Clear Sky Science · en
Scoping review of preclinical and clinical studies on the role of HMGB1 in heart disease
Why this heart protein matters
Heart attacks and heart failure remain leading causes of death, and doctors still lack treatments that truly repair the heart. This article explores a powerful protein called HMGB1 that behaves like a double-edged sword: in some situations it helps the heart heal, in others it fans the flames of damage. By pulling together hundreds of animal and human studies, the authors ask a simple but crucial question: when is this protein a friend, and when is it a foe—and could we safely target it to prevent or treat heart disease?
A danger signal with two faces
HMGB1 normally lives inside the cell nucleus, where it helps organize DNA and supports basic cell functions. When heart cells are stressed or injured—by a heart attack, infection, chemotherapy, or long-term high blood pressure—HMGB1 can move out of the nucleus into the cell fluid and eventually be released outside the cell. There, it acts as an alarm signal that calls in immune cells. The review shows that this alarm function is not uniform: the chemical “redox” state of HMGB1 and its location inside or outside cells change what it does. A fully reduced form tends to support repair and cell migration, while a partially oxidized form tends to drive inflammation and clot formation.
What the studies reveal across heart diseases
The authors systematically examined 352 experimental and clinical studies covering many heart conditions, including heart attacks, heart failure, myocarditis, sepsis-related heart weakness, diabetes-related heart damage, and chemotherapy-induced cardiomyopathy. Across these disorders, levels of HMGB1 in blood and heart tissue were frequently elevated and often tracked with how sick patients or animals were. Treatments that improved heart function—including certain drugs, antibodies, and plant-derived compounds—were commonly associated with lower HMGB1 levels, suggesting that excessive HMGB1 is usually a marker of worse disease and sometimes a driver of it.
When HMGB1 harms and when it heals
In heart attacks and acute coronary syndromes, HMGB1 is quickly released from dying heart cells and later from immune cells. Higher blood levels tend to correlate with larger heart damage and poorer outcomes. Experiments that added HMGB1 from outside often worsened injury through specific cell-surface sensors that trigger inflammation, while blocking HMGB1 reduced damage. Yet other studies reported the opposite: boosting