Clear Sky Science · en
Salinomycin as a death switch: how gastric cancer cells choose their demise
Why this research matters for patients
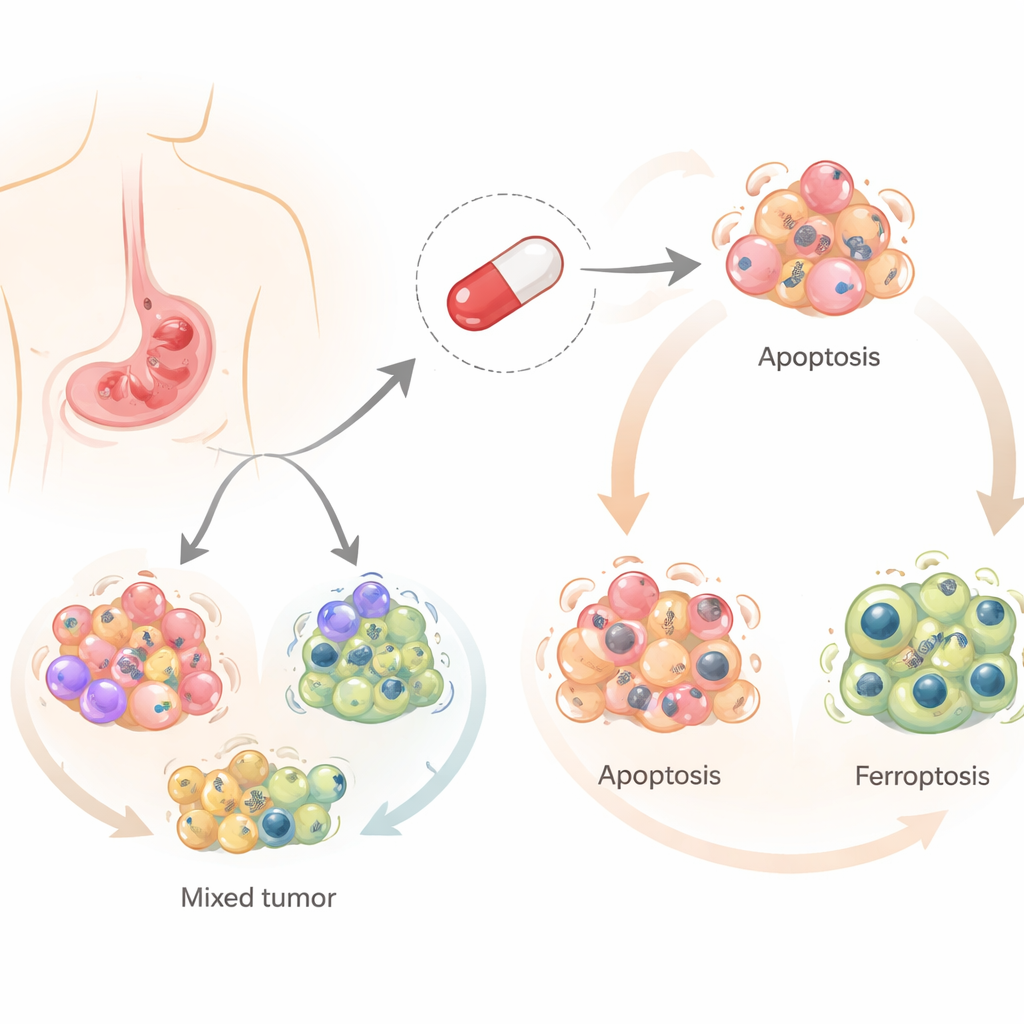
Stomach cancer is still one of the world’s deadliest cancers, largely because many tumors learn to ignore standard drugs. A big part of the problem is that tumors are made of many different kinds of cells, including a small but stubborn group called cancer stem cells that can restart the disease after treatment. This study explores whether an old veterinary drug, salinomycin, can be turned into a new weapon against stomach cancer by forcing these cells to die in more than one way.
