Clear Sky Science · en
Programmable nanomicelles rewire myeloid immunity for durable control of primary and metastatic breast cancer
A new way to help the immune system fight breast cancer
Most cancer treatments focus on killing tumor cells directly, but many tumors survive by quietly turning the body’s own immune cells into allies. This study describes a tiny, programmable medicine that aims to flip those cells back to being cancer fighters, offering long‑lasting control of aggressive breast cancers that have spread to other organs.
Why breast cancer can be so hard to cure
Breast cancer is the most commonly diagnosed cancer in women, and its deadliest feature is its tendency to spread, especially to the lungs. One subtype, triple‑negative breast cancer, is especially difficult to treat because it lacks common hormone and growth‑factor targets. Even modern immunotherapies that block the PD‑L1 “shield” on tumor cells help only a fraction of patients and often fail to extend survival. A major reason is the tumor’s microenvironment—the immediate neighborhood of cells and molecules around the cancer—which is packed with immune cells that have been reprogrammed to suppress, rather than support, anti‑tumor attacks.
The hidden influence of support cells around tumors
Among these surrounding cells, two types of myeloid cells play an outsized role: dendritic cells and macrophages. In healthy conditions, dendritic cells act as sentinels, picking up suspicious molecules and showing them to T cells to trigger a targeted immune attack. Macrophages can either support inflammation (M1‑like) and help clear threats, or calm things down and promote tissue repair (M2‑like). In breast tumors from patients and mouse models, the authors found fewer mature dendritic cells and a glut of M2‑like macrophages. These “peace‑keeper” macrophages were linked to poor T‑cell infiltration and worse outcomes, suggesting that if scientists could rewire these cells back into fighters, they might unlock more powerful and durable immune responses against cancer.
