Clear Sky Science · en
Engineering of genetically encoded programmable calcium channel inhibitory binders
Turning Down Cellular Volume Knobs
Many of our cells rely on tiny bursts of calcium to decide when to move, divide, or unleash an immune attack. When this calcium signaling runs too hot, it can fuel rare genetic disorders, chronic inflammation, and even limit the power of cancer-fighting T cells. This study introduces a set of tailor‑made protein tools called CRABs that act like programmable dampers on a major calcium entry route in cells, opening new possibilities for both basic research and future therapies.
How Cells Use Calcium as an On–Off Switch
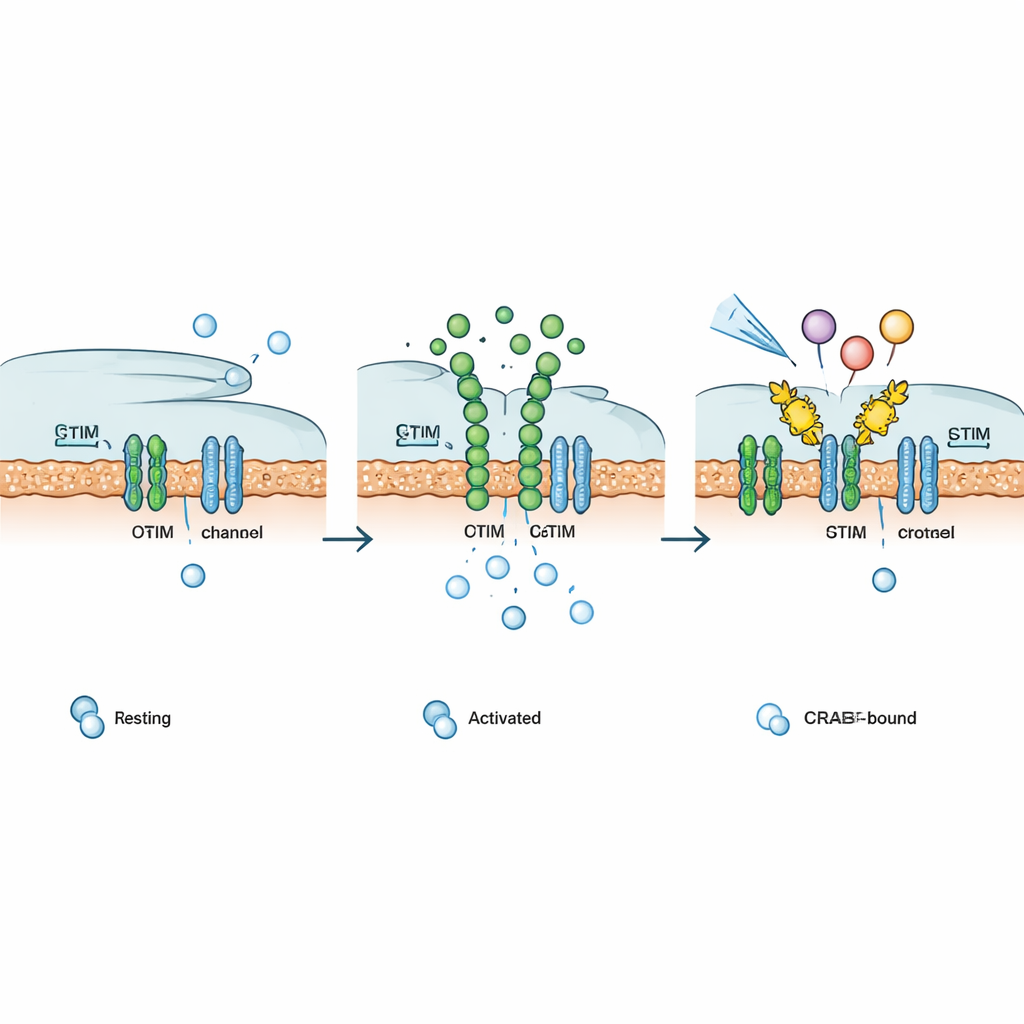
In most non‑nerve cells, a main doorway for calcium is a structure known as the CRAC channel, formed by two partners: STIM, which senses calcium levels inside an internal storage compartment, and ORAI, a pore in the outer cell membrane. When the internal stores run low, STIM changes shape, moves to regions where the internal and outer membranes touch, and latches onto ORAI. This opens the channel, allowing calcium to rush in and trigger responses ranging from short‑term movement to long‑term changes in gene activity. In immune cells, this pathway is vital for activating transcription factors such as NFAT that turn on genes needed for defense, but if left unchecked it can drive disease.
