Clear Sky Science · en
Membrane interfacial potential governs surface condensation and fibrillation of α-Synuclein in neurons
Why this matters for brain health
Parkinson’s disease and related disorders are marked by clumps of a protein called alpha synuclein inside brain cells, but how these clumps first appear and spread from cell to cell has remained a puzzle. This study shows that the outer skin of nerve cells, their membranes, can quietly gather alpha synuclein into tiny droplets at very low levels of the protein, and that these droplets can harden into damaging fibers that distort and even rupture the membrane. Understanding this hidden staging ground on the cell surface offers a fresh way to think about how harmful protein clumps start, grow, and move through the brain.
How a nerve cell’s skin can gather loose proteins
The authors focused on the thin fatty layer that surrounds cells, made of a mix of different lipids. They found that alpha synuclein, which usually floats freely inside neurons, can be drawn to this membrane and form dense droplets there, even when its overall concentration is extremely low. Strikingly, this only happened when the membrane contained a particular blend of two common lipids, one neutral and one negatively charged, in a 6:4 ratio. That specific mix creates just the right electrical conditions at the membrane surface to attract and hold alpha synuclein. When these conditions were not met, or when the same lipids were present as free particles rather than as a continuous surface, the protein did not condense into droplets.
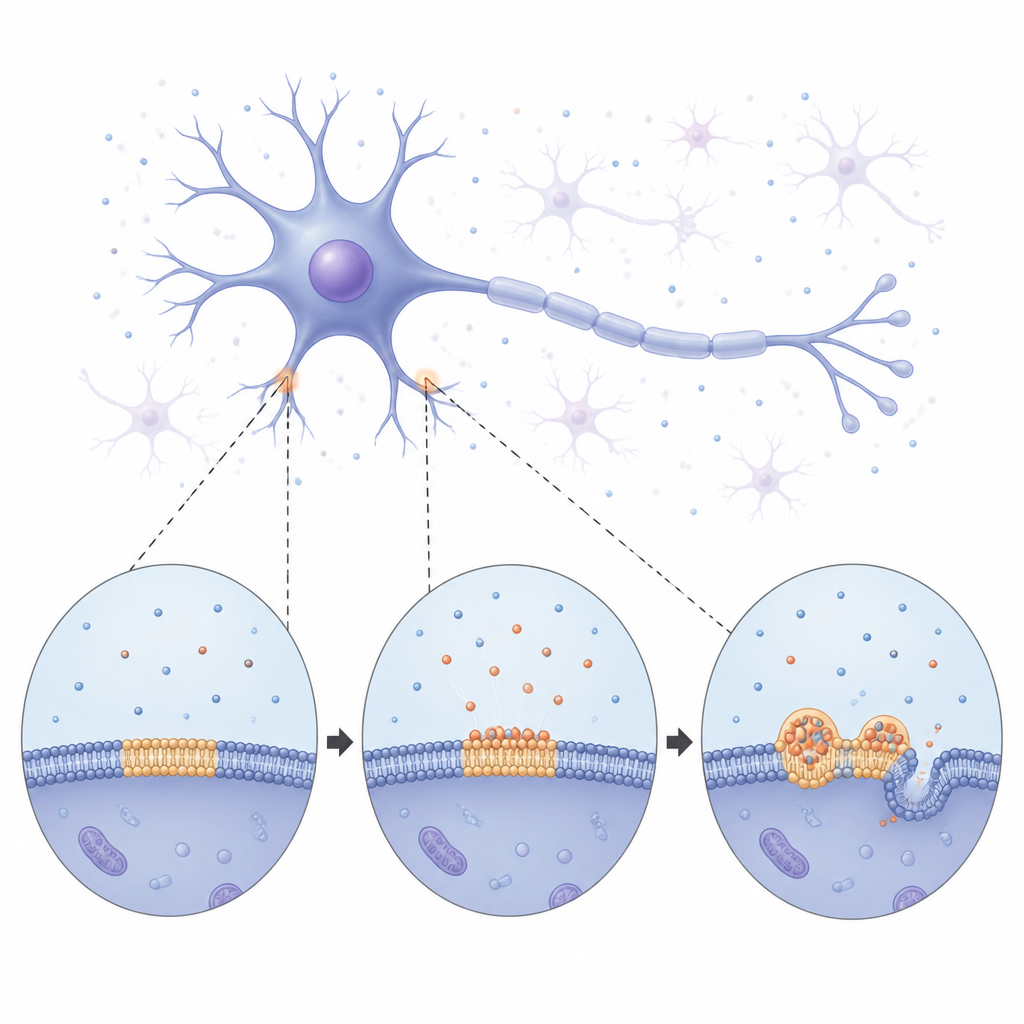
From soft droplets to rigid fibers
Using long term imaging, the team watched alpha synuclein slowly coat the membrane and then break up into bright, round condensates spread across the surface. At first these droplets behaved like tiny liquid blobs: they merged with one another and grew by slowly draining material from smaller neighbors. Over time, however, the droplets became more rigid. Measurements of how easily fluorescent signals recovered inside them showed that both the protein and the surrounding lipids moved less and less, signaling a shift from a fluid to a more solid state. Eventually, thin fiber like structures began to sprout from the droplets, bending and tubulating the underlying membrane. These observations suggest that the membrane bound droplets act as temporary, liquid staging areas where alpha synuclein can reorganize before locking into the solid fibers associated with disease.
