Clear Sky Science · en
Combinations of colistin and bacteriocins prevent the selection of colistin resistance in Acinetobacter baumannii
Why this matters for hospital infections
Drug-resistant infections picked up in hospitals are becoming harder and harder to treat. One of the most worrying culprits is a bacterium called Acinetobacter baumannii, which can shrug off nearly all available antibiotics. Doctors often have to fall back on colistin, a powerful “last-resort” drug. But even colistin is starting to fail as the bacteria evolve resistance. This study explores whether partnering colistin with natural bacterial peptides called bacteriocins can both kill these stubborn microbes more effectively and slow down the rise of resistance.
The tough germs that outsmart antibiotics
The researchers focused on several recent clinical isolates of A. baumannii, collected from real patients and already resistant to many commonly used drugs. Genetic analysis showed that these strains were diverse, both in their DNA and in the types of resistance genes they carried. Standard tests confirmed that most were still sensitive to colistin, while one strain was highly resistant. When the scientists tried combining colistin with other conventional antibiotics often used in hospitals, such as tigecycline or imipenem, the pairings produced at best an additive effect, not the true synergy that could significantly boost killing and curb resistance.
Natural bacterial weapons join the fight
Many bacteria make their own tiny protein weapons, called bacteriocins, to fend off competitors. The team screened a panel of these molecules and found that several produced by harmless Gram-positive bacteria could inhibit A. baumannii, though only at high doses. They selected two small, helical peptides—Lacticin Z and Enterocin L50—for deeper study. In test-tube membrane models, both strongly attached to mixtures of fats that mimic the inner membrane of A. baumannii and could poke holes in these artificial vesicles. However, intact bacteria were much less affected, suggesting that the bacterium’s outer surface layer acts as a shield, preventing the peptides from reaching their preferred target.
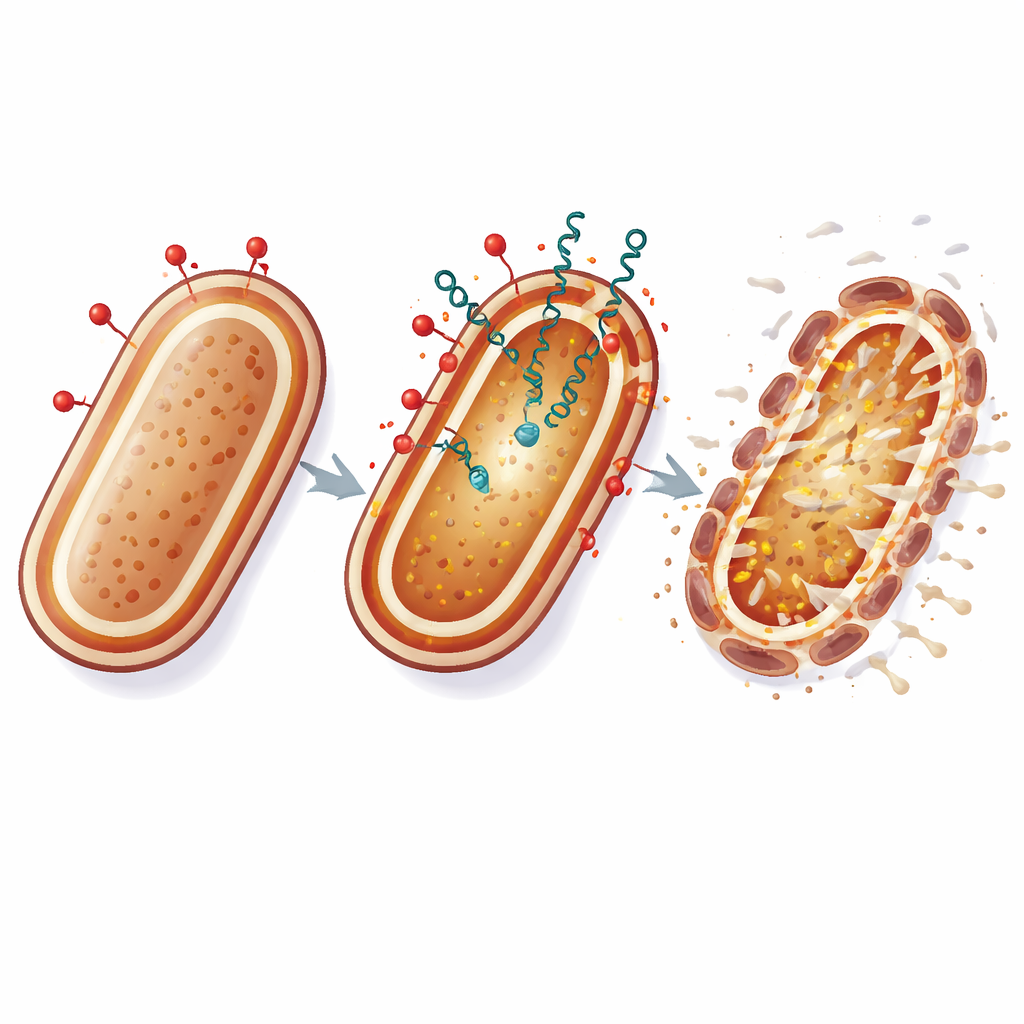
Colistin opens the door, bacteriocins finish the job
Colistin works by binding to a lipid component in the outer coating of Gram-negative bacteria and disturbing the structure of their membranes. In A. baumannii, the authors showed that colistin not only makes the outer membrane leaky but also disrupts the inner membrane’s electrical charge and drains the cell’s supply of ATP, its main energy currency. On its own, colistin treatment often failed to wipe out all cells in a culture: some bacteria survived and, over time, mutants with changes in specific membrane-related genes appeared and became highly resistant. When the researchers added low, otherwise inactive doses of Lacticin Z or Enterocin L50 together with colistin, bacterial survival dropped dramatically. The combination also prevented or greatly reduced the regrowth that usually signaled the rise of resistant mutants, even when they mimicked strains that had partially lost their typical outer-layer molecules.
Targeting the cell envelope from two sides
To understand how this partnership works, the team probed different ways of weakening the bacterial surface. Chemicals that block the synthesis of the outer-layer lipid (lipid A) or that destabilize the outer membrane made A. baumannii more vulnerable to the bacteriocins, supporting the idea that once the outer shield is compromised, the peptides can reach and damage the inner membrane. The bacteriocins also boosted the activity of an outer cell-wall–targeting antibiotic, imipenem, when that drug had already started to disturb the envelope. Importantly, combining colistin with Lacticin Z or Enterocin L50 not only killed more cells but also sharply reduced the appearance of stable, genetically resistant mutants. The surviving cells tended to be transiently tolerant rather than permanently resistant.
What this could mean for future treatments
In everyday terms, colistin acts like a crowbar that pries open the bacterium’s outer armor, while bacteriocins slip through the cracks and attack the inner life-support membrane. By hitting the cell envelope at two vulnerable points at once, this strategy makes it much harder for A. baumannii to adapt through simple genetic tweaks. Although these experiments were done in the lab, the results suggest that carefully chosen combinations of traditional antibiotics with natural bacterial peptides could extend the useful life of last-resort drugs and offer new options against otherwise untreatable hospital infections.
Citation: Rubio, T., zur Nedden, T., Zedek, S. et al. Combinations of colistin and bacteriocins prevent the selection of colistin resistance in Acinetobacter baumannii. Commun Biol 9, 599 (2026). https://doi.org/10.1038/s42003-026-09883-6
Keywords: antibiotic resistance, Acinetobacter baumannii, colistin, bacteriocins, combination therapy