Clear Sky Science · en
Complete inhibition of β-tryptase by tetramer dissociation and active site allostery due to a single antibody residue
Why this matters for allergies and asthma
People with asthma, severe allergies, or anaphylaxis often have trouble because certain immune cells in their tissues release powerful chemicals that tighten airways and damage tissue. One of the key culprits is an enzyme called beta‑tryptase, stored in mast cells and unleashed during allergic reactions. This study describes a specially engineered antibody that can switch beta‑tryptase fully off in a new way, potentially opening the door to better treatments for mast‑cell–driven diseases.
The troublemaker inside mast cells
Mast cells sit as sentinels in tissues such as the lungs, gut, and skin. When they sense allergens, venoms, or microbes, they quickly dump packets filled with histamine, signaling proteins, and enzymes. Beta‑tryptase is the most abundant of these enzymes and helps drive airway narrowing, extra collagen production, and muscle contraction in the lungs, all of which worsen asthma and other inflammatory diseases. Beta‑tryptase normally assembles into a four‑part ring, or tetramer, whose active sites point into a central pore. This architecture hides the active centers from most natural inhibitors in the body, making beta‑tryptase especially hard to block with conventional drugs.
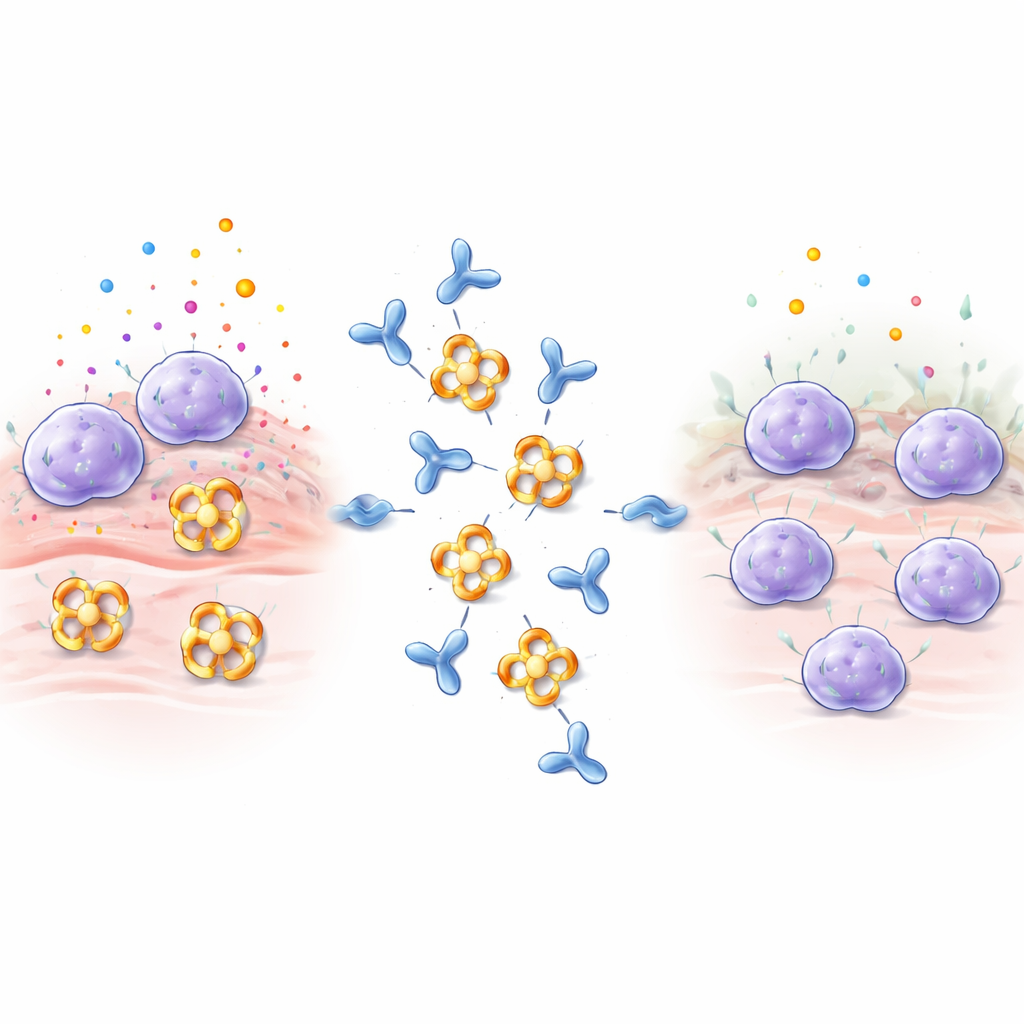
Designing an antibody that shuts the enzyme down
The researchers immunized rabbits with human beta‑tryptase and screened the resulting antibodies, focusing on one called E82.AS. Unlike earlier antibodies that only partly disabled the enzyme, E82.AS could suppress beta‑tryptase activity even under conditions that usually keep it strongly active. Detailed binding tests showed that both the full‑length antibody and its smaller fragment (Fab) latch onto beta‑tryptase very tightly and inhibit it in a dose‑dependent manner. When the team mixed E82.AS with the four‑part enzyme and ran it through separation columns, they saw that the tetramer broke apart into single units that traveled together with the antibody, indicating that E82.AS forces the ring to fall apart into monomers.
A distant grip that flips the off switch
To understand how E82.AS works so effectively, the scientists used hydrogen–deuterium exchange measurements and high‑resolution cryo‑electron microscopy. These approaches revealed that the antibody does not bind in the enzyme’s central pore or catalytic groove. Instead, it grabs a patch on the surface—an “exosite”—quite far from the chemical cutting center. Yet this distant grip triggers a cascade of shape changes that travel through flexible loops of the enzyme into the active site itself. In the antibody‑bound form, critical structural features that normally stabilize the cutting reaction, such as a small pocket that holds the reacting bond and a key negative charge that attracts basic amino acids, are distorted or moved. As a result, the active site can no longer accommodate either natural protein substrates or bulky protein‑based inhibitors.
