Clear Sky Science · en
Engineering programmable CAR and antigen pairing via drug-gated light activation
Teaching immune cells to hunt smarter
Many people have heard of CAR T cells, a type of personalized cancer treatment that reprograms a patient’s own immune cells to attack tumors. These therapies work well for some blood cancers but have struggled against solid tumors like those in the breast, prostate, or lung. This study describes a new way to both sharpen and restrain CAR T cells so they can better find diverse tumor cells while sparing healthy tissues.
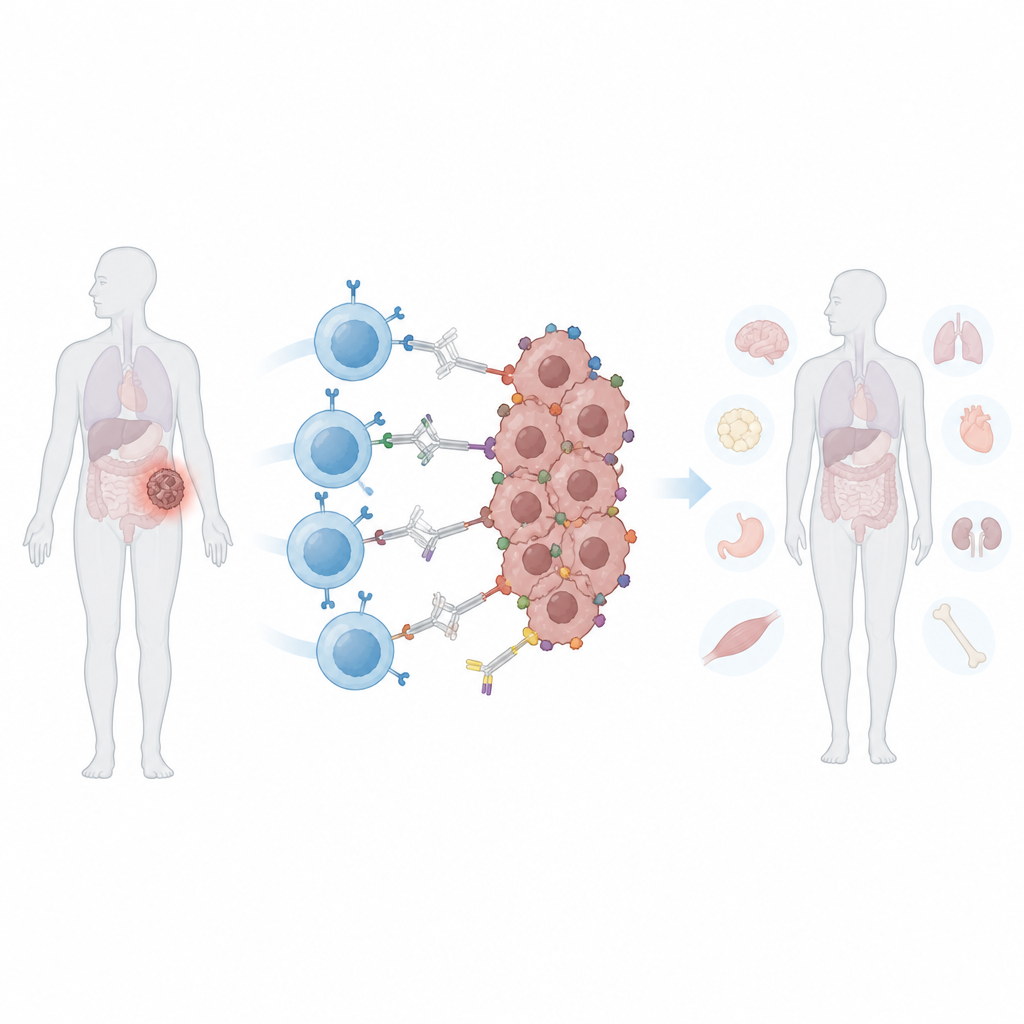
The challenge of shape-shifting tumors
Standard CAR T cells are built to recognize one specific molecular “flag” on cancer cells. Solid tumors often dodge this attack in two ways. First, they can lose or lower that flag, a process called antigen escape, allowing some cells to survive and regrow the tumor. Second, different cells within the same tumor may display different flags, a problem known as heterogeneity. Expanding CAR T cells to recognize more flags can help, but it also raises the risk that they will attack normal tissues that share those markers. Once conventional CAR T cells are infused, they are always on, making it hard for doctors to turn their activity up or down if side effects appear.
A universal plug for targeting many tumor types
The researchers created a “programmable” CAR that does not bind tumor cells directly. Instead, it recognizes a safe, brightly colored protein called phycoerythrin, commonly used in foods and lab tests. They engineered a small human-derived binding domain, called a PEbody, onto the CAR so that it latches onto any antibody carrying this protein tag. By choosing which PE-tagged antibody to infuse, doctors could in principle redirect the same batch of CAR T cells to different cancer markers, one after another or several at once. In lab tests, these PEbody CAR T cells efficiently killed leukemia and multiple solid tumor cell lines when provided with matching tagged antibodies, and they could be retargeted to new markers when the first one was lost.
A light and drug safety switch
To improve safety, the team added a layered control system that lets them switch CAR expression on only where and when needed. They used a two-part genetic switch that responds first to a common drug, tamoxifen, and then to blue light. Only T cells that see both cues activate a recombinase enzyme that permanently flips on the CAR gene. This “drug-gated light activation” confines powerful CAR activity to illuminated tumor regions. In cell culture, CAR levels and tumor killing rose sharply in T cells exposed to both tamoxifen and light, while cells in the dark or without drug remained quiet. In mice, the same system drove strong gene activation only in tumors on the side of the body that received light, with negligible activity on the shaded side.