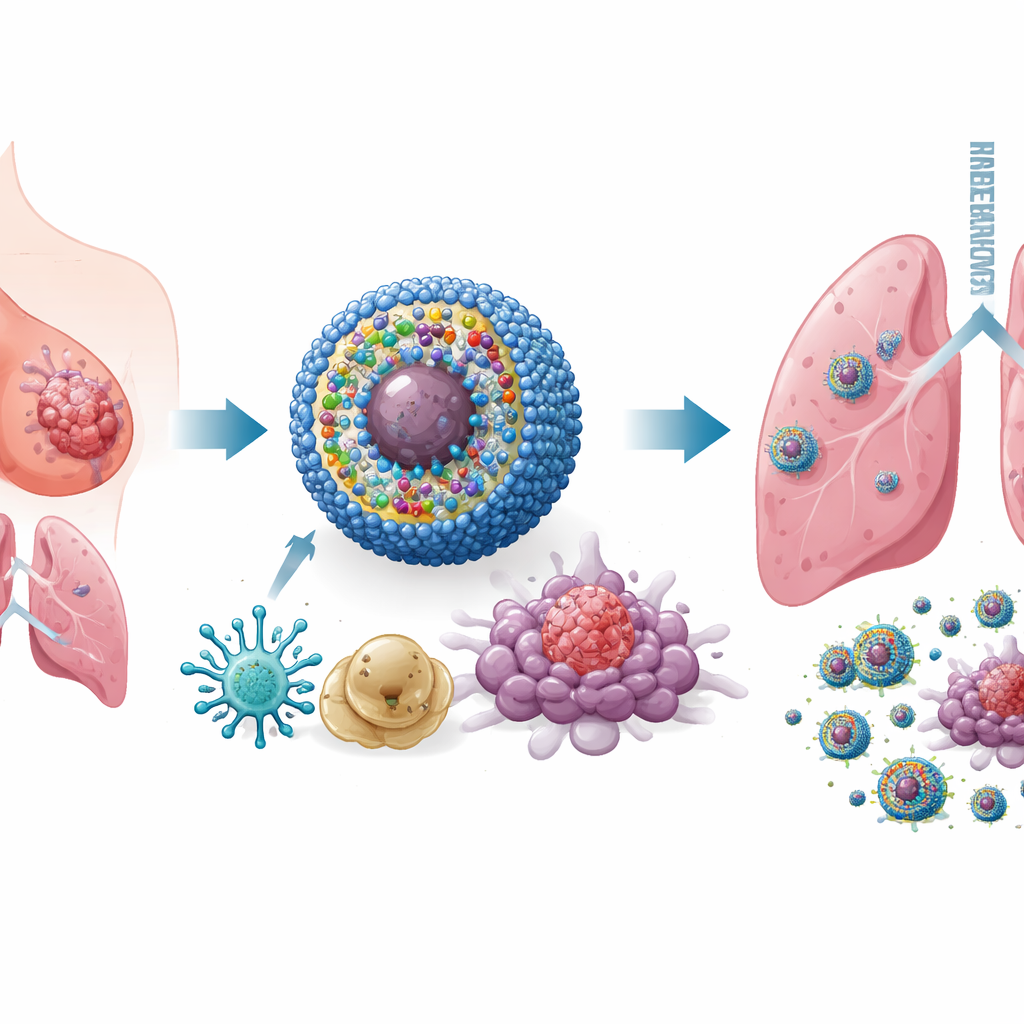
Designing a smart, multi‑tool nanomedicine
To tackle this problem, the researchers built a nanoscale delivery system they call IPANP—tiny spherical micelles that circulate in the bloodstream and accumulate in tumors. Each nanomicelle carries three components. First, a light‑activated dye (IR825) that, when illuminated with near‑infrared light from outside the body, generates reactive molecules that stress and kill tumor cells in a way that makes them highly visible to the immune system. Second, an antibody against PD‑L1, which helps remove a molecular brake that tumors use to turn off T cells. Third, a short piece of genetic material (antagomiR‑182) that blocks a microRNA, miR‑182, found at high levels in tumor‑promoting macrophages. The carrier is sensitive to the chemically reactive environment inside tumors, so it releases its cargo mainly where it is needed, limiting exposure to healthy tissues.
Turning suppressor cells into attackers
In cell cultures and multiple mouse models of breast cancer, including difficult triple‑negative and spontaneous tumor systems, IPANP combined with gentle near‑infrared light did more than just shrink tumors. The light‑activated dye caused cancer cells to die in an “immunogenic” fashion, spilling out danger signals that pushed dendritic cells to mature and present tumor fragments more efficiently to T cells. At the same time, blocking miR‑182 in macrophages lifted a brake on a key sensing pathway (TLR4/MYD88/NF‑κB), nudging them from the M2‑like, tumor‑supporting state toward an M1‑like, inflammatory state. As these two myeloid cell types shifted roles, more killer T cells flooded into tumors, became more active, and were less likely to shut down from exhaustion. The result was strong control of primary tumors, sharp reductions in lung metastases, and prolonged survival in treated animals.
