Clear Sky Science · en
Lgmn targets two distinct GPCRs, PAR2 and µ-OR1, and induces cell death in acute lymphoblastic leukemia through an intracellular Ca²⁺ imbalance triggered by ER Ca²⁺ release
How a Parasite Enzyme May Help Fight Childhood Leukemia
Acute lymphoblastic leukemia (ALL) is the most common childhood blood cancer, and although current treatments can cure many children, they often come with harsh side effects and the risk of relapse. In an unexpected twist, researchers studying a protein used by common gut parasites to attack human cells have discovered that this same molecule can selectively push leukemia cells toward self-destruction. By tracing how this protein perturbs the balance of calcium inside cancer cells, the study points to fresh ways of killing leukemia cells while hinting at new drug targets.
A Hidden Weapon from Common Parasites
The work centers on legumain, an enzyme secreted by intestinal and urogenital protozoa such as Blastocystis and Trichomonas. These parasites use legumain as part of their arsenal to invade and damage host tissues. Earlier research showed that legumain can cut a receptor on human cells known as PAR2 and, in doing so, trigger a rise in calcium inside the cell. But even when PAR2 was removed or made resistant to cutting, legumain still managed to boost calcium levels, implying that another trigger was at work. Because a widely used leukemia drug, L-asparaginase, also acts on PAR2 and on a second receptor called μ-OR1, the authors asked whether legumain might likewise use more than one route to disturb the inner life of leukemia cells.
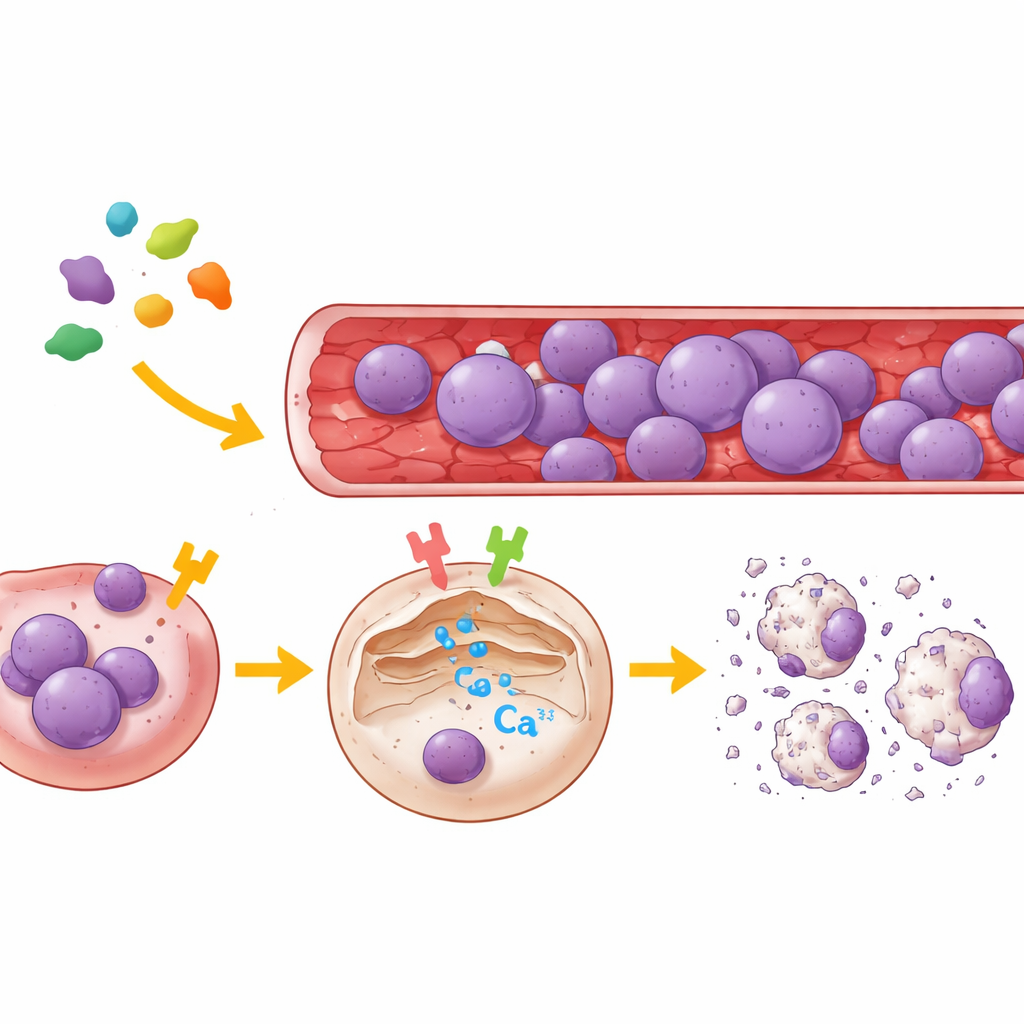
Two Cell Switches that Control Calcium