An old drug with a new job
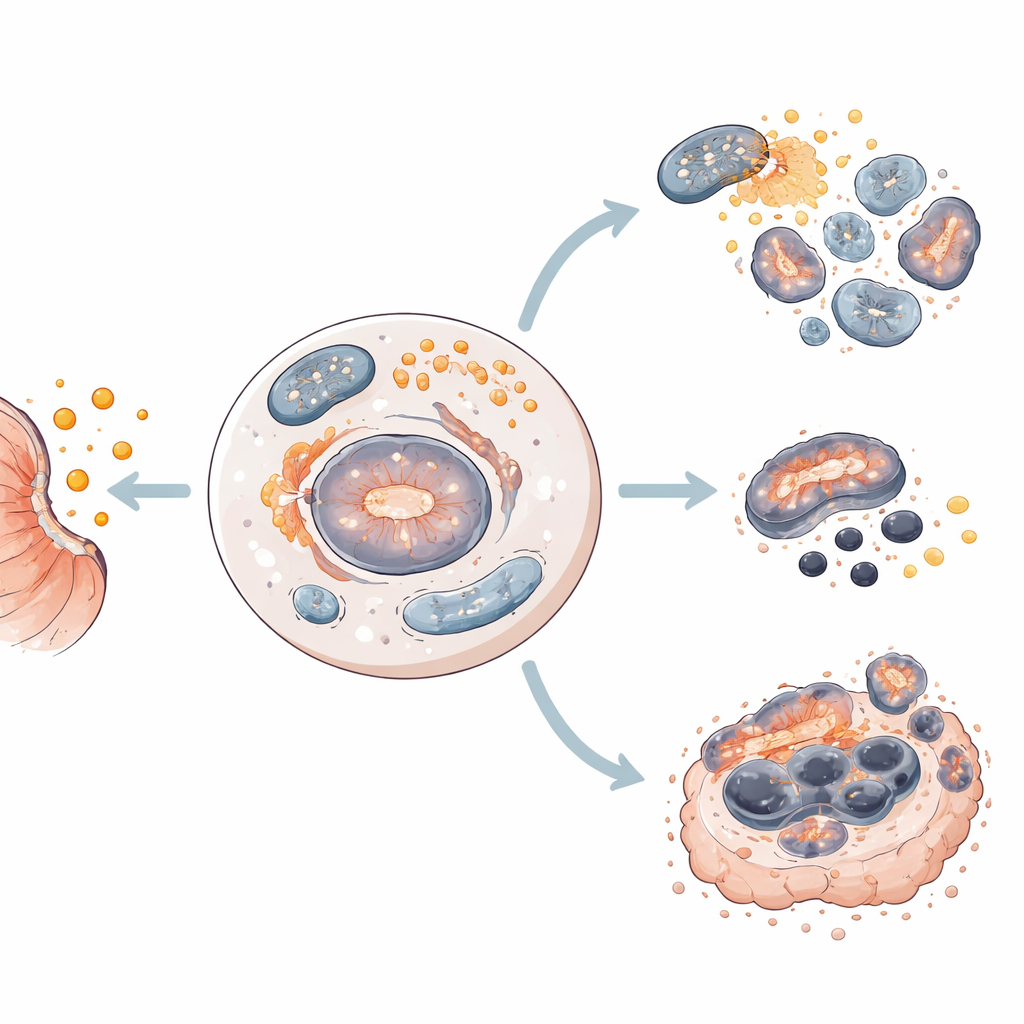
Salinomycin was originally developed to prevent infections in farm animals, but over the last decade it has drawn attention for its ability to kill cancer cells, especially cancer stem cells. Unlike most chemotherapy drugs, which usually rely on a single main route to kill cells, salinomycin can trigger several controlled forms of cell death. The researchers focused on three of these: classic programmed cell death (apoptosis), a recycling-related process (autophagy), and a newer, iron-dependent type of death driven by fat damage (ferroptosis). Understanding which of these paths are activated in stomach cancer is important, because tumors that evade one route might still be vulnerable to another.
How tumor cells choose their way to die
The team tested salinomycin on four different human stomach cancer cell lines that represent the diversity seen in patients. They found that the same drug pushed different cells toward different fates. In two cell lines, salinomycin mainly caused apoptosis, where cells quietly dismantle themselves. In the other two, it drove ferroptosis, a more explosive death linked to iron, reactive oxygen molecules, and damage to fatty components of the cell. In every case, autophagy switched on early, suggesting a shared starting point from which cells are steered toward one death route or the other.
Signals that guide the death decision
Digging deeper, the researchers measured key proteins that act like traffic lights inside the cell. In cells heading for apoptosis, salinomycin turned down growth and survival signals (such as mTOR and survivin) and activated proteins that cut up cell contents and damage mitochondria, the cell’s power plants. In cells undergoing ferroptosis, the drug again reduced mTOR and survivin, but also suppressed two important protectors against fat damage, SLC7A11 and GPX4. By analyzing gene activity data, they identified a 20-gene pattern that marks cells predisposed to ferroptosis rather than apoptosis, suggesting that each tumor’s molecular “wiring” helps determine how it will succumb to treatment.
Targeting the roots of relapse
The study also examined cancer stem cells, identified by surface markers CD44 and CD133, which are linked to drug resistance and ferroptosis control. In the most drug-resistant cell lines, these stem-like cells were common, formed robust 3D “spheroids,” and generated many colonies. Salinomycin sharply reduced the fraction of cells carrying these markers, shrank or eliminated spheroids, and blocked colony growth, indicating that it attacks the very cells most likely to cause relapse. When the team applied the ferroptosis-related gene pattern to large patient datasets, they could separate groups with different survival prospects, showing that this molecular fingerprint has real-world prognostic value.
Testing the approach in patient-like mini-tumors
To move closer to the clinic, the researchers tested salinomycin on patient-derived stomach cancer organoids—tiny 3D structures grown from human tumor samples that mimic many features of real cancers. Across all organoid models, salinomycin reduced viability and disrupted structure. In most cases it activated apoptotic enzymes; in one model that did not show apoptosis, it instead triggered hallmarks of ferroptosis, including changes in mitochondrial activity and loss of protective ferroptosis proteins. Autophagy increased in all models, reinforcing its role as a common early step in salinomycin-induced death.
What this could mean for future treatment
Overall, this work portrays salinomycin as a “death switch” that can flip stomach cancer cells into either apoptotic or ferroptotic self-destruction, with autophagy as a shared prelude. By simultaneously hitting the bulk tumor and its stem-like core, and by offering an alternative death route when the usual one is blocked, salinomycin may help overcome multidrug resistance. The ferroptosis-based gene signature developed here could help doctors identify which patients are more likely to benefit, opening the door to more personalized and durable treatment strategies for gastric cancer.
Citation: Laurenziello, P., Luongo, M., Lospinoso Severini, F. et al. Salinomycin as a death switch: how gastric cancer cells choose their demise. Cell Death Discov. 12, 171 (2026). https://doi.org/10.1038/s41420-026-03058-2
Keywords: gastric cancer, salinomycin, cancer stem cells, ferroptosis, drug resistance