Clear Sky Science · en
Using an integrated omics approach to uncover the mechanisms underlying chemotherapy-induced peripheral neuropathy (CIPN)
Why cancer treatment can hurt the nerves
Chemotherapy saves lives, but for many people it comes with a troubling cost: burning, tingling, or numbness in the hands and feet that can last for months or years. This condition, called chemotherapy-induced peripheral neuropathy, can make walking, sleeping, and even buttoning a shirt difficult, and sometimes forces doctors to reduce or stop otherwise effective cancer drugs. The reviewed article explains how cutting‑edge “omics” tools that scan thousands of molecules at once are starting to reveal what goes wrong in nerves during treatment—and how this knowledge could lead to earlier warning signs and gentler, more personalized cancer care.
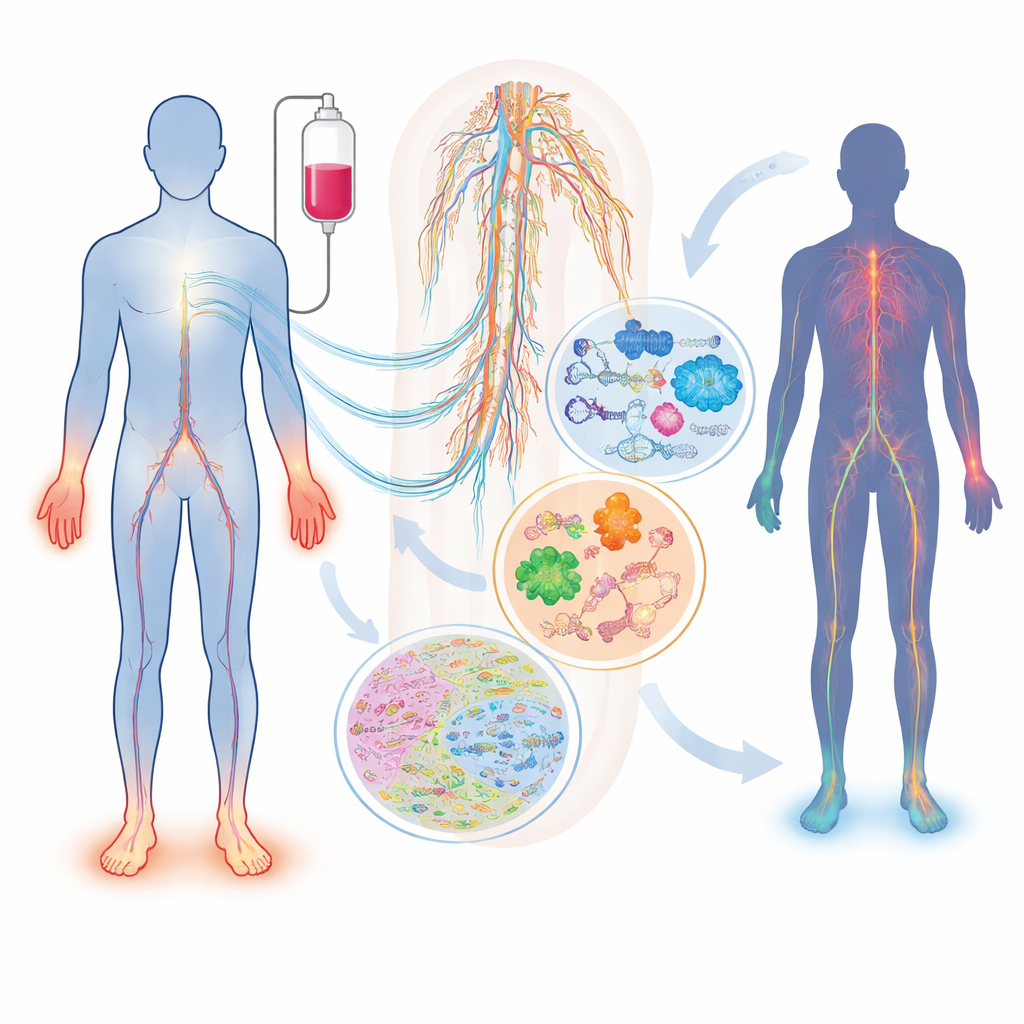
What happens to nerves during chemotherapy
Not all patients exposed to the same drug and dose develop nerve damage, and those who do can experience very different symptoms. The review describes how widely used drugs—such as taxanes (paclitaxel, docetaxel), platinum compounds (cisplatin, oxaliplatin), and proteasome inhibitors (bortezomib, carfilzomib)—can injure sensory, motor, and autonomic nerves in distinct ways. Factors like age, diabetes, vitamin shortages, kidney or thyroid problems, and inherited differences in genes that control drug handling or nerve structure all shift an individual’s risk. One simple blood marker, neurofilament light chain, can already track axonal damage in both animals and patients, but on its own it cannot capture the full complexity of the condition or explain why it develops in the first place.
