Clear Sky Science · en
Parallelized droplet microfluidic mechanoporation enables robust and clogging-resistant intracellular gene delivery
Why this matters for future medicines
Many of the most exciting new therapies—like gene editing and mRNA-based treatments—depend on getting fragile biomolecules safely inside living cells. Today’s methods can be powerful but often damage cells, work inconsistently across cell types, or are hard to scale up to the billions of cells needed for therapies. This paper introduces a new microchip-based approach that delivers genetic material into cells efficiently, gently, and at very high throughput, while solving a stubborn practical problem: devices clogging and failing during operation.
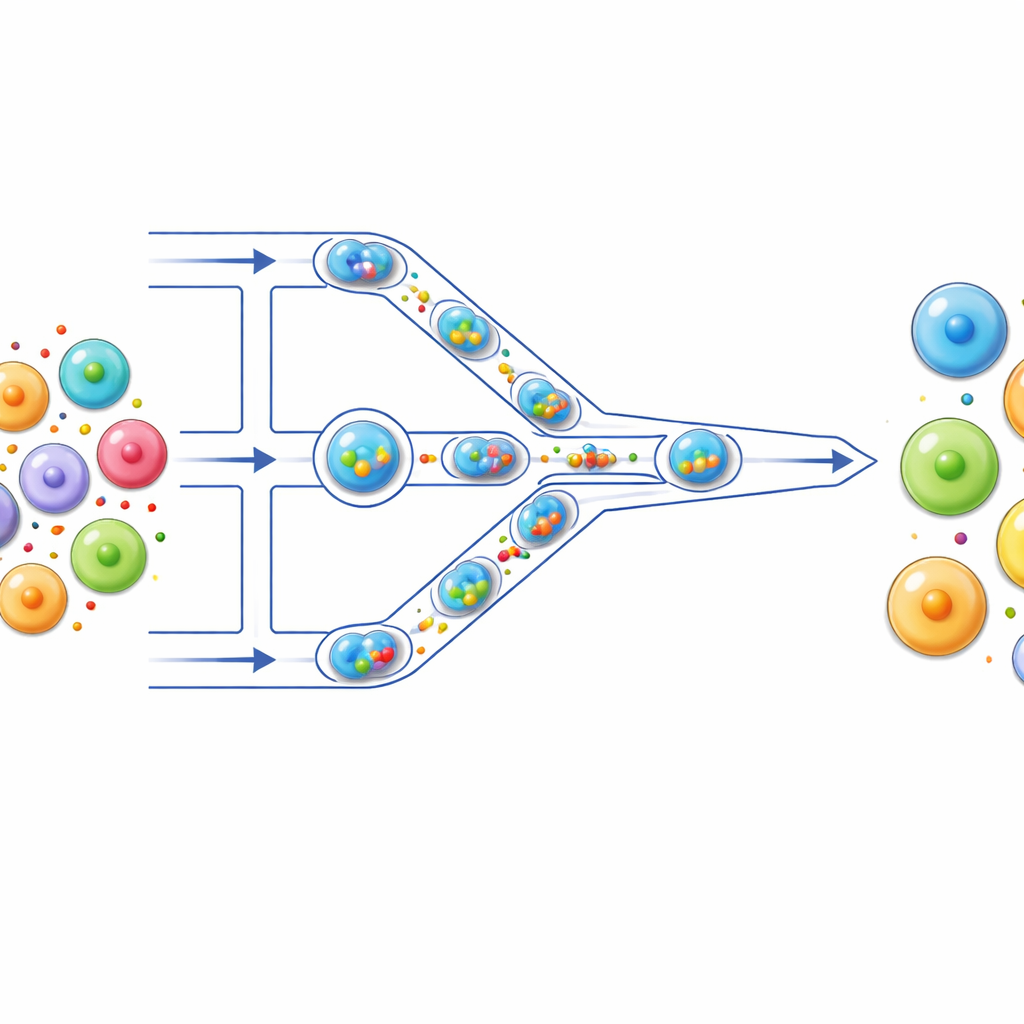
A tiny assembly line for cells
The authors built a microfluidic “assembly line” where cells travel through hair‑thin channels on a transparent chip. First, each cell is packaged together with its cargo—such as fluorescent dye, mRNA, or CRISPR components—inside a microscopic droplet of water surrounded by oil. These droplets act like individual reaction vessels, keeping each cell and its cargo isolated. As droplets are pushed along the channel, they encounter a series of narrow constrictions that are smaller than the cells themselves. When a droplet squeezes through, the cell inside is briefly deformed and then allowed to relax, a cycle that repeats several times as it passes multiple constrictions.
How gentle squeezing opens the cell door
When a soft cell is forced through a constriction that is narrower than its diameter, its membrane stretches, forming temporary nanoscale openings. Because the cell is inside a droplet moving quickly through the constriction, fluid flows across the cell surface and pushes cargo molecules through these tiny openings into the cell interior. Once the droplet exits the narrow region, the cell snaps back to its round shape and the membrane reseals, now with the cargo trapped inside. High‑speed imaging and quantitative measurements showed that higher flow rates and carefully chosen constriction sizes increase membrane stretching and delivery, but the researchers tuned these parameters so that most cells—over 80%—remain alive and healthy.
