Clear Sky Science · en
Histone lactylation-driven feedback loop modulates pyrimidine metabolism to promote oral carcinogenesis
Why this matters for mouth health
Oral cancer often starts as a small white patch in the mouth and can quietly progress into a life‑threatening disease. This study uncovers how changes in the way precancerous mouth cells burn sugar create a self‑reinforcing loop that drives them toward full‑blown cancer. By tracing this loop step by step, the researchers reveal new molecular "pressure points" that might be targeted to prevent or slow oral tumors.
From normal tissue to dangerous patches
Oral squamous cell carcinoma (OSCC) is a common and deadly cancer of the mouth. It usually develops gradually: healthy lining cells become abnormal (a condition called oral leukoplakia, which appears as white patches) and then progress to cancer. The team examined human samples from normal tissue, leukoplakia, and OSCC, as well as data from public cancer databases and single‑cell sequencing. They found that as tissue moved from normal to precancerous to cancerous, its metabolism shifted: cells relied more on rapid sugar burning (glycolysis) and accumulated higher levels of lactate, a metabolic by‑product. At the same time, a specific chemical mark on DNA‑packaging proteins, called histone lactylation at a site named H3K18, became progressively stronger.
When lactate turns into a genetic switch
Lactate is usually thought of as metabolic waste, but here it acts more like a signal. The researchers showed that high lactate levels feed into histone lactylation, which changes how genes are switched on or off. In both cell cultures and a mouse model of tongue cancer, blocking glycolysis or lowering lactate reduced overall histone lactylation and specifically the H3K18 mark. This dampened cell growth, colony formation, and the ability of cells to move and invade—key features of cancer. In mice exposed to a chemical that induces tongue tumors, treatment with glycolysis blockers shrank lesion areas, reduced the number of cancerous spots, and did not cause obvious liver or kidney damage, suggesting that targeting this axis could be both effective and tolerable.
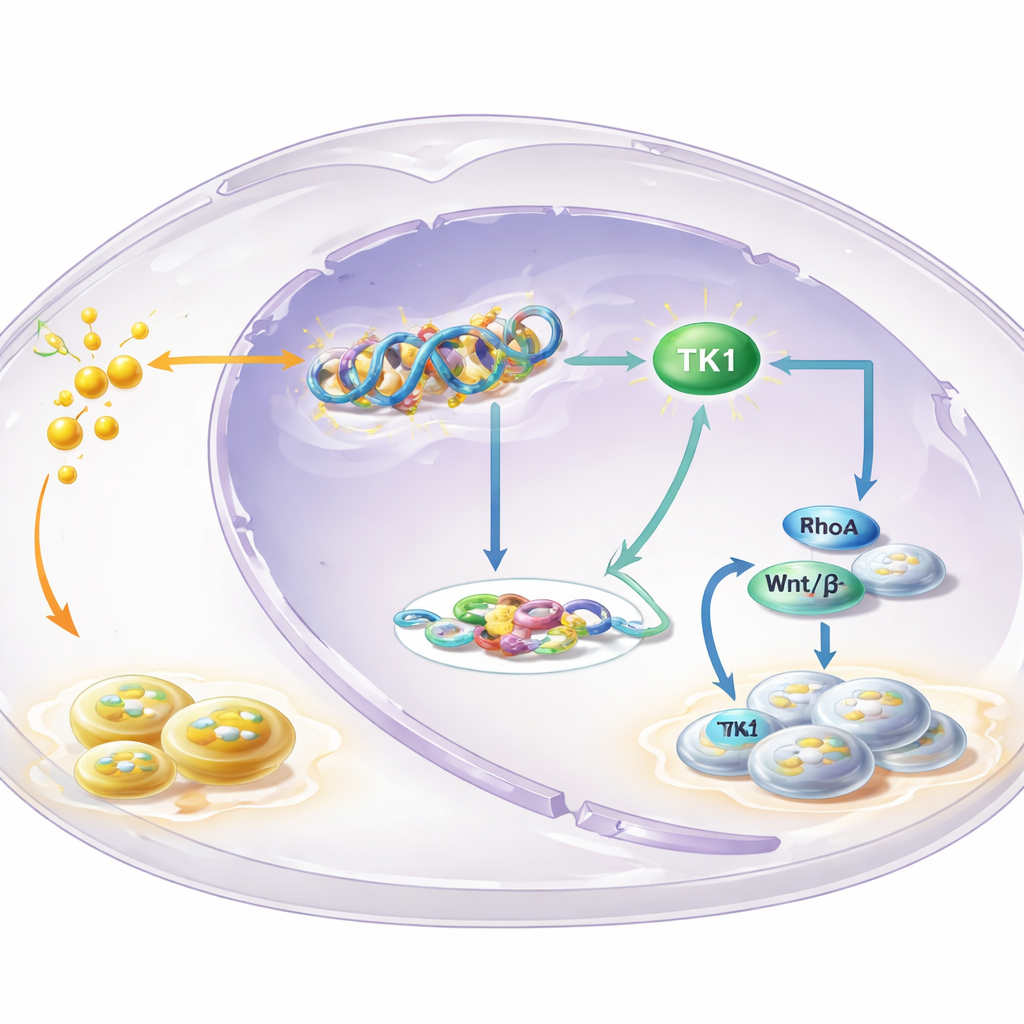
A key enzyme at the heart of the loop
To pinpoint which genes are controlled by H3K18 lactylation, the team mapped where this mark sits on the genome and combined that with gene‑expression data. One standout was thymidine kinase 1 (TK1), an enzyme that helps build the building blocks of DNA. They found that H3K18 lactylation was enriched at the TK1 gene’s control region and that blocking glycolysis reduced TK1’s activity and the pool of DNA precursors in cells, especially one called dTTP. TK1 was more abundant in leukoplakia and OSCC than in normal tissue, and patients whose tumors had higher TK1 levels tended to have worse survival. Experimentally lowering TK1 slowed cell growth, pushed cells to pause in an early phase of the cell cycle, and shrank tumors in mice, marking TK1 as a driver rather than a bystander.