How omics give a molecular X‑ray of injured nerves
The authors show how transcriptomics (RNA profiling), proteomics (large‑scale protein analysis), and metabolomics (systematic study of small molecules) each offer a different lens on nerve injury. Modern single‑cell RNA sequencing can distinguish many subtypes of sensory neurons and supporting cells in the dorsal root ganglia—clusters of nerve cell bodies near the spine—and track how each type responds to different chemotherapy drugs. For example, specific touch‑sensitive fibers show strong gene changes after paclitaxel, and some genes linked to nerve aging and senescence are activated after cisplatin. Proteomics has revealed early shifts in proteins that maintain the cell skeleton, energy factories, and stress responses, often days before animals show pain behaviors. Metabolomics highlights major disturbances in lipids, amino acids, and bile acids, pointing to problems with energy use, membrane stability, and inflammation that can differ by drug, age, and even gut microbes.
Seeing where damage happens inside tissues
Beyond cataloguing which molecules change, newer “spatial” mass spectrometry methods can show exactly where in the tissue those changes occur. Imaging mass cytometry and mass spectrometry imaging map proteins, lipids, metabolites, and even the chemotherapy compounds themselves directly on nerve sections and spinal cord slices, preserving the architecture of neurons, glial cells, and surrounding tissue. Early work has used these tools to localize pain‑related neuropeptides and to chart how lipids shift in precise regions after nerve injury. They have also tracked long‑lasting deposits of platinum from oxaliplatin in skin, suggesting that drug molecules that linger in non‑cancer tissues may help sustain chronic symptoms. Although only a few such studies have been done specifically in chemotherapy‑treated nerves, the technology is poised to clarify which cell types and micro‑regions are hit first, hardest, and longest.
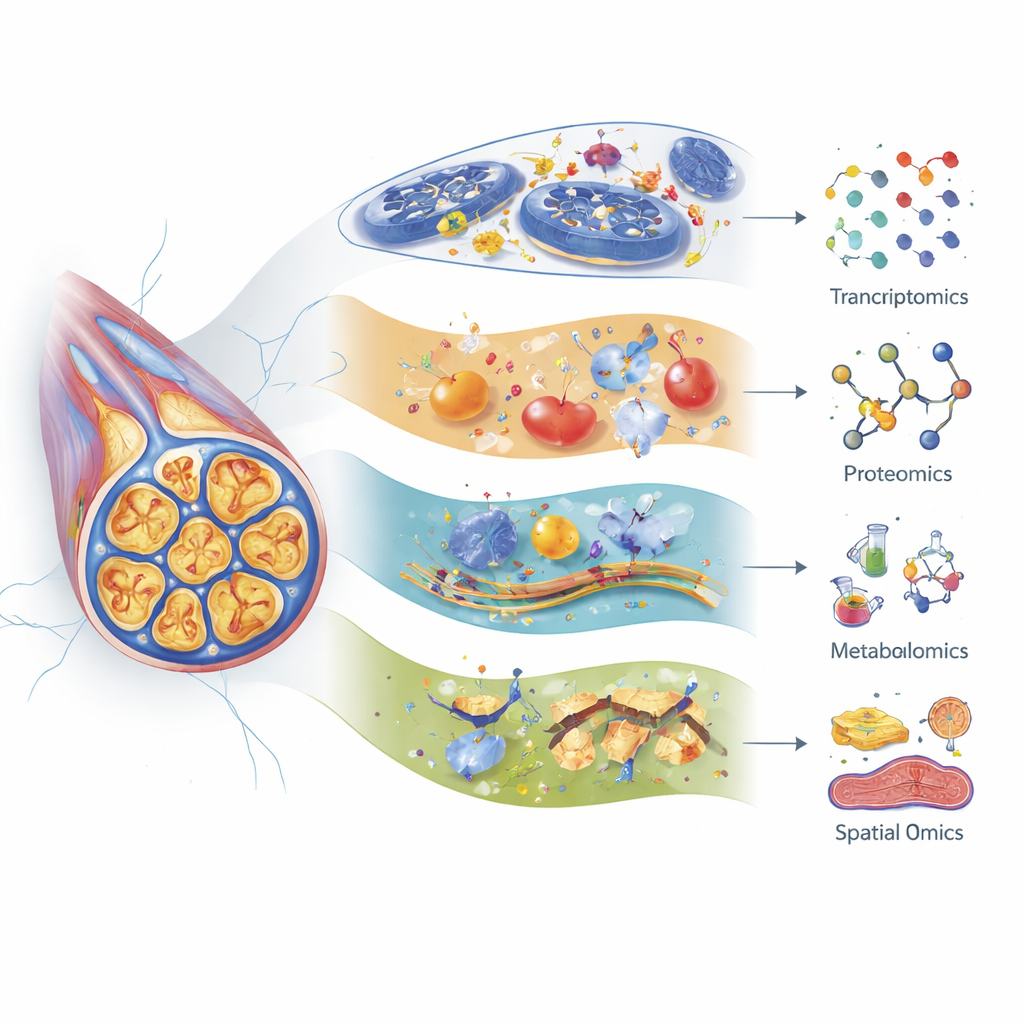
Putting all the molecular pieces together
