Clear Sky Science · en
Aberrant calcium signaling and neuronal activity in the L271H CACNA1D (Cav1.3) iPSC model of neurodevelopmental disease
When a Single Gene Change Ripples Through Brain Development
Rare changes in genes can subtly alter how brain cells handle electrical and chemical signals. This study follows one such change in a calcium channel gene linked to developmental delay, seizures, and autism-like symptoms. By turning a patient’s blood cells into stem cells and then into miniature brain tissues and neurons in a dish, the researchers map how a tiny molecular tweak can distort early brain development and neuronal activity.
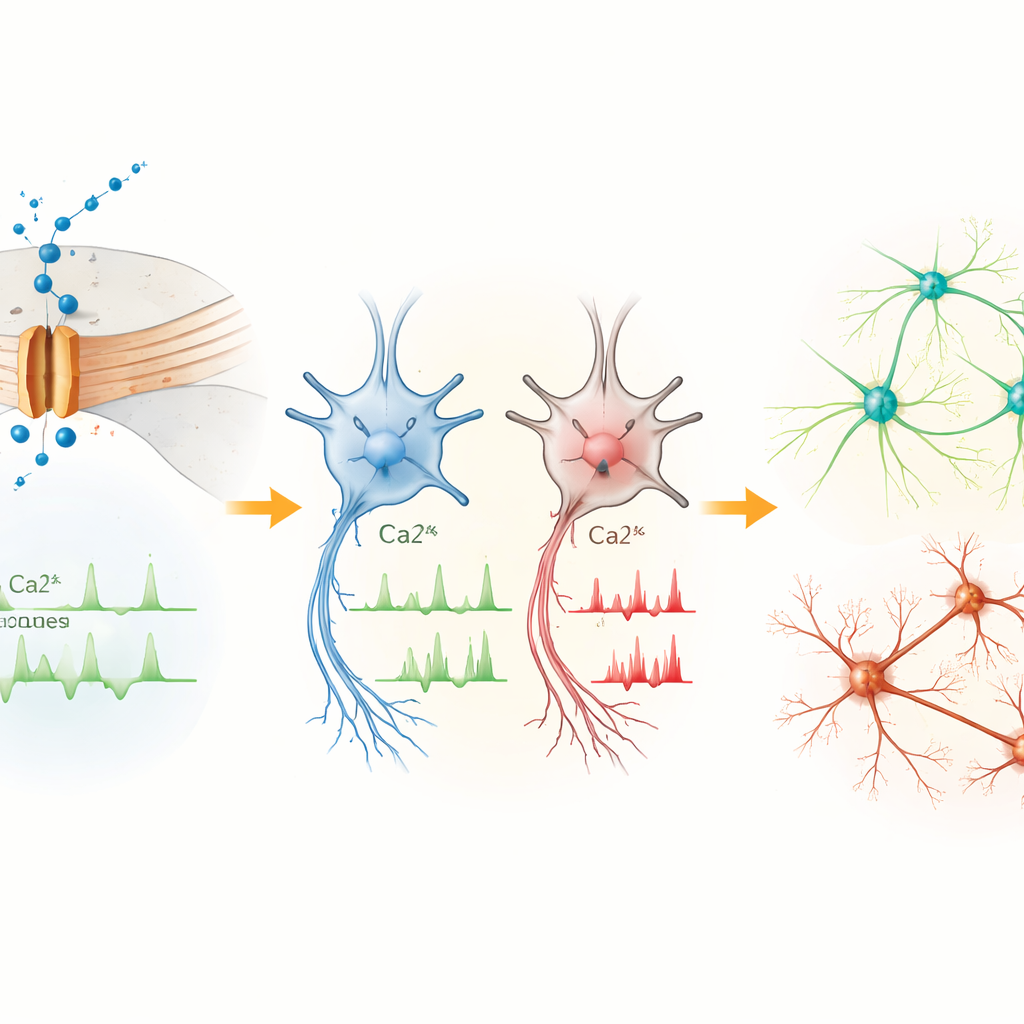
Gatekeepers That Let Calcium Into Brain Cells
Brain cells rely on a controlled trickle of calcium ions to grow, talk to one another, and switch genes on and off. Specialized “gates” in the cell membrane, called voltage-gated calcium channels, open during electrical activity and let calcium flow in. One such gate, known as Cav1.3, is important in many organs, including the brain and hormone-producing tissues. In several patients, rare inherited changes in the Cav1.3 gene have been tied to a mix of symptoms: hormonal imbalances, muscle weakness, seizures, delayed development, and traits on the autism spectrum. The variant studied here, called L271H, had been shown in non‑neuronal cells to make the channel open more easily, but its impact on human brain cells was unknown.
