Clear Sky Science · en
Accelerating Leigh syndrome drug discovery through deep learning screening in brain organoids
Finding New Hope for a Devastating Childhood Brain Disease
Leigh syndrome is a rare, often fatal disorder that strikes babies and young children, robbing their brains of the energy needed to grow and function. Families currently have no proven treatments, only supportive care. This study brings together cutting‑edge biology and artificial intelligence to search for existing drugs that might help. By combining computer‑guided predictions with tiny lab‑grown brain models, the researchers identify two medicines that partially restore brain‑like activity in models of Leigh syndrome, pointing to a faster way to discover therapies for rare childhood diseases.
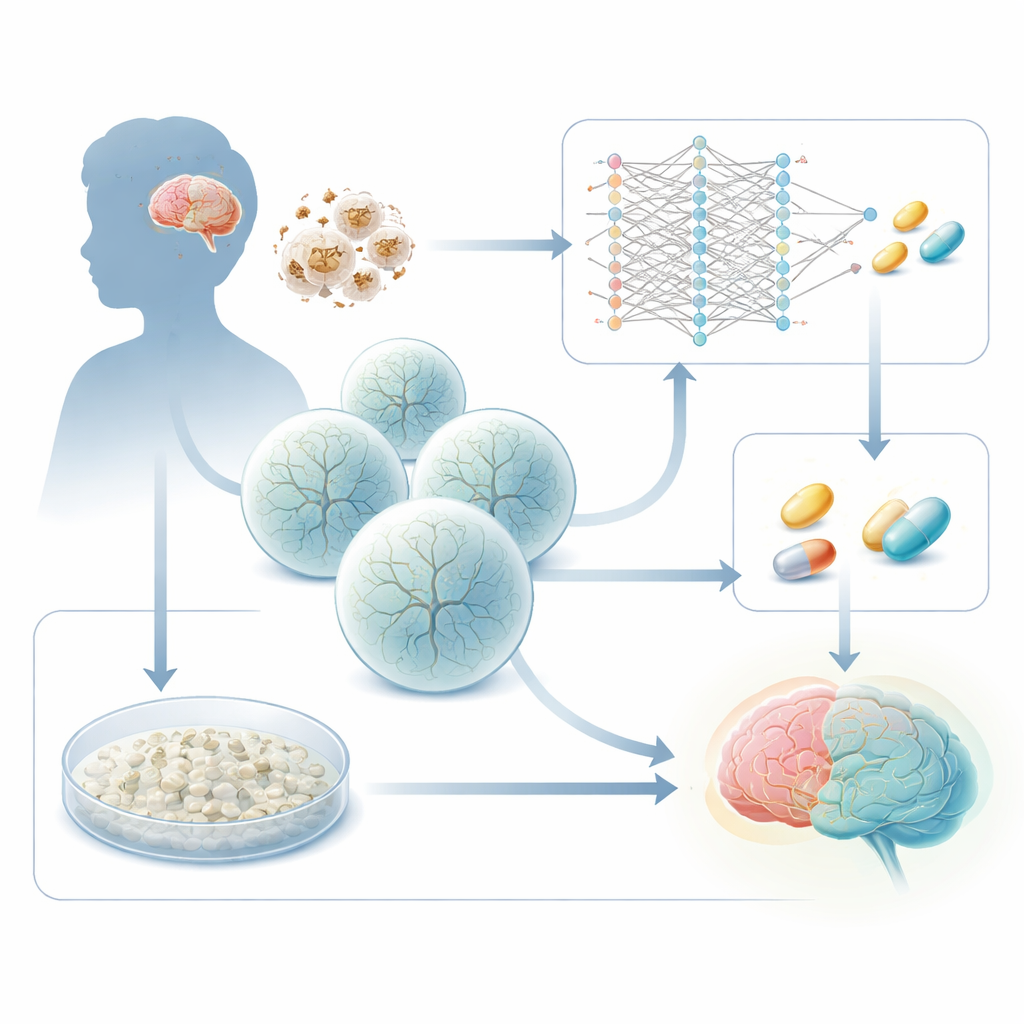
How Energy Failure Harms the Developing Brain
Leigh syndrome is caused by inherited faults in genes that power mitochondria, the cell’s “batteries.” One common culprit is a gene called SURF1, which is needed for a key step in the cell’s energy‑producing machinery. When SURF1 is defective, brain regions that demand a lot of energy—especially deep structures in the midbrain and basal ganglia—begin to fail. Earlier work from this group used patient‑derived stem cells to grow 3D brain organoids and showed that Leigh organoids form neurons poorly, with abnormal branching and wiring. The new study asks a practical question: can these disease‑specific changes be exploited as a read‑out to find drugs that push sick cells back toward normal development?