How the membrane itself is changed
The study also asked what alpha synuclein does to the membrane while all this is happening. Experiments with model membranes showed that when the protein inserts into the surface, it makes the lipid layer more tightly packed and less compressible, altering its tension and curvature. Giant synthetic vesicles aspirated into micropipettes relaxed and slipped out once alpha synuclein was added, revealing a rapid drop in membrane tension and shape changes. Over hours, the protein’s grip on the membrane strengthened. These mechanical shifts create a stiffer, more ordered environment at the interface, which in turn favors more protein crowding and stable contacts, feeding back into further droplet growth and eventual fibril formation.
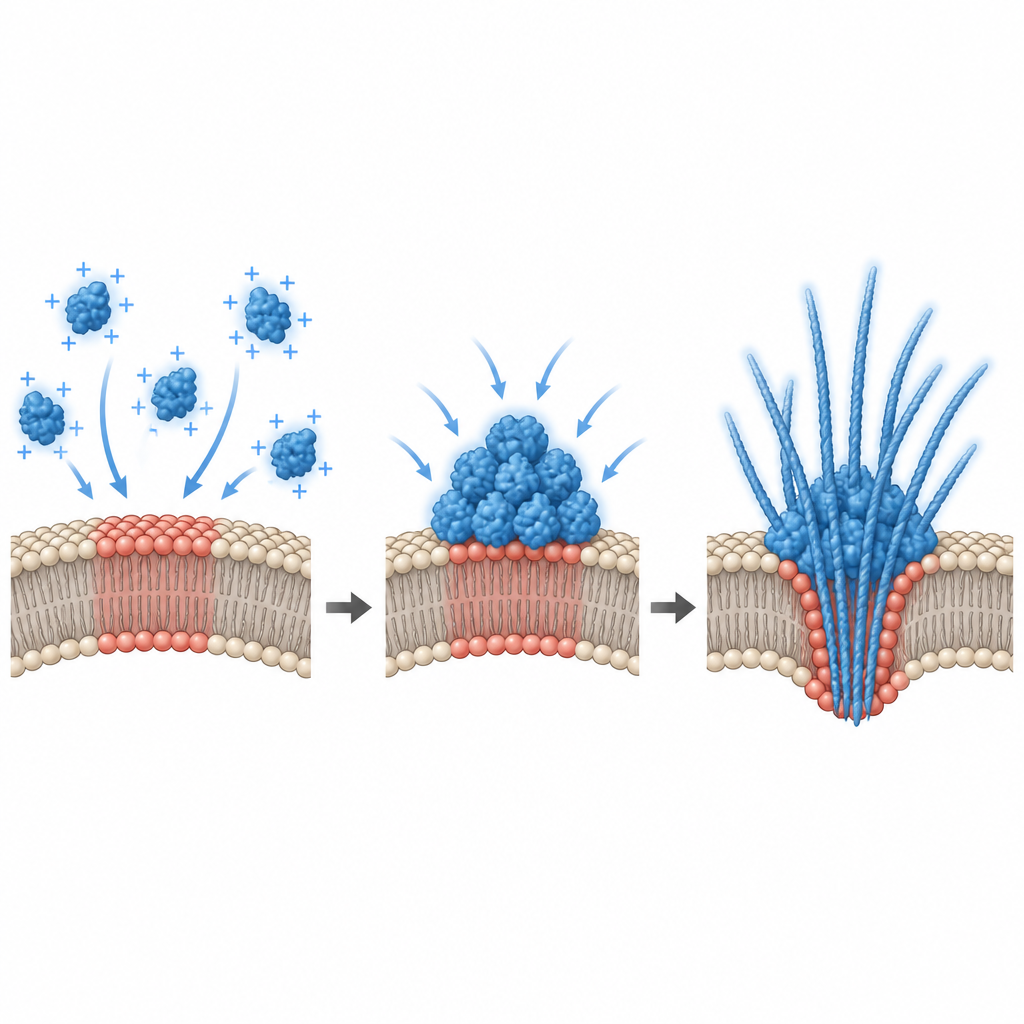
The hidden role of electrical surface charge
To explain why only certain lipid mixtures triggered condensation, the researchers measured the electrical potential at the membrane surface. They found that alpha synuclein binding occurred only within a narrow window of negative surface charge, and that robust condensation appeared near a threshold value in this range. Computer simulations backed this up, showing that an attractive membrane can cause a “prewetting like” transition: a thick protein rich layer forms on the surface even when the bulk solution is too dilute to phase separate on its own. In living cells, the team altered this interfacial potential in two ways. Adding extra negatively charged lipids to cultured hippocampal cells changed the potential and shrank pre existing alpha synuclein puncta, while depolarizing primary neurons with high potassium or glutamate increased the number and release of alpha synuclein droplets. This links everyday electrical activity of neurons directly to how and where the protein condenses.
What this means for neurodegenerative disease
Together, these findings paint a picture in which specific membrane patches on neurons act as silent assembly lines for alpha synuclein droplets at protein levels close to those found in the healthy brain. Slight changes in lipid makeup or electrical state can tip these surfaces into a regime where condensation occurs, droplets stiffen, and fibrils emerge, deforming and sometimes rupturing the membrane while releasing protein aggregates outside the cell. This membrane driven pathway offers a plausible route for alpha synuclein to propagate from neuron to neuron during neurodegeneration, and it suggests that tuning membrane composition or surface potential could one day help control harmful protein condensation at its earliest stages.
Citation: Shaikh, J., Nagarajan, A., Mitra, T. et al. Membrane interfacial potential governs surface condensation and fibrillation of α-Synuclein in neurons. Nat Commun 17, 4247 (2026). https://doi.org/10.1038/s41467-026-70840-2
Keywords: alpha synuclein, Parkinsons disease, neuronal membranes, protein condensation, lipid composition