Clear Sky Science · en
Epigenetic landscape of the H3K27me3 mark in macrophages transformed by Theileria annulata
Why a cattle parasite matters to cancer biology
Tropical theileriosis is a severe disease of cattle caused by the parasite Theileria annulata, transmitted by ticks. Once inside the animal, the parasite turns certain white blood cells into endlessly dividing, mobile cells that behave much like cancer. These transformed cells spread throughout the body and drive disease. Intriguingly, when such infected cells are grown in the lab for a long time, they gradually lose their ability to spread. This paper asks a simple but far-reaching question: what changes in the infected cells’ DNA packaging accompany this loss of aggressiveness, and what does that teach us about cancer-like growth more broadly?
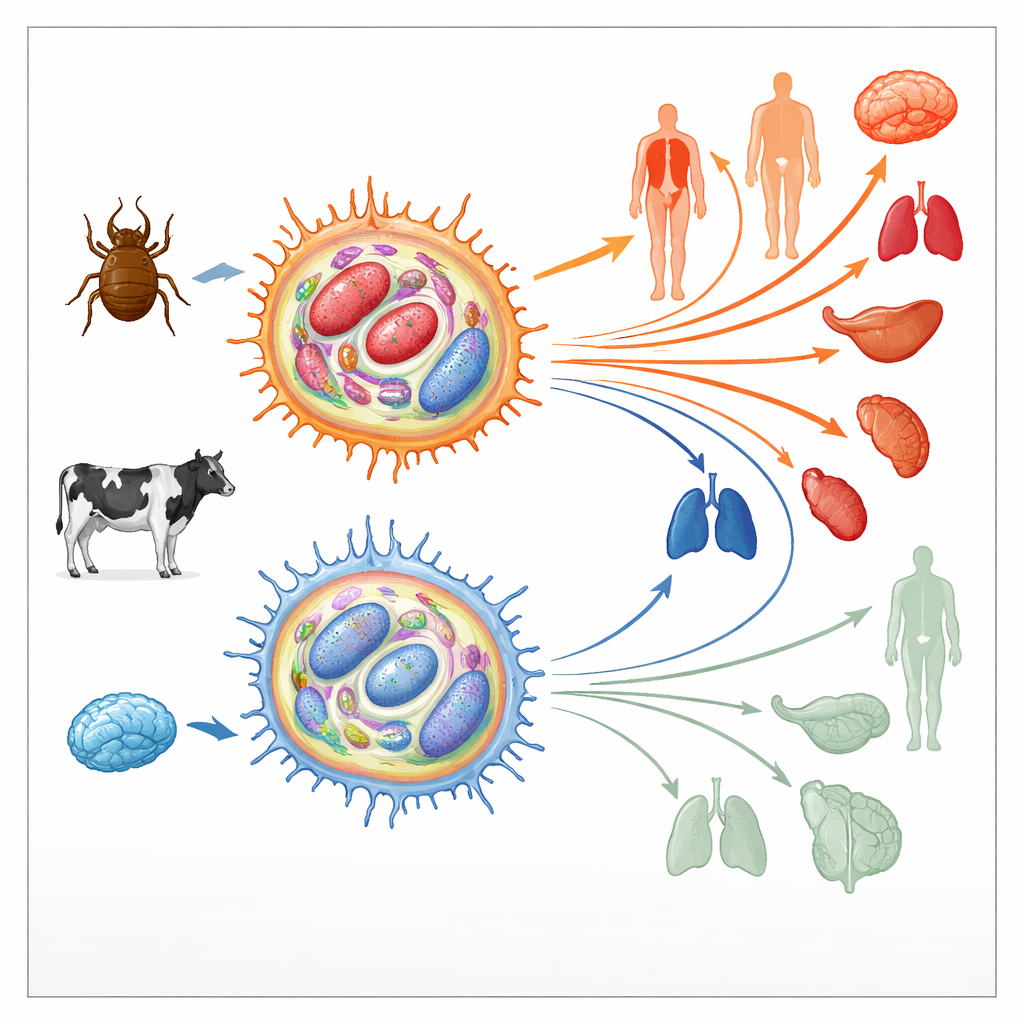
How the parasite retools host immune cells
Theileria annulata lives inside bovine immune cells called macrophages. There it manipulates key signaling pathways that normally control cell death, division, and movement, pushing the host cell into a tumor-like state. These hijacked cells can then divide without limit and migrate through tissues, spreading the parasite. Early in culture, such infected macrophages are highly “virulent,” meaning they disseminate readily in animals. After hundreds of cell divisions, however, their ability to spread drops sharply; they become “attenuated” and are even used as live vaccines. Because similar transitions from aggressive to less aggressive states occur in human cancers, the authors examined whether changes in the cells’ epigenetic landscape—the chemical marks that help turn genes on or off without altering DNA sequence—might underlie this switch.
