Clear Sky Science · en
Structural insights into the gating mechanism of the fission yeast phosphate exporter SpXpr1
Why tiny gateways for phosphate matter
Every cell must carefully balance phosphate, a small but indispensable ingredient in molecules such as DNA, membranes, and the cell’s energy currency. Too little phosphate stalls growth; too much can be toxic. This study looks at how fission yeast, a model fungus, pushes excess phosphate out of the cell through a molecular doorway called SpXpr1. By revealing this doorway’s three-dimensional structure and how it opens and closes, the work sheds light on how very different organisms—from fungi to humans—use related proteins to keep phosphate levels in check, with implications for brain disease, kidney health, agriculture, and antifungal strategies.
Keeping the balance inside a cell
Phosphate homeostasis is a universal problem: cells must import phosphate for metabolism and building blocks, store some of it safely, and export any surplus. In fission yeast, uptake systems switch on under scarcity and are shut down when phosphate is abundant, while a storage complex packs excess phosphate into long chains. Genetic studies had suggested that a protein called SpXpr1 acts as a dedicated exporter, helping prevent dangerous overload when storage pathways are compromised. SpXpr1 belongs to the XPR1 family of exporters found across eukaryotes, including plants and humans, where related proteins are linked to disorders such as brain calcification and kidney dysfunction. Despite this shared family background, it was unclear whether the yeast version worked in the same way as the human protein.
Seeing the protein and proving it is a channel
The researchers first tested whether SpXpr1 truly moves phosphate across membranes. They inserted the yeast protein into human cells that had their own XPR1 removed, then measured how much phosphate leaked into the surrounding solution. Cells with SpXpr1 exported phosphate more than control cells did, confirming its activity, although it was less efficient than the human exporter. Using electrical recordings, the team then showed that cells bearing SpXpr1 carried phosphate-dependent currents that increased when the small signaling molecule inositol hexakisphosphate (InsP6) was present. This established SpXpr1 as a bona fide channel that allows phosphate ions to pass and that can be tuned by inositol-containing messengers.
Two doors along a shared pathway
To understand how SpXpr1 works at the atomic level, the authors used cryo–electron microscopy to determine its structure in the absence and presence of InsP6. Each SpXpr1 unit forms part of a paired assembly in the membrane and contains an external transport core and an internal regulatory domain. Along the central pathway, they observed two distinct spots where phosphate can sit, coordinated by positively charged amino acids; these sites are conserved in human and plant counterparts, highlighting a shared blueprint for how phosphate itself is recognized. Yet, unlike its relatives, the yeast protein builds two physical gates into this pathway. On the inside, a flexible segment called the N-loop, supported by a nearby short helix and by an extended end of one membrane-spanning helix, folds into the pore entrance and blocks phosphate from getting in. On the outside, a small loop between two helices forms a plug that caps the exit. Mutating or trimming either of these gates caused the channel to leak more phosphate, confirming that they normally hold the protein in a self-inhibited, closed state.