When Calcium Signaling Goes Wrong
Inherited changes that permanently flip STIM or ORAI into an “on” position can cause multi‑system disorders, such as Stormorken syndrome, marked by low platelet counts, bleeding problems, muscle weakness, and other issues. Milder but chronic overactivation of the same pathway contributes to autoimmune disease, cancer progression, and exhaustion of engineered CAR‑T cells used in leukemia therapy. Drug developers have tried to block CRAC channels with small molecules, but many of these compounds hit other ion channels and organs, leading to side effects and stalled clinical progress. A more specific, adjustable, and genetically targeted way to rein in CRAC activity has been lacking.
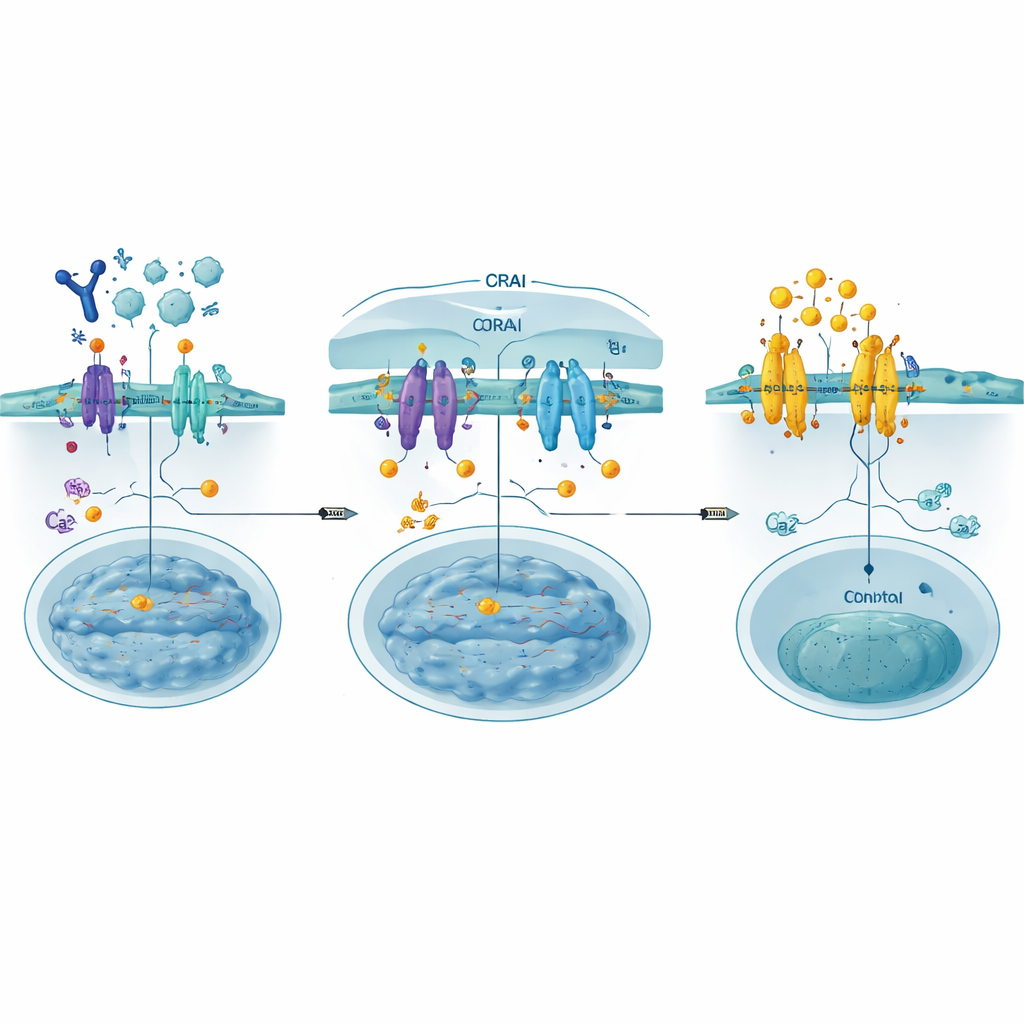
Building Decoys to Block the Gate
The authors designed CRABs (CRAC channel inhibitory binders) as short, genetically encoded protein fragments that mimic part of ORAI where STIM normally binds. Anchored at the cell membrane, these decoys catch activated STIM before it can latch onto real ORAI channels, effectively closing the gate upstream of calcium flow. Using deep mutational scanning—systematically testing hundreds of small sequence changes—they identified a particularly strong binder, dubbed PM‑CRAB, that potently reduced calcium entry and NFAT movement into the nucleus in human cells. Structural modeling suggested that a single amino‑acid swap makes the CRAB peptide fit more snugly into STIM’s binding groove, stabilizing the partnership and improving its blocking power.
From Fish Platelets to Programmable Dimmers
To show that CRABs work in living animals, the team used zebrafish engineered to carry a disease‑causing STIM1 mutation that keeps CRAC channels open and mimics Stormorken syndrome. These fish have markedly fewer fluorescently labeled thrombocyte (platelet‑like) cells. When PM‑CRAB was co‑expressed, the production of these cells largely recovered, indicating that the decoy could counter the harmful calcium overload. The researchers then created a family of CRAB variants with different strengths: monomers, dimers, and tetramers that progressively increased channel inhibition. Crucially, they also built versions that can be switched on by blue light (Opto‑CRAB) or by small molecules such as rapamycin or a non‑immunosuppressive analog (Chemo‑CRAB). In cultured cells, these switches allowed rapid, reversible control of calcium entry and NFAT activation in response to light or drug timing.