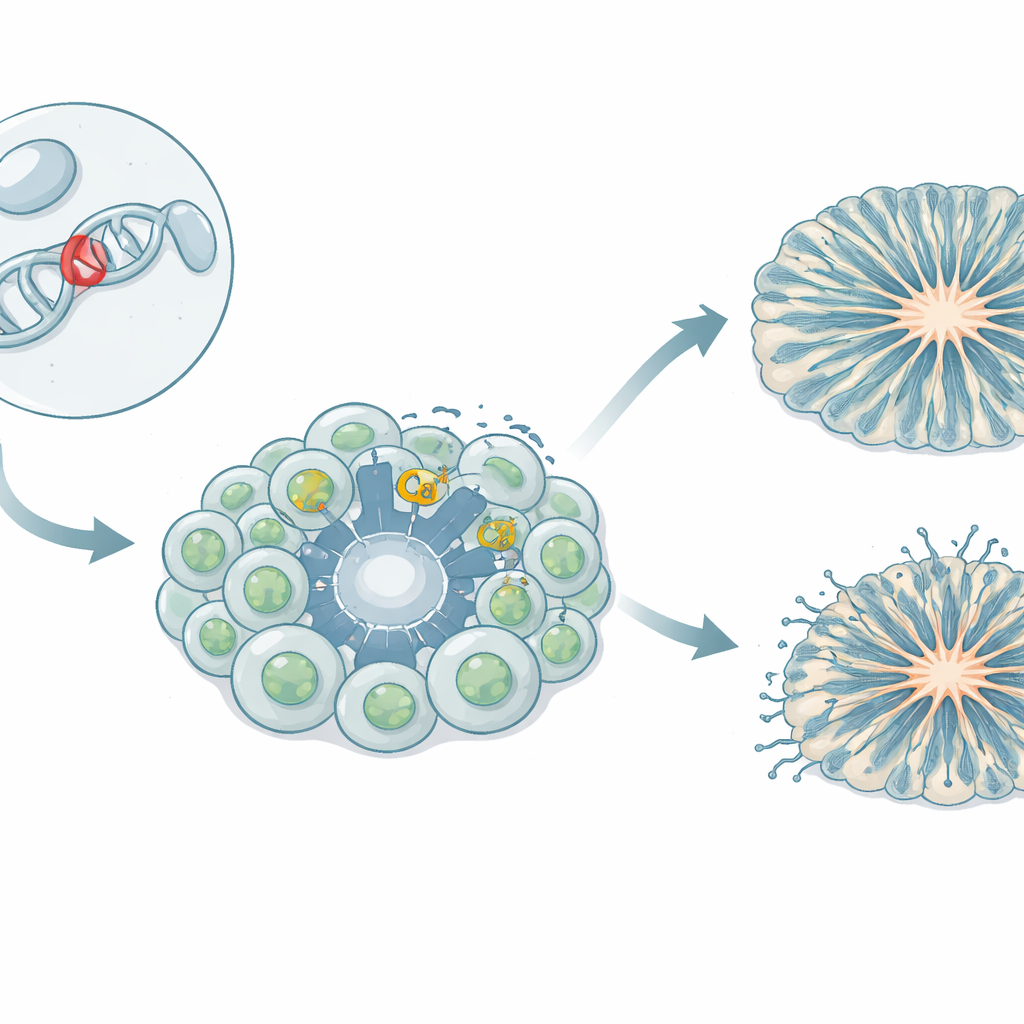
Building Patient-Specific Brain Cells in the Lab
The team created an iPSC model, in which blood cells from a girl carrying the L271H variant were reprogrammed into stem cells capable of forming any tissue. These induced pluripotent stem cells were then guided to become neural progenitor cells—the early building blocks that later generate neurons—and into midbrain neurons related to dopamine signaling. The researchers also grew three-dimensional cortical organoids, tiny “mini-brain” structures that mimic early stages of human cortex formation. By comparing patient-derived cells with several control lines from healthy donors, they could separate the effects of the variant from natural genetic variation.
