Clear Sky Science · en
The oncogenic CCDC6-RET fusion protein is a dual ATP- and ADP-dependent kinase
Why this cancer study matters
Cancers of the lung and thyroid are often driven by faulty versions of cell communication proteins called kinases. One such faulty version, the CCDC6-RET fusion protein, crops up frequently in papillary thyroid cancer and some lung cancers, especially after radiation exposure. This study uncovers how that fusion protein switches itself on and why it may be unusually well adapted to the stressed, energy-poor environment inside tumors, opening new angles for targeted treatment.
A genetic mash-up that fuels tumors
In healthy cells, the RET protein sits in the cell membrane and helps relay growth signals. In many thyroid cancers, and in a subset of lung cancers, DNA breaks and rejoins in the wrong places, creating a hybrid gene that fuses part of the CCDC6 protein to the business end of RET. The result, called CCDC6-RET, is a stand-alone signaling engine that no longer needs normal external cues. The authors produced this fusion protein in large quantities using insect cells and showed that it forms very stable pairs, or dimers, in solution. These dimers were highly active, adding phosphate groups to themselves at many sites and responding strongly to known RET-blocking drugs, confirming that the laboratory-made protein behaves like its cancer-driving counterpart.
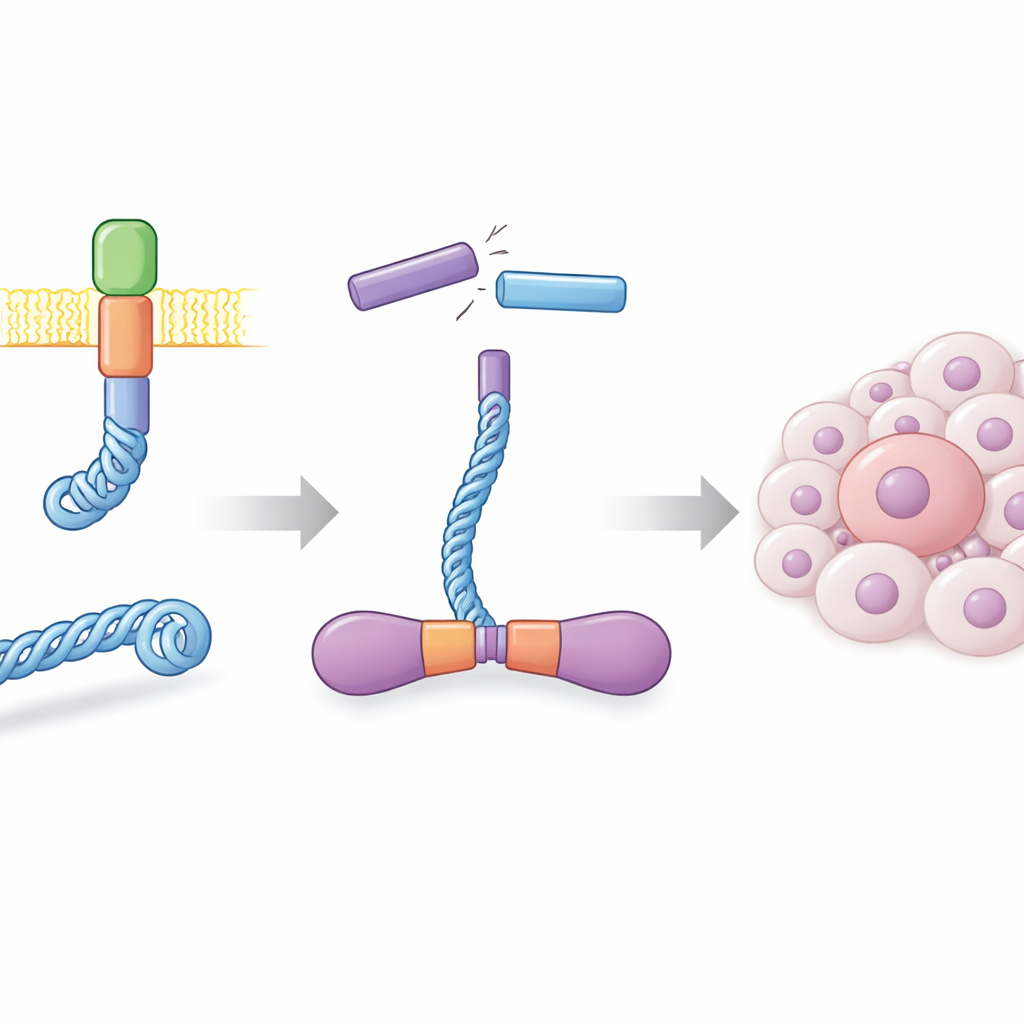
A kinase that runs on two kinds of fuel
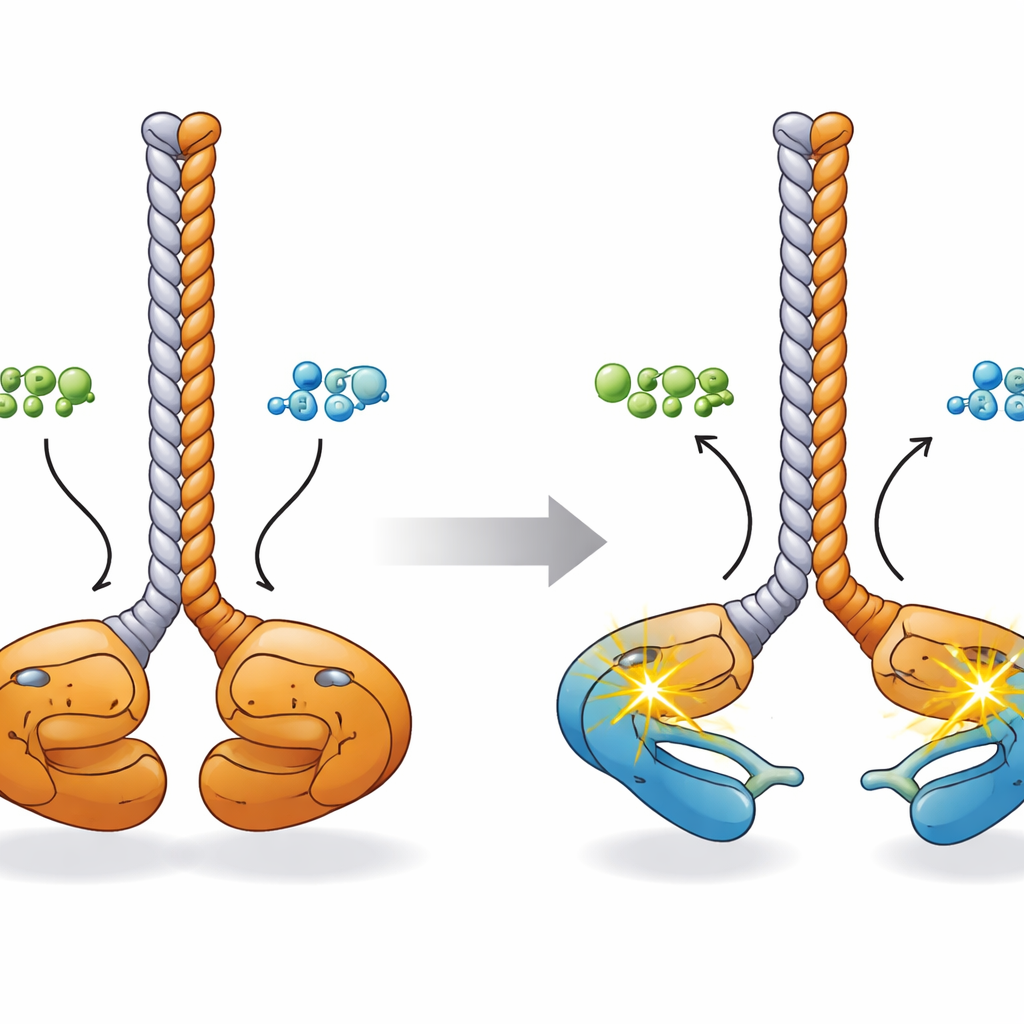
Most kinases use ATP, the cell’s primary energy currency, as the source of phosphate for their reactions. When the authors analyzed CCDC6-RET’s activity using biochemical assays and time-resolved mass spectrometry, they noticed something surprising: the protein’s phosphorylation increased not only with ATP but also with ADP, the lower-energy product left after ATP is spent. Follow-up experiments showed that CCDC6-RET binds both nucleotides and can use either as a phosphate donor, although it works faster with ATP. At moderate ADP levels the fusion remains active, but very high ADP concentrations start to interfere with ATP-driven activity, suggesting that ADP can both fuel and regulate this cancer enzyme depending on its abundance.
Fine-tuned control switches on the protein surface