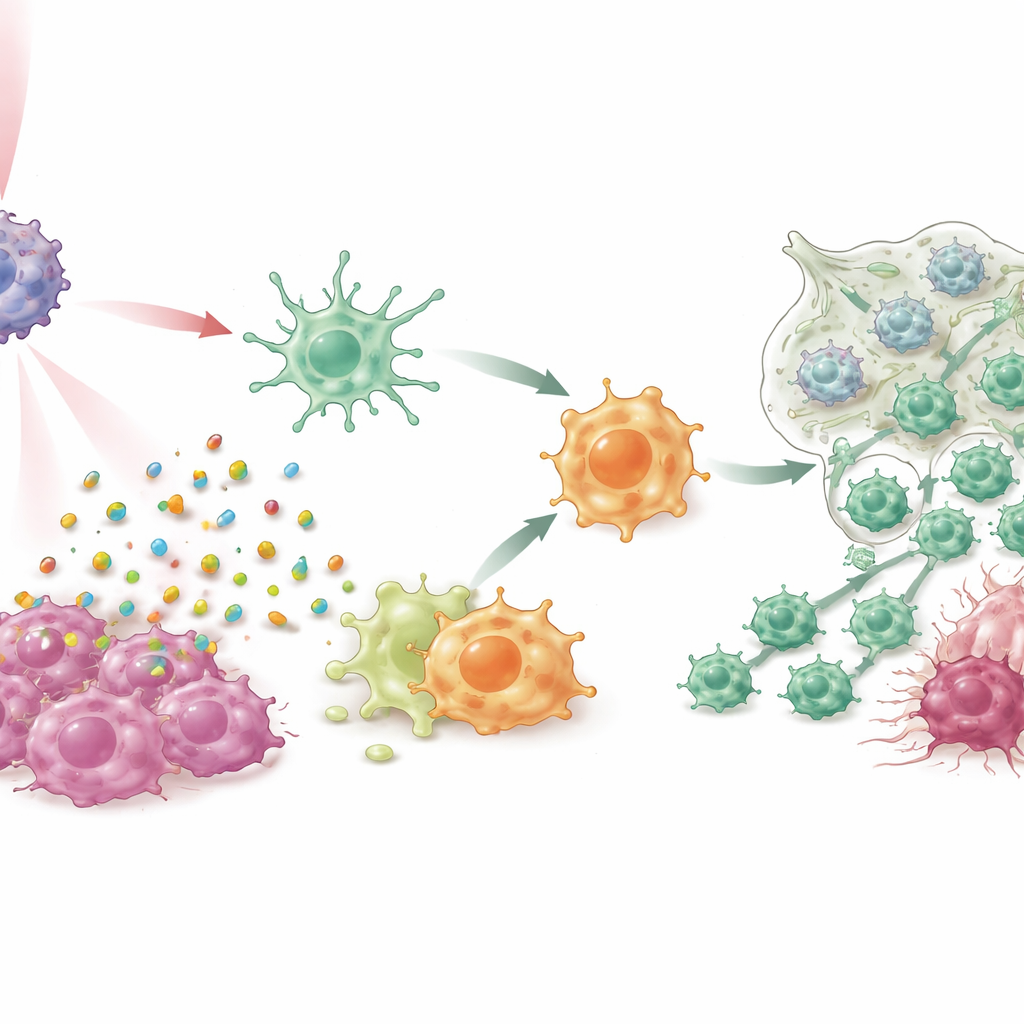
Lasting protection and signs of real‑world promise
Strikingly, mice whose tumors were cleared by IPANP and light were protected when researchers tried to re‑implant new tumors months later, indicating the formation of immune memory similar to that created by a vaccine. The team also tested the approach on freshly removed breast cancer samples from patients, kept alive as small tissue fragments, and in patient‑derived tumor grafts in humanized mice. In these settings, the treatment again pushed dendritic cells and macrophages into more active, anti‑tumor states, reduced growth‑promoting signals, boosted beneficial inflammatory molecules, increased killer T‑cell presence, and caused tumor cell death—all while showing good safety in long‑term animal studies.
What this could mean for future cancer treatment
This work shows that it may be possible to treat aggressive breast cancers not just by attacking tumor cells directly, but by re‑educating the immune cells that surround them. By packaging light‑activated therapy, checkpoint blocking, and genetic reprogramming into one smart nanomicelle, the researchers created a coordinated push that turned a suppressive tumor neighborhood into one that supports lasting immune defense. While much more testing is needed before this strategy could reach patients, it offers a blueprint for future treatments that remodel the immune environment to keep both primary and metastatic tumors under long‑term control.
Citation: Yang, J., Chang, D., Li, Y. et al. Programmable nanomicelles rewire myeloid immunity for durable control of primary and metastatic breast cancer. Nat Commun 17, 4082 (2026). https://doi.org/10.1038/s41467-026-70859-5
Keywords: breast cancer immunotherapy, nanomedicine, tumor microenvironment, macrophage reprogramming, dendritic cell activation