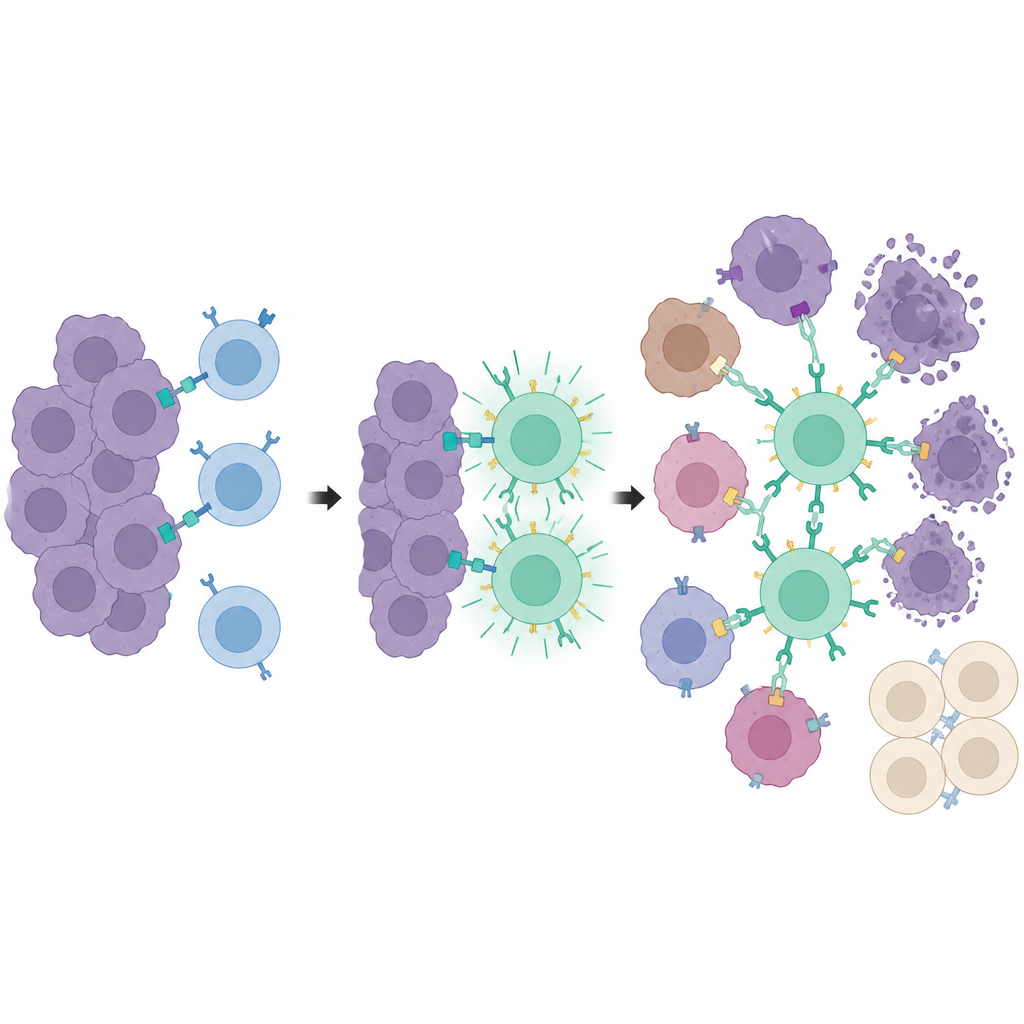
Training centers inside the tumor
Going a step further, the researchers separated tumor sensing from tumor killing. They used a second engineered receptor, called synNotch, that turns on the PEbody CAR only after a T cell encounters a special marker, a truncated version of CD19, placed on tumor cells. That marker itself is not naturally present but is induced locally by the same drug-and-light system. Only tumor cells that receive the light signal start displaying this training marker. When synNotch T cells arrive, those marked cells act as “training centers,” activating many T cells and equipping them with PEbody CARs. Once trained, these T cells can then attack the broader tumor mass using PE-tagged antibodies against common tumor markers such as PD-L1 or PSMA, even on cells that never expressed the training marker.
Local attack with distant tissues spared
In several mouse models of breast and prostate cancer, the combined drug, light, and synNotch system led to strong tumor control at illuminated sites. Tumors that received tamoxifen and light, followed by programmable T cells and PE-tagged antibodies, shrank or grew much more slowly than unlit tumors in the same animal. Importantly, the T cells circulated throughout the body but were only fully activated within the light-primed tumors, limiting damage to other organs that carried the same surface markers. Compared with a conventional CAR T treatment directed at the PSMA marker, the new strategy achieved similar tumor control with less weight loss, lower inflammatory signals, and reduced signs of liver damage.
What this could mean for future cancer care
For a lay reader, the key idea is that the researchers have built a kind of universal, rewritable CAR T system with a built-in local safety lock. Choice of tagged antibodies decides which tumor features to target, so doctors might adapt treatment as a cancer evolves without rebuilding the T cells from scratch. At the same time, the drug-and-light and training-center design helps ensure that full T cell attack power is unleashed only inside the tumor, not in healthy tissues that share the same markers. While much work remains before this approach reaches patients, the study outlines a flexible blueprint for safer, more adaptable cell therapies against complex solid tumors.
Citation: Huang, Z., Limsakul, P., Wu, Y. et al. Engineering programmable CAR and antigen pairing via drug-gated light activation. Nat Commun 17, 4252 (2026). https://doi.org/10.1038/s41467-026-70855-9
Keywords: CAR T cells, solid tumors, optogenetics, antigen escape, synthetic immunotherapy