The power of a single atom‑level tweak
One of the most striking findings is that a single amino‑acid change in the antibody can uncouple binding from function. The team altered one residue in the antibody’s light chain—changing an aspartate to glycine at position 93. This tiny modification barely weakened how well the antibody bound beta‑tryptase, but it completely abolished its ability to break apart the tetramer or block enzymatic activity. Structural analysis shows that the original aspartate side chain pushes against a loop in beta‑tryptase, nudging a nearby residue and, through a disulfide bond, transmitting the distortion into the heart of the active site. Removing that side chain eliminates the mechanical push, so the enzyme stays assembled and active despite the antibody’s presence. Conversely, by adding positive charges at two positions in the heavy chain, the researchers strengthened electrostatic attraction to a negatively charged patch on the enzyme and improved inhibitory potency up to nearly fivefold.
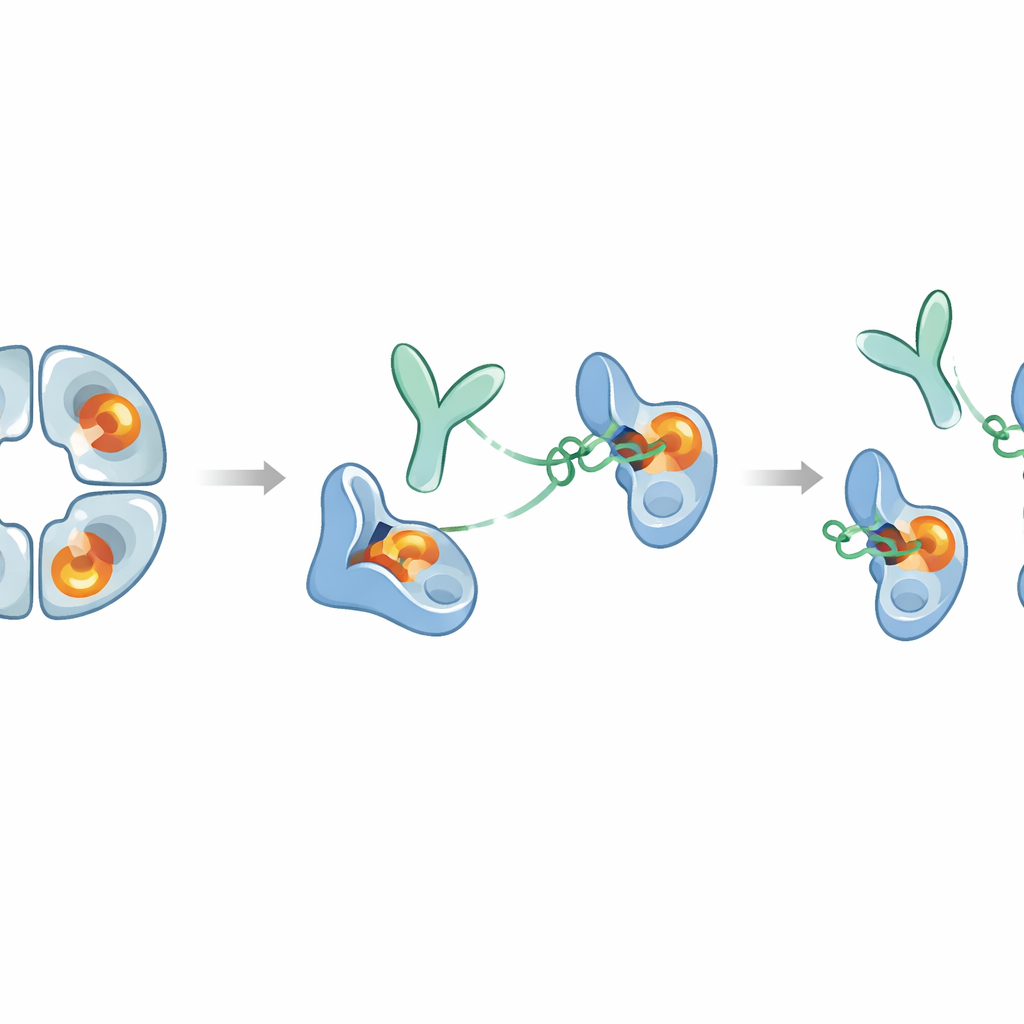
What this could mean for future treatments
Taken together, the work reveals a rare example of an antibody that completely silences a difficult‑to‑drug enzyme by pulling on a remote site rather than plugging the active center. E82.AS both pries apart the beta‑tryptase tetramer and locks each monomer into a “zymogen‑like” inactive shape, even when a helper molecule called heparin is present to boost activity. Because the antibody fragment alone can do this, it may be possible to design smaller versions better suited to reaching inflamed airways. At the same time, beta‑tryptase has beneficial roles in regulating immune signals, so any therapy will need to balance blocking harmful overactivity without suppressing useful functions for too long. This study offers a detailed molecular blueprint for that balance and suggests that similar allosteric strategies could be used to design highly selective inhibitors for other enzymes involved in human disease.
Citation: Maun, H.R., Azumaya, C.M., Walters, B.T. et al. Complete inhibition of β-tryptase by tetramer dissociation and active site allostery due to a single antibody residue. Nat Commun 17, 3393 (2026). https://doi.org/10.1038/s41467-026-70491-3
Keywords: mast cells, beta-tryptase, allosteric antibody, asthma, enzyme inhibition