Teaching Computers to Suggest Helpful Drugs
One arm of the project uses a deep learning framework built specifically for non‑cancer cells. Instead of simply matching disease gene signatures to a catalog of drug responses, the algorithm learns how gene‑control networks shift as young brain cells progress from stem‑like radial glia, to intermediate progenitors, to mature neurons. Using single‑cell RNA sequencing from Leigh and healthy cerebral organoids, the model infers which transcription factors must be nudged to move diseased cells along the normal developmental path. It then searches a large database of drugs and predicts which compounds should “steer” the network from a Leigh‑like state toward a healthier neuronal state. From thousands of candidates, it narrows the list to a few dozen drugs with promising effects on cellular metabolism.
Letting Yeast and Mini‑Brains Vote on the Best Candidates
In parallel, the team runs a very different screen in yeast. Baker’s yeast has its own version of the SURF1 gene, and deleting it makes the cells vulnerable when nutrients are scarce—an echo of the energy stress in Leigh syndrome. The researchers expose SURF1‑deficient yeast to 2,250 already‑approved drugs and measure which chemicals best rescue survival. Intriguingly, both the artificial‑intelligence screen and the yeast survival test point to the same chemical family: azoles, a group better known as antifungal and dermatology drugs. From these, the team focuses on two compounds, talarozole and sertaconazole, that appear safe enough for long‑term exposure in human cell models and that strongly improve early neuron formation in Leigh‑derived neurons in a dish.
Testing the Drugs in Human Midbrain Organoids
To move closer to the affected brain regions in patients, the researchers generate midbrain organoids from patient‑derived neural progenitor cells. These 3D structures contain dopamine‑producing neurons, support cells, and developing circuits. Compared with organoids from healthy cells, Leigh organoids grow larger but have poorer neuronal structure, release more lactic acid into the culture medium, and respond abnormally when subjected to acute energy stress. Treating Leigh midbrain organoids for several weeks with low doses of talarozole or sertaconazole slows their excessive growth, reduces lactate release by about one‑fifth, and improves some measures of neuronal function. Talarozole, in particular, increases the number of dopamine‑like neurons and restores a stronger calcium signal when the organoids are challenged with sudden energy deprivation, suggesting a partial recovery of network responsiveness.