To map legumain’s targets, the team used both human kidney cells and several types of ALL cells from patients. They selectively reduced either PAR2 or μ-OR1 in these cells, or blocked them with highly specific small-molecule inhibitors, then watched calcium movements in single cells using a fluorescent probe that reports calcium release from the endoplasmic reticulum, the cell’s main internal calcium store. The results were clear: legumain drove a strong burst of calcium out of this store only when at least one of the two receptors was active. Inhibiting μ-OR1 sharply blunted the calcium surge, while blocking PAR2 produced a smaller but noticeable reduction. When both receptors were disabled, legumain could no longer provoke calcium release. Across several B‑cell and T‑cell leukemia lines, μ-OR1 emerged as the dominant switch, with PAR2 providing a supporting role.
Unbalanced Signals Tip Cells Toward Death
Having established that legumain hits both receptors, the researchers then followed the signaling chains that connect these surface switches to events deep inside the cell. PAR2 couples to a G-protein that directly activates an enzyme called PLC, which in turn unlocks calcium valves on the endoplasmic reticulum. μ-OR1, by contrast, primarily signals through a different G-protein that damps down production of the messenger molecule cAMP and thereby quiets an enzyme known as protein kinase A (PKA). Normally, PKA puts inhibitory “brakes” on PLC and maintains the activity of a survival protein called BAD in a non-lethal state. When legumain stimulates μ-OR1, cAMP levels fall, PKA activity drops, these brakes are released, and PLC is free to drive more calcium out of storage. At the same time, BAD shifts into a form that weakens the cell’s built-in defenses against death.
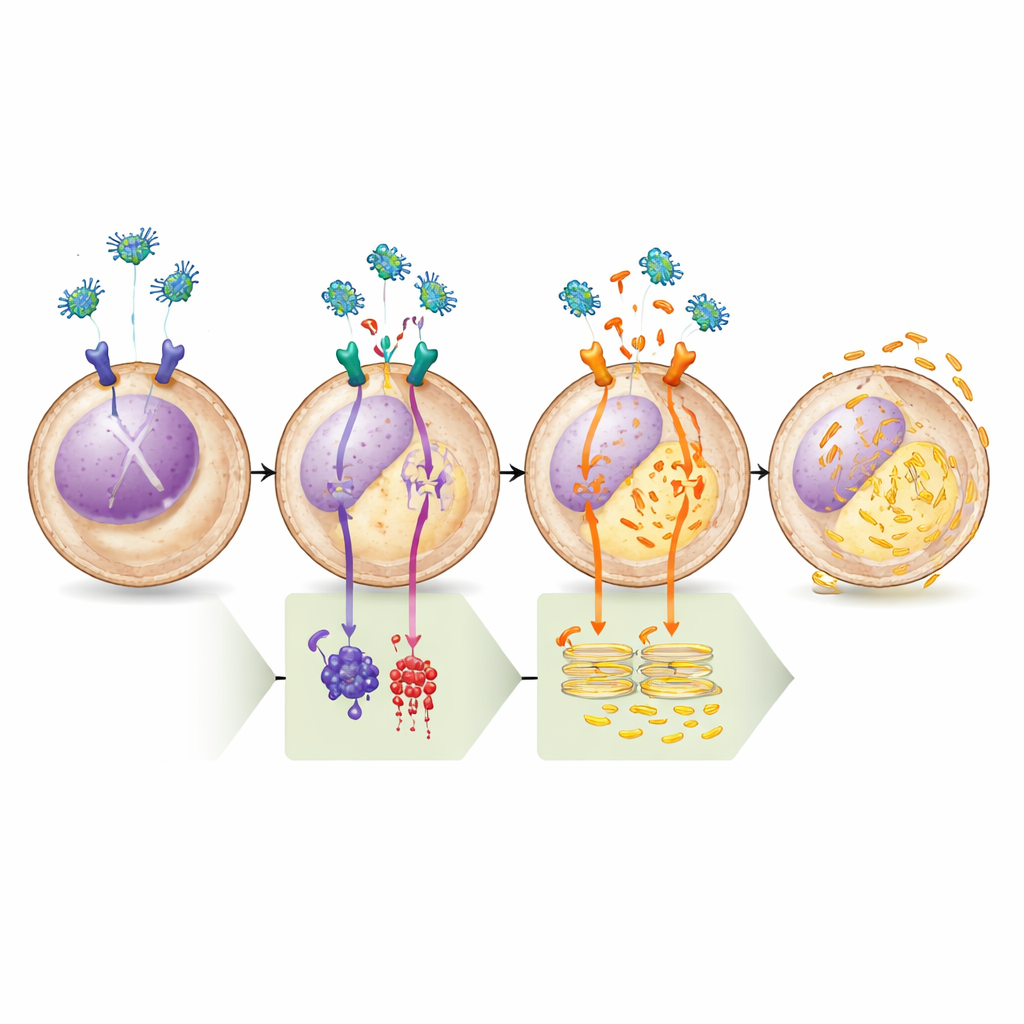
Proving the Pathway Step by Step
The team verified this model with a series of pharmacological tests. Drugs that boosted cAMP or directly activated the enzyme that makes it (adenylate cyclase) completely blocked legumain’s ability to empty calcium from the endoplasmic reticulum, underscoring the importance of the μ-OR1–cAMP–PKA branch. Conversely, a