Early Cells Signal Too Much, Mature Neurons Fire Too Little
In the earliest neural progenitors, the Cav1.3 channel was the dominant form of this calcium gate. Cells carrying the L271H variant showed more frequent spontaneous calcium surges than controls, even though their basic electrical properties were similar. This suggests that, at this stage, the variant mainly disrupts calcium balance inside the cell rather than overt firing of electrical impulses. As cells matured into midbrain neurons, the picture flipped. Although another calcium channel type became more abundant overall, neurons with the L271H variant showed fewer calcium spikes, a more depolarized resting state, and were harder to drive into repeated firing. Their action potentials were smaller and slower to rise and fall, indicating that the variant ultimately dampens the neurons’ ability to send robust electrical signals.
Mini-Brains That Grow Faster but Organize Poorly
In cortical organoids, the L271H variant left a strong mark on structure. Compared with controls, patient-derived organoids had fewer and smaller ventricle-like cavities—ring-shaped zones where progenitor cells normally line up in an orderly fashion. Radial glial cells, which act as scaffolds and guide newborn neurons, were scattered throughout the tissue instead of forming neat, polarized layers. Intermediate progenitor cells and neurons appeared earlier and then declined sooner, suggesting that progenitors were pushed to differentiate too quickly. By 60 days, total neuron numbers caught up, but the early mis-timing and disorganization point to a disrupted blueprint for building cortical circuits rather than an absolute failure to make neurons.
Gene Activity Shifts Toward Neurodevelopmental Risk Programs
To understand how altered calcium handling feeds into gene control, the team sequenced RNA from patient and control cells at different stages. Differences were modest in stem cells but grew larger in progenitors and neurons as Cav1.3 expression increased. Many genes turned up or down in the patient cells are involved in controlling transcription, brain development, and neurogenesis. Several, including PTN, MEIS2, POU3F2, CNTN4, CNTNAP2, and AUTS2, are known risk genes for autism and related neurodevelopmental conditions. These changes suggest that abnormal calcium entry through Cav1.3 reshapes activity-dependent gene programs that guide how brain cells proliferate, migrate, and wire together.
What This Means for Patients and Future Therapies
Put simply, this work shows how a single calcium channel variant can first overstimulate early brain cells, then leave mature neurons underpowered, while at the same time nudging gene networks associated with neurodevelopmental disorders off track. The result is a brain that may look largely normal from the outside yet is built from circuits that developed too fast and organized imperfectly. By recreating this patient’s mutation in human cells and organoids, the study provides a detailed roadmap linking a molecular defect to altered cell signaling, tissue architecture, and gene regulation. This model now offers a platform to test targeted drugs and genetic strategies aimed at restoring more normal calcium signaling in Cav1.3-related channelopathies.
Citation: Tisch, M., Geisler, S.M., Gabassi, E. et al. Aberrant calcium signaling and neuronal activity in the L271H CACNA1D (Cav1.3) iPSC model of neurodevelopmental disease. Mol Psychiatry 31, 2927–2940 (2026). https://doi.org/10.1038/s41380-025-03429-8
Keywords: calcium channels, neurodevelopmental disorders, induced pluripotent stem cells, brain organoids, autism risk genes