HMGB1 in heart muscle cells or giving carefully timed doses after a heart attack improved healing, encouraged new blood vessel growth, and limited scarring. Similar contradictions emerged in chronic heart failure models, where nuclear HMGB1 inside heart cells generally protected against long-term stress, but HMGB1 outside cells often promoted harmful thickening and stiffening of the heart.
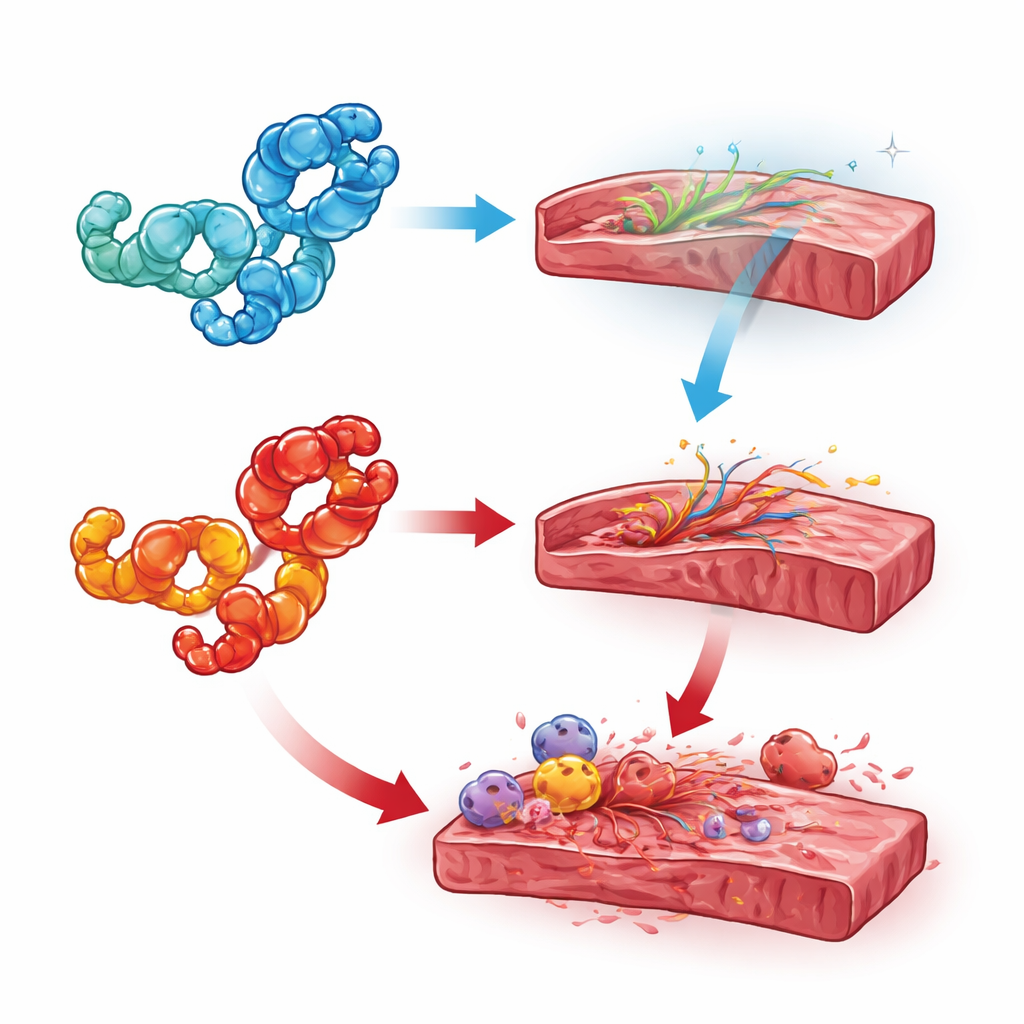
Clues from chemistry and cell location
To resolve these conflicts, the reviewers highlight two key factors: where HMGB1 is and what chemical form it takes. When HMGB1 stays in the nucleus, it appears to guard the heart by supporting DNA integrity and preventing excessive cell death; deleting it from heart cells or immune cells usually made outcomes worse. Once HMGB1 moves into the cytoplasm and is partially oxidized, it can help sustain self-digestion pathways and stress responses that, in excess, become harmful. Outside cells, the fully reduced form tends to cooperate with a natural signaling molecule to guide stem and repair cells to injured tissue, whereas the disulfide (oxidized) form binds different receptors that spark inflammation, fibrosis, and blood clotting. Diseases characterized by ongoing metabolic or inflammatory stress—such as diabetes, sepsis, and autoimmune myocarditis—appear to push HMGB1 toward this damaging oxidized role.
What this means for future treatments
The review concludes that HMGB1 is not simply good or bad; its impact on the heart depends on its chemical state, its location, and the disease context. In broad terms, keeping HMGB1 in the nucleus and preserving its fully reduced form near sites of injury seems beneficial, while sustained accumulation of oxidized HMGB1 outside cells promotes chronic inflammation and scarring. Current tools that block HMGB1 do not distinguish between its helpful and harmful versions, which may explain why some interventions improved outcomes while others worsened them. The authors argue that new laboratory methods to reliably tell these forms apart in blood and tissue, and drugs that selectively target the damaging oxidized version, will be essential if we are to harness HMGB1 safely as a diagnostic marker and therapeutic target for heart disease.
Citation: Mao, SH., Luo, R.GJ., Lee, C. et al. Scoping review of preclinical and clinical studies on the role of HMGB1 in heart disease. npj Cardiovasc Health 3, 20 (2026). https://doi.org/10.1038/s44325-026-00119-4
Keywords: HMGB1, heart disease, inflammation, biomarkers, cardiac repair