The team went on to map exactly where CCDC6-RET phosphorylates itself. They found multiple sites not only in the catalytic core of RET but also in the CCDC6 portion, including previously unknown ones. Two neighboring tyrosines in a flexible segment called the activation loop played distinct roles. One site (Tyr 900) appears crucial for proper folding and stability of the fusion protein, while the other (Tyr 905) is essential for efficient phosphorylation of other proteins. Mutating Tyr 905 largely spared the protein’s ability to modify itself but crippled its ability to act on an added peptide substrate, revealing that “self-activation” and “signaling outward” are controlled somewhat separately. This nuanced control may help the fusion protein stay on without losing structural integrity.
Revealing the protein’s moving parts in 3D
To understand how these chemical changes relate to shape, the researchers pieced together a three-dimensional picture using predictive artificial-intelligence models, electron microscopy, small-angle X-ray scattering, and cross-linking mass spectrometry. In its resting state, the CCDC6-RET dimer adopts a “face-to-face” configuration: the two kinase domains sit close together at the base of a shared coiled-coil stalk formed by the CCDC6 segments, with their activation loops touching. Upon binding ATP or ADP, those kinase lobes swing apart like the tips of a pair of chopsticks. Computer simulations suggest that in this more open state each kinase can reach in and phosphorylate its own activation loop (“in cis”), priming the dimer for rapid signaling to other cellular targets.
What this means for cancer and future therapies
Tumor cells often live on the edge of an energy crisis, with lower ATP and higher ADP levels than healthy cells. A fusion kinase that can function with either ATP or ADP—and even shift its target preference toward different amino acids under ADP-rich conditions—may therefore have a survival advantage in the harsh microenvironments of solid tumors. By clarifying how CCDC6-RET is built, how it dimerizes, how it uses both forms of cellular fuel, and which phosphorylation sites matter most, this work offers a detailed blueprint for designing next-generation drugs. Such drugs could aim not only to block the active site, but also to disrupt the dimer, interfere with activation-loop switching, or exploit the enzyme’s unusual dependence on the cell’s energy balance.
Citation: Martín-Hurtado, A., Contreras, J., Sánchez-Wandelmer, J. et al. The oncogenic CCDC6-RET fusion protein is a dual ATP- and ADP-dependent kinase. Nat Commun 17, 3595 (2026). https://doi.org/10.1038/s41467-026-69833-y
Keywords: RET fusion, papillary thyroid cancer, lung adenocarcinoma, protein kinase activation, ATP ADP metabolism