Clear Sky Science · en
AcuB senses cellular energy charge to coordinate acetyl-CoA synthesis in bacteria
How Bacteria Decide When to Make Cellular Fuel
Bacteria live in constantly changing environments, where food and energy supplies can swing from feast to famine. To survive, they must carefully manage a central fuel molecule called acetyl-CoA, which feeds many vital processes. This study uncovers how a bacterial protein named AcuB acts as an internal "energy meter," helping cells decide when to turn acetyl-CoA production on or off so that they neither waste resources nor starve. 
A Molecular Switchboard for Acetate Use
The work focuses on the soil bacterium Bacillus subtilis and its relatives, which can use the simple molecule acetate both as a waste product and as a nutrient. One enzyme, AcsA, converts acetate into acetyl-CoA, feeding growth when preferred sugars like glucose are scarce. Another route, involving two enzymes called Pta and Ack, pushes the reaction in the opposite direction, turning acetyl-CoA back into acetate and releasing it from the cell. Running both directions at once would be wasteful, so the cell must coordinate these pathways. Earlier studies showed that AcsA can be turned off by attaching a small chemical tag (an acetyl group) and turned back on when that tag is removed, but how these two opposing steps were coordinated was not known.
The Hidden Role of AcuB Revealed
Right next to the acsA gene sits a cluster of three genes, called the acu operon, encoding proteins AcuA, AcuB, and AcuC. AcuA places the acetyl tag on AcsA, inactivating it, while AcuC removes the tag and reactivates the enzyme. AcuB’s role had remained mysterious. In this study, the authors produced AcuB proteins from several bacterial species in the lab and analyzed their behavior. They discovered that AcuB naturally binds small energy-carrying molecules—AMP, ADP and ATP—as well as a stress signal molecule called Ap4A. Using biochemical tests, they showed that when AcuB is loaded with AMP, it strongly binds to AcuC and shuts down its deacetylase activity, preventing AcsA from being reactivated. 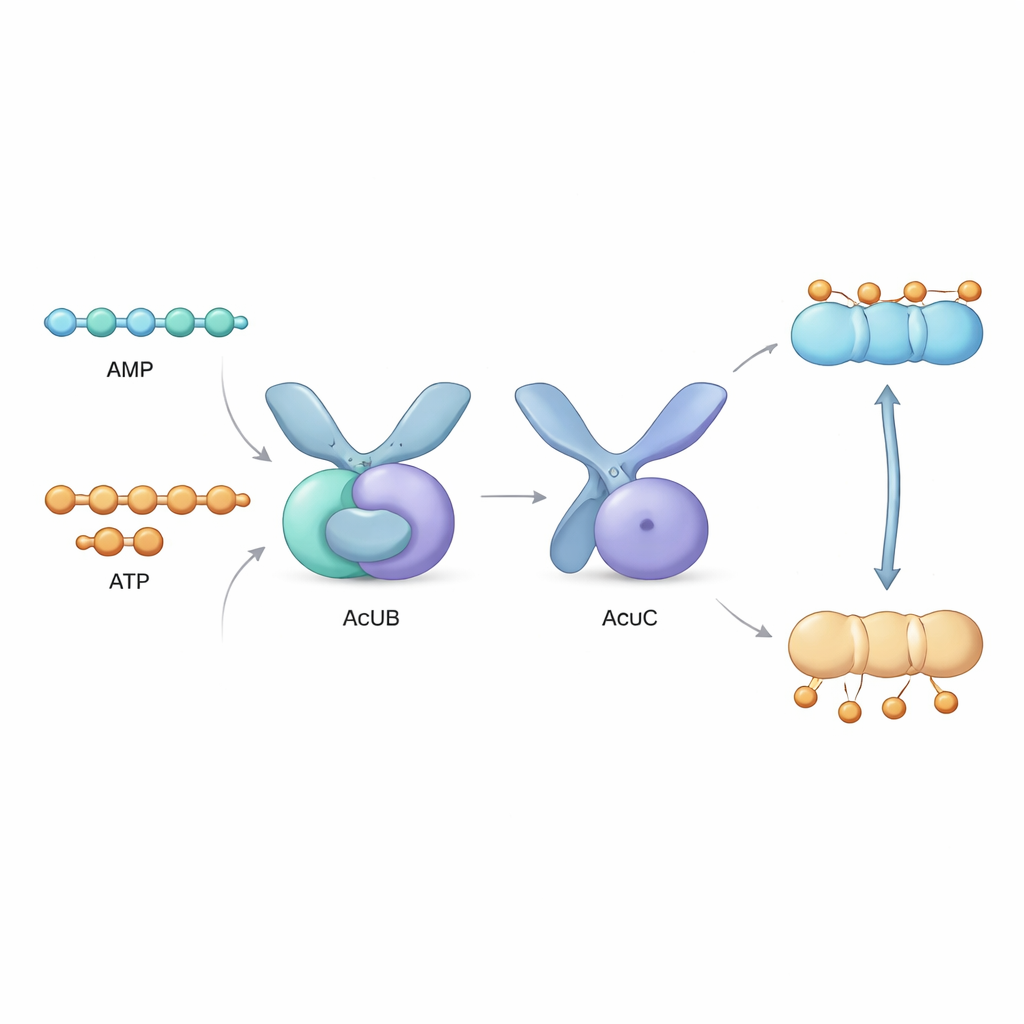
Energy Reading by Shape-Shifting Proteins
To see how this works at the atomic level, the team used X-ray crystallography to solve the three-dimensional structures of AcuB from two bacteria. AcuB forms a scissor-like pair of protein units, each with a twin-pocket “sensor” section that binds adenine-based nucleotides and a “handle” section that contacts AcuC. Four nucleotide molecules can slot into each AcuB dimer. The researchers observed how these pockets are lined with positively charged amino acids that interact differently with AMP, ADP, ATP, or Ap4A. Computer simulations showed that when AMP is bound, AcuB tends to adopt a compact conformation that is well suited to embracing and blocking AcuC. In contrast, when ATP or ADP bind, the protein shifts toward more extended or flexible shapes that weaken its grip on AcuC and allow the deacetylase to function.
A Built-In Safety Mechanism for Fuel Production
Bringing these findings together, the authors propose that AcuB is an energy sensor that reads the balance between AMP and ATP inside the cell. When energy is low, AMP levels rise. AcuB binds AMP, clamps onto AcuC, and prevents it from reactivating AcsA. As a result, AcsA remains acetylated and inactive, and the cell avoids committing precious energy to making more acetyl-CoA than it can afford. When energy is plentiful and ATP dominates, AcuB releases AcuC, which then removes acetyl tags from AcsA, allowing acetate to be converted into acetyl-CoA and fueling biosynthesis.
Why This Matters Beyond One Bacterium
This work identifies AcuB as the long-sought link that coordinates two opposing acetate pathways with the cell’s energy status. By coupling the chemical “energy charge” to enzyme activity through reversible protein–protein interactions and conformational changes, bacteria ensure efficient use of resources and avoid conflicting metabolic flows. The discovery also hints that similar energy-sensing switches may be widespread, and that stress signals like Ap4A can fine-tune this control, helping microbes cope with harsh conditions.
Citation: Janetzky, M., Geist, N., Schulze, S. et al. AcuB senses cellular energy charge to coordinate acetyl-CoA synthesis in bacteria. Nat Commun 17, 3815 (2026). https://doi.org/10.1038/s41467-026-71006-w
Keywords: bacterial metabolism, energy sensing, acetyl-CoA, protein acetylation, allosteric regulation