No single type of omics can fully explain why some patients develop severe neuropathy and others do not. The review argues for integrated, multi‑layer approaches that combine genetic variation, RNA changes, protein networks, metabolite shifts, spatial maps, nerve function tests, and patient‑reported symptoms. Early multi‑omics studies already link coordinated changes in lipid signaling, inflammatory pathways, mitochondrial stress, and nerve‑modulating systems such as endocannabinoids and opioid signaling. Others tie specific lipid messengers and gut‑derived bile acids to pain, suggesting new drug targets like sphingolipid receptors or chemokine receptors that might blunt nerve injury without weakening anti‑cancer effects. Large, carefully designed human studies that align laboratory data with clinical patterns will be crucial to turn these insights into practical tools.
What this means for future cancer care
For a lay reader, the central message is hopeful: by reading the molecular fingerprints left behind in blood, nerve tissue, and even stool, scientists are beginning to predict who is most at risk of nerve damage, understand how different drugs injure the nervous system, and identify weak points in those pathways that could be protected. The authors conclude that integrated omics, especially when applied to human samples over time, could eventually guide doctors in choosing cancer drugs and doses that fit each person’s biology, while also pointing to new medicines that shield nerves from harm. In time, this systems‑level view may make it possible to keep the benefits of powerful chemotherapies while greatly reducing the long‑lasting pain and disability that so often follow.
Citation: Piga, I., Bonomo, R., Chinello, C. et al. Using an integrated omics approach to uncover the mechanisms underlying chemotherapy-induced peripheral neuropathy (CIPN). Commun Med 6, 269 (2026). https://doi.org/10.1038/s43856-026-01622-6
Keywords: chemotherapy-induced peripheral neuropathy, multi-omics, nerve damage, cancer treatment side effects, biomarkers