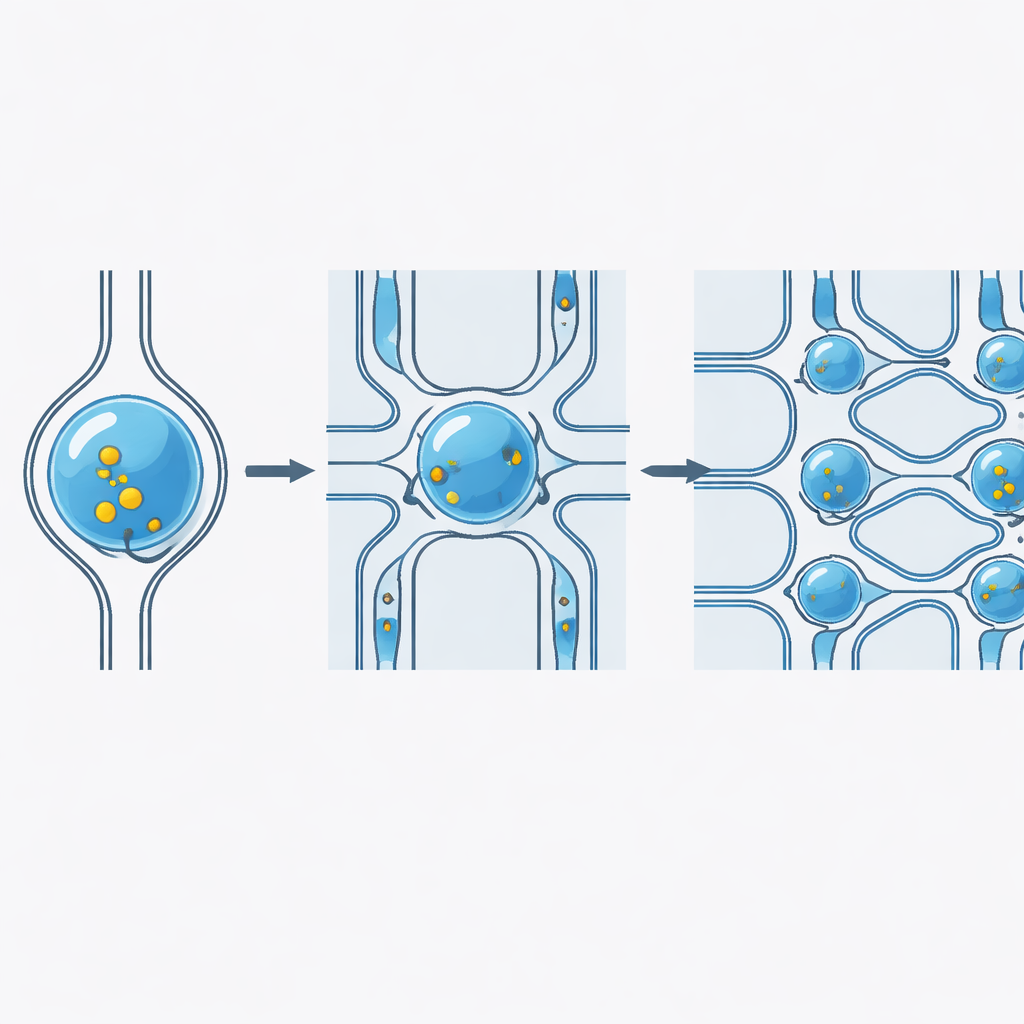
A smart design that resists clogs
A major barrier to using such chips outside the lab is clogging: dust, cell clumps, or bits of material can block a single narrow channel and shut down the system. To overcome this, the team arranged many constrictions in parallel and linked them with bypass channels that allow flow to reroute around obstructions. This parallel, interconnected layout stabilizes pressure inside the chip and keeps droplets moving even when some paths are partially blocked. Tests comparing chips with and without bypasses showed that the new design not only kept delivery efficiency high during clogging events but also improved how much material each cell received, with only modest effects on cell survival.
Works across cargos, cell types, and speeds
The researchers systematically probed how the platform behaves under real‑world conditions. They delivered fluorescent molecules of very different sizes, from small dyes to large polymers, and consistently achieved delivery efficiencies above 98%, with the amount entering each cell controlled by the cargo concentration. The chip worked on a range of human and animal cell lines, including blood‑derived and adherent cells, by adjusting constriction width to match cell size and stiffness. Performance remained stable even when processing up to 100 million cells per hour, indicating that the device can handle the cell numbers required for cell‑based therapies without losing efficiency or cell viability.
From glowing proteins to edited genes
To demonstrate practical impact, the authors delivered mRNA encoding a fluorescent protein into leukemia cells and compared their method to two common non‑viral techniques: lipid‑based transfection and electroporation. Their droplet‑squeezing chip matched or exceeded electroporation in both fraction of cells expressing the protein and intensity of expression, while keeping more cells alive than either alternative. They then used the platform to deliver CRISPR/Cas9 gene‑editing complexes into T cells to knock out a key surface receptor. The edit worked in over 99% of cells, with fluorescence signals from the targeted receptor dropping more than twenty‑fold, and cell viability staying above 80% over several days.
What this means for future cell engineering
In simple terms, this study shows that gently squeezing droplet‑encapsulated cells through a cleverly designed, clog‑resistant microchannel array can load genetic cargo into almost every cell while keeping most of them alive, and can do so at industrially relevant speeds. The platform handles different molecules and cell types, rivals or outperforms standard non‑viral methods, and can be scaled up by adding more channels and droplet generators. With improved cell‑recovery steps, this approach could become a practical workhorse for manufacturing engineered cells for therapies, as well as a versatile tool for basic research that needs reliable, quantitative delivery into large cell populations.
Citation: Liu, Q., Chung, A.J. Parallelized droplet microfluidic mechanoporation enables robust and clogging-resistant intracellular gene delivery. Microsyst Nanoeng 12, 157 (2026). https://doi.org/10.1038/s41378-026-01273-6
Keywords: microfluidic gene delivery, droplet mechanoporation, intracellular mRNA transfection, CRISPR cell engineering, clogging-resistant microdevices