How TK1 pushes signaling and feeds back on metabolism
The story does not stop at DNA building blocks. The products of TK1’s activity alter the balance between two related molecules, GTP and GDP, which control switches known as small GTPases. The study focused on one such switch, RhoA, known to influence cell shape, movement, and growth. When glycolysis was blocked, the GTP/GDP ratio fell and RhoA activity dropped—but both were restored by forcing cells to make more TK1. This, in turn, boosted the Wnt/β‑catenin pathway, a powerful growth‑promoting signaling route. Active β‑catenin increased production of c‑Myc and the glycolytic enzyme LDHA, which further raises lactate levels. More lactate then fuels more histone lactylation and TK1 activation, closing a self‑reinforcing loop that continually pushes cells toward uncontrolled growth.
Different paths in different oral cancers
The researchers also compared HPV‑negative and HPV‑positive oral cancer cells. While blocking glycolysis and lactylation strongly slowed HPV‑negative cells, HPV‑positive cells were less dependent on this pathway, likely because viral cancer‑causing proteins provide alternative growth signals. In patient samples, the H3K18 lactylation mark correlated with abnormal patterns of the tumor‑suppressor protein p53, which is often mutated in OSCC and normally restrains glycolysis and DNA replication. This suggests that loss of p53 and the lactate‑driven loop may cooperate to accelerate early tumor development.
What this means for future treatments
Overall, the study shows that in early mouth lesions, excess sugar burning produces lactate, which does not just sit as waste—it decorates histones, turns on TK1, boosts DNA building and growth signals, and activates a Wnt‑driven cycle that feeds back to generate even more lactate. Breaking this glycolysis–lactylation–TK1–β‑catenin feedback loop at any of its key steps—such as glycolysis, histone lactylation, TK1, RhoA, or Wnt signaling—could offer new ways to prevent leukoplakia from becoming cancer or to treat established OSCC, especially in HPV‑negative patients.
Citation: Wang, Y., Geng, Y., Chen, Y. et al. Histone lactylation-driven feedback loop modulates pyrimidine metabolism to promote oral carcinogenesis. Cell Death Dis 17, 316 (2026). https://doi.org/10.1038/s41419-026-08580-w
Keywords: oral cancer, tumor metabolism, histone lactylation, pyrimidine synthesis, Wnt signaling