highly selective PKA inhibitor alone was enough to trigger calcium release, and adding legumain afterward produced no further effect, showing that once PKA is switched off, the pathway has already been fully engaged. Biochemical analyses confirmed that legumain treatment lowered the phosphate “tags” on both PLC and BAD at sites known to be controlled by PKA, matching the predicted change in their activity. Finally, when the researchers blocked the calcium channel on the endoplasmic reticulum or removed a key scaffolding protein required for this channel, legumain no longer induced cell death, firmly tying the lethal outcome to calcium imbalance.
What This Could Mean for Future Leukemia Treatments
By the end of this signaling cascade, leukemia cells experience a sustained flood of calcium from their internal stores, coupled with the shutdown of a critical survival circuit. Together, these changes push the cells into programmed death, or apoptosis. For a lay reader, the message is that legumain acts like a two-handed saboteur: it presses both of the cell’s “go” buttons for calcium release while at the same time cutting the wires to its safety systems. Although legumain itself comes from parasites and would require careful evaluation before use as a therapy, the study highlights μ-OR1, PAR2, and PKA as promising drug targets. Medicines that mimic legumain’s selective disruption of calcium balance in leukemia cells—or that strategically inhibit PKA—could one day help treat patients whose cancers resist current therapies.
Citation: Lee, J.K., Riabowol, K. & Lee, KY. Lgmn targets two distinct GPCRs, PAR2 and µ-OR1, and induces cell death in acute lymphoblastic leukemia through an intracellular Ca²⁺ imbalance triggered by ER Ca²⁺ release. Cell Death Discov. 12, 143 (2026). https://doi.org/10.1038/s41420-026-03003-3
Keywords: acute lymphoblastic leukemia, legumain, calcium signaling, G protein coupled receptors, apoptosis