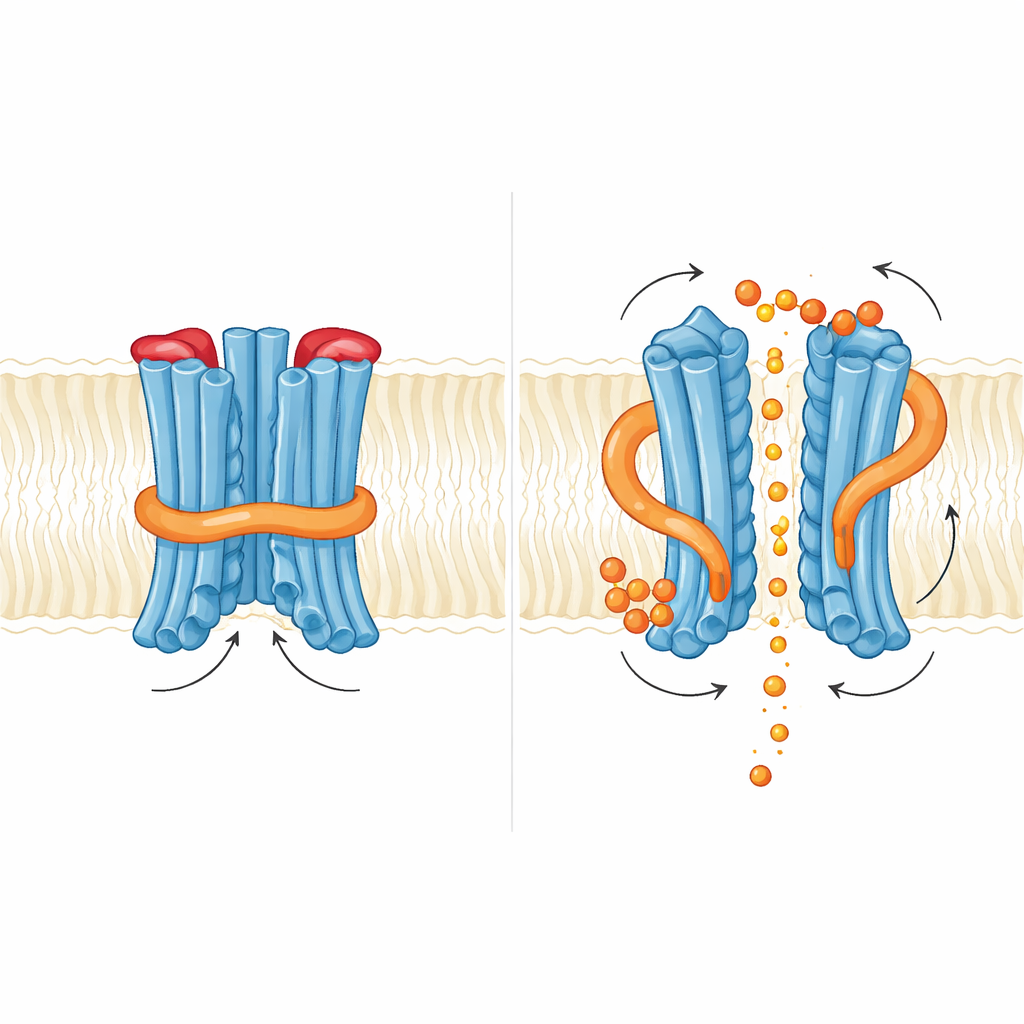
How a small messenger nudges the gates
The team then examined what happens when InsP6 binds. They found two binding pockets rich in basic residues: one where the regulatory domain meets the membrane core, and another at the interface between the two regulatory domains in the dimer. InsP6 binding caused a subtle rotation of the regulatory region and destabilized the short helix that braces the N-loop, but the loop itself still sat in the way of phosphate in the captured structures. Functionally, adding InsP6 increased phosphate currents, but mutations that weakened InsP6 binding did not abolish export and sometimes even enhanced it. This suggests that, in yeast, the inositol messenger acts more like a fine adjuster that loosens an internal lock than like an on–off switch. The authors propose that higher-charged inositol pyrophosphates, such as InsP7 or InsP8, may be the real physiological triggers capable of fully swinging both the inner N-loop gate and the outer plug out of the way.
What this means beyond yeast
Together, these structural and functional experiments support a working model in which SpXpr1 normally sits in a dual-locked state, with an inner and an outer gate guarding the phosphate pathway. Rising levels of inositol-based signals produced during phosphate excess gradually release these locks, allowing phosphate to flow out and protect the cell. Compared with the human exporter, which uses a different movable segment to plug and unplug its pore, the yeast solution shows how evolution can rewire the same basic scaffold to suit different lifestyles—strict conservation in fungi versus rapid export in mammals. Understanding these design principles could help researchers tune phosphate use in crops, design targeted antifungal drugs that disrupt fungal phosphate control, and better interpret disease-causing changes in the human XPR1 protein.
Citation: Yang, H., Wang, Y., Yue, C. et al. Structural insights into the gating mechanism of the fission yeast phosphate exporter SpXpr1. Cell Discov 12, 27 (2026). https://doi.org/10.1038/s41421-026-00883-8
Keywords: phosphate homeostasis, ion channels, cryo-EM structure, inositol polyphosphates, membrane transport