Tuning Immune and Signaling Pathways on Demand
Because many surface receptors in the body—such as certain hormone, growth factor, and G‑protein‑coupled receptors—ultimately rely on CRAC channels to sustain calcium signals, the authors tested whether Chemo‑CRAB could intervene across diverse pathways. Once activated by its chemical trigger, Chemo‑CRAB blunted calcium responses driven by engineered light‑ or caffeine‑gated calcium actuators, synthetic and natural growth factor receptors, and several distinct G‑protein‑coupled receptors. In T cells, a membrane‑anchored CRAB abolished CRAC currents and sharply lowered NFAT‑dependent gene activity. In human CAR‑T cells targeting CD19‑positive cancer cells, PM‑CRAB reduced the release of key inflammatory cytokines, while Chemo‑CRAB enabled dose‑dependent, on‑demand dampening of activation, hinting at a way to limit exhaustion and toxicity without permanently disabling the cells.
What This Could Mean for Future Therapies
To a non‑specialist, CRABs can be thought of as highly specific, genetically encoded volume knobs for calcium signaling. By acting at the precise handshake between STIM and ORAI, they shut down a central calcium entry route without broadly interfering with other channels or upstream receptors. The work demonstrates that these tools can correct disease‑like traits in zebrafish, dissect complex signaling networks in cells, and modulate engineered immune cells in a programmable way. While challenges remain—such as safely delivering the genes and avoiding excessive long‑term blockade in healthy tissues—the study lays out a versatile platform that could eventually help treat rare calcium channel disorders and fine‑tune powerful but delicate therapies like CAR‑T cells.
Citation: Liu, X., Ali, S., Lan, TH. et al. Engineering of genetically encoded programmable calcium channel inhibitory binders. Nat Commun 17, 3472 (2026). https://doi.org/10.1038/s41467-026-71769-2
Keywords: calcium signaling, CRAC channels, optogenetics, chemogenetics, CAR-T cells