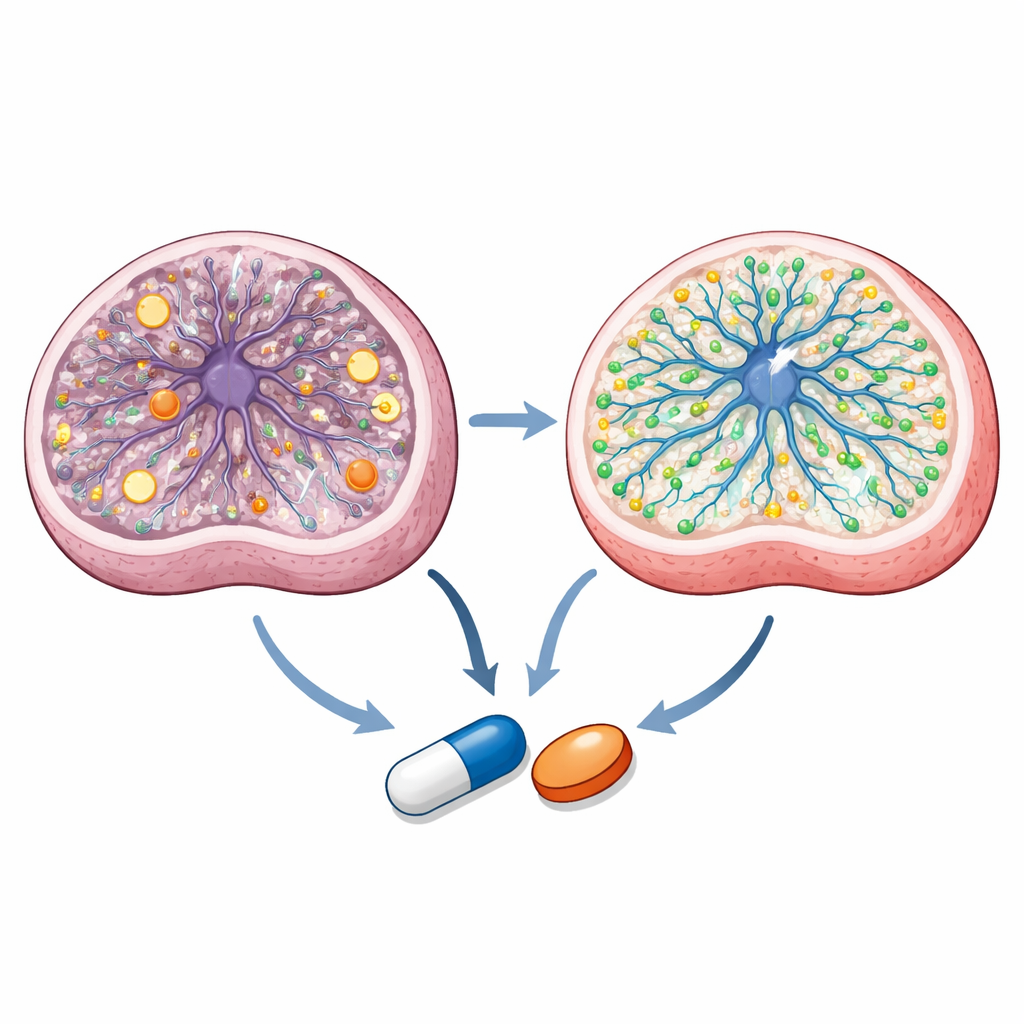
Uncovering How the Compounds Work Inside Cells
Digging deeper, the team finds that the two azole drugs act through overlapping but distinct routes. Detailed metabolite measurements show that Leigh neural progenitor cells have an imbalanced energy state and disrupted mitochondrial chemistry. Talarozole normalizes key energy ratios and several intermediates in the cell’s central metabolic cycle, while both drugs correct specific metabolic bottlenecks. Lipid profiling and imaging reveal that sertaconazole has a strong impact on membrane‑associated fats and cholesterol, restoring certain sphingolipids and increasing membrane‑bound cholesterol that is reduced in Leigh cells. Single‑cell RNA sequencing of treated organoids indicates that both drugs promote gene programs linked to neuron generation and lipid metabolism while dialing down overactive mitochondrial gene expression. Computational docking and live‑cell reporter assays further suggest that talarozole engages enzymes that break down retinoic acid—a vitamin A derivative crucial for brain development—and activates a nuclear receptor called PPARγ, which has been implicated as a therapeutic lever in mitochondrial diseases.
What This Could Mean for Children with Leigh Syndrome
Although talarozole and sertaconazole do not fully reverse all disease features in these models, they consistently soften several hallmarks of Leigh syndrome across yeast, 2D neurons, and 3D human midbrain organoids. The work shows that combining deep learning with human organoids and simple yeast screens can rapidly home in on repurposable drugs for a rare pediatric disorder that currently lacks treatments. For families, this does not yet translate into an approved therapy, but it provides two concrete drug candidates and, perhaps more importantly, a general strategy: use advanced computation to filter huge drug libraries, then test the most promising hits in realistic human mini‑brain models. This approach may shorten the road from basic biology to meaningful treatment options for Leigh syndrome and other neurodevelopmental diseases rooted in mitochondrial dysfunction.
Citation: Menacho, C., Okawa, S., Álvarez-Merz, I. et al. Accelerating Leigh syndrome drug discovery through deep learning screening in brain organoids. Nat Commun 17, 3570 (2026). https://doi.org/10.1038/s41467-026-71391-2
Keywords: Leigh syndrome, brain organoids, deep learning, drug repurposing, mitochondrial disease