Focusing on a powerful gene-silencing mark
The study zooms in on a specific chemical tag on histone proteins, called H3K27me3. This tag is added by a protein complex known as Polycomb Repressive Complex 2 (PRC2) and is famous for shutting down genes that control cell identity and development. Using chromatin immunoprecipitation followed by sequencing (ChIP-seq), the authors mapped where H3K27me3 sits across the bovine genome in virulent versus attenuated, parasite-infected macrophages. They also mapped H3K4me3, a mark associated with active genes, for comparison. They confirmed that their antibody detects the host-cell version of H3K27me3 but not the parasite’s own histones, so the maps reflect changes in the cow cell rather than the microbe.
From sharp peaks to broad silent domains
The overall amount of H3K27me3 was higher in attenuated cells, while the activating mark H3K4me3 was lower. More striking than the total levels, however, was the change in pattern. In virulent macrophages, H3K27me3 formed narrow peaks centered near gene start sites, a configuration often linked to precise, PRC2-driven gene control. In attenuated cells, the mark spread out, forming broad domains that extended far into surrounding regions, sometimes covering many neighboring genes. A vivid example is the SKAP2 gene, involved in macrophage adhesion: in virulent cells it carries a small local patch of H3K27me3, but in attenuated cells it lies within a >500 kilobase domain of this repressive mark and is strongly switched down. Across the genome, such spreading was common, suggesting a wholesale remodeling of how DNA is packaged as cells shift from virulent to attenuated behavior.
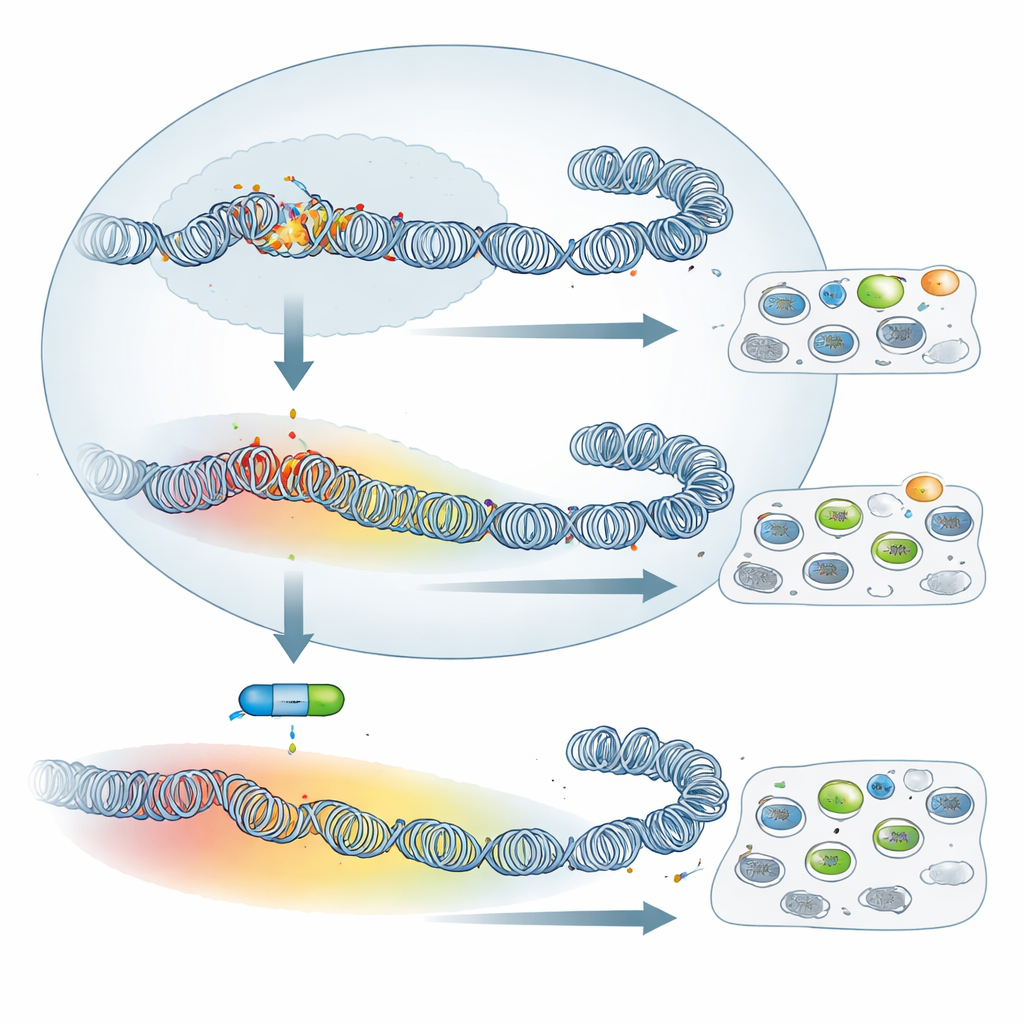
Blocking the silencing machinery has limited impact
