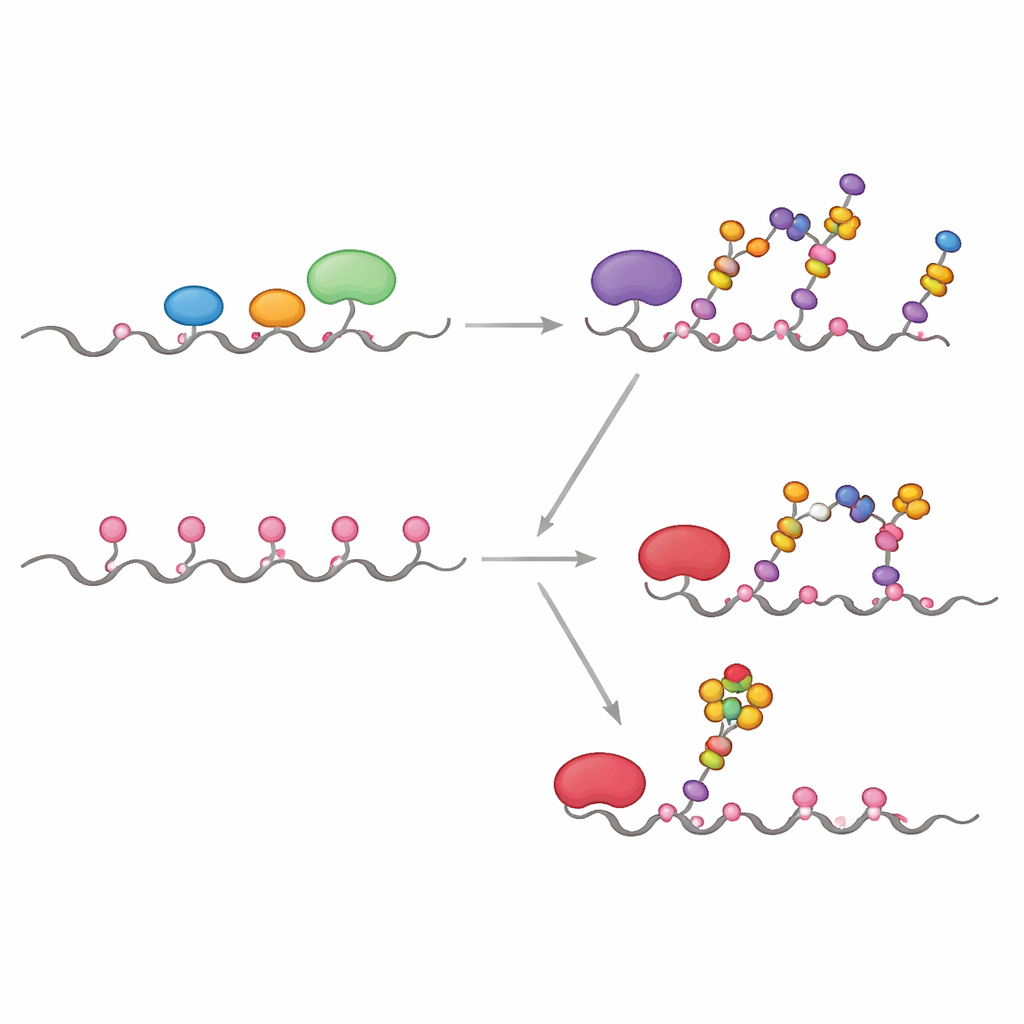
In healthy cells, the first enzymes in the process, called GALNTs, mostly reside in an early part of the Golgi and work side-by-side with other enzymes that extend or cap the initial sugar units. The team showed that under these "co-localized" conditions, GALNTs primarily modify a subset of sites on MUC1—what the authors call direct sites—while two slower, lectin-dependent sites remain mostly untouched. As soon as the first sugar (GalNAc) is added at the direct sites, downstream enzymes such as C1GALT1 quickly extend them, or ST6GALNAC1 and other sialyltransferases add sialic acid, effectively blocking GALNTs from using their lectin-based mechanism to densely fill all remaining sites. The result is a more modest, controlled sugar pattern typical of normal epithelial MUC1.
Simulating the cancer-like reshuffling of enzymes
Many tumors show a striking relocation of GALNT enzymes from the Golgi back to the ER, along with changes in the levels of several glycosylation enzymes. The researchers mimicked this by letting GALNTs act first, in isolation, for longer periods before introducing the downstream enzymes. Under these conditions, the initial sugars gradually spread from the easy direct sites to the slower lectin-dependent sites, eventually occupying all five positions on MUC1. Only after this saturation step were extension and capping enzymes added. This ER-like scenario yielded a highly decorated MUC1 resembling tumor-associated MUC1, rich in the so-called Tn and sialyl-Tn (sTn) antigens known to correlate with cancer progression, immune evasion, and metastasis.
Zeroing in on a single hot spot site
One of the most clinically important features of tumor MUC1 is the presence of sialyl-Tn, a short, sialic-acid–capped sugar attached directly to GalNAc. To see exactly where this appears on MUC1, the team combined kinetic experiments, computer docking, and hybrid quantum/classical simulations. They found that the enzyme ST6GALNAC1 is uniquely responsible for adding sialic acid in an α2-6 linkage to Tn and T antigens on MUC1, and that it shows a strong preference for one particular threonine site in the PDTR region (called T13 in the paper) once all five sites carry GalNAc. Structural models and reaction-path simulations reveal that at this site the sugar and catalytic amino acids align in a geometry that lowers the energy barrier for the reaction more than at neighboring sites, explaining why sTn appears there first and most strongly.
What this means for understanding and targeting cancer
Together, these experiments and simulations show that the difference between normal and tumor-associated MUC1 is driven less by which enzymes are present and more by where they sit in the cell and how long they can act before being interrupted. When GALNTs are confined to the Golgi with competing enzymes, they mostly decorate only the fast sites, giving rise to typical core 1 sugars and limited sialylation. When they are relocated to the ER, as in many cancers, they gain time to fill both fast and slow sites, creating a densely GalNAc-covered MUC1 that becomes an especially good substrate for ST6GALNAC1 at the T13 site, sharply boosting sTn levels. This systems-level view links enzyme localization, reaction timing, and molecular geometry into a single framework that helps explain how a seemingly subtle change in the cell’s sugar factory can generate potent tumor-specific markers and suggests more precise targets for diagnostics, vaccines, and glycan-focused cancer therapies.
Citation: Nashed, A., Dilsook, K., Senapathi, T. et al. An in vitro approach for simulating divergent Golgi O-glycosylation of tumor-associated MUC1 from normal MUC1. Nat Commun 17, 3619 (2026). https://doi.org/10.1038/s41467-026-72151-y
Keywords: protein glycosylation, MUC1, cancer biomarkers, Golgi apparatus, sialyl-Tn antigen