Given this sweeping reconfiguration, one might expect that turning off PRC2—and thus erasing H3K27me3—would dramatically change how these cells behave. The team treated both virulent and attenuated macrophages with UNC1999, a drug that inhibits PRC2’s enzyme activity, and verified that H3K27me3 was largely lost. Yet classic tumor-like traits barely budged: the cells’ ability to stick to fibronectin in a dish, and their capacity to disseminate in immune-deficient mice, were not significantly altered by the drug. When the authors examined gene activity by RNA sequencing, they found that many more genes were reactivated in virulent than in attenuated cells. In other words, even though attenuated cells have broader H3K27me3 coverage, their gene silencing depends less on PRC2. Still, a small shared group of genes, including known or suspected tumor suppressors such as Granzyme A and Follistatin, were reawakened by the inhibitor in both cell types, showing that a core PRC2 program remains intact.
What this means for disease and treatment
This work reveals that as parasite-transformed macrophages lose their ability to spread, their epigenetic landscape is profoundly reshaped: a key silencing mark spreads from sharp peaks into wide domains, yet this does not translate into a simple, stronger shutdown of genes. Instead, global PRC2-driven repression is dampened in attenuated cells, while a subset of crucial genes still relies on this machinery to stay off. For livestock health, the findings suggest that targeting PRC2 alone is unlikely to be an effective way to control Theileria-induced tumors in animals. More broadly, the study illustrates how infection can push host cells through cancer-like epigenetic states, and how dramatic changes in chromatin structure do not always have straightforward functional outcomes. Understanding these subtle epigenetic shifts may help refine future strategies that aim to tweak gene regulation in both infectious disease and cancer.
Citation: Sakura, T., Tajeri, S., Rchiad, Z. et al. Epigenetic landscape of the H3K27me3 mark in macrophages transformed by Theileria annulata. Commun Biol 9, 478 (2026). https://doi.org/10.1038/s42003-026-09735-3
Keywords: epigenetics, Theileria, macrophages, histone modification, PRC2