Clear Sky Science · en
SH3 domains selectively activate the PI3 kinase through non-conventional tertiary contacts
How Cells Keep Growth Signals Under Control
Our cells constantly decide whether to grow, divide, or stay quiet. A key decision-maker in this process is an enzyme called PI3 kinase (PI3K), which helps control cell metabolism and survival and is frequently overactive in cancers. This study asks a subtle but important question: when so many similar molecular “plug-and-socket” parts crowd the cell, how does PI3K tell apart harmless encounters from the few that actually flip it from "off" to "on"? The answer reveals a hidden layer of selectivity that could help design more precise cancer therapies.
Many Similar Plugs, Only a Few Real Switches
Cells are full of reusable building blocks called protein domains—compact modules that bind to short stretches of other proteins. One widespread family, the SH3 domains, recognize simple proline-rich motifs that are very common across the human proteome. Because these motifs are so abundant and SH3 domains bind them only weakly and somewhat indiscriminately, it has been puzzling how SH3-containing proteins participate in highly specific signaling pathways without causing chaos. This puzzle is especially pressing for PI3K, whose misactivation drives cancer and other diseases. Earlier work had shown that the regulatory subunit of PI3K, called p85, can be turned on by SH3 domains from certain Src-family kinases, but not others, even though many of these SH3 domains recognize similar proline-rich tags.
The Hidden Helper Contacts on the PI3K Switch
The authors focused on how SH3 domains from different proteins interact with p85 and how this affects PI3K activity. Using precise binding measurements and structural methods, they compared the interaction of the Fyn SH3 domain with a short proline-rich peptide from p85 versus with nearly full-length p85. While the short peptide engaged Fyn in a standard way, the full p85 fragment bound more tightly and with a different energetic signature, hinting that extra surfaces were coming into play. By dissecting p85 into pieces and rebuilding it as chimeras, the team showed that its C‑terminal regions—the two SH2 domains and the coiled iSH2 segment that normally clamp down on the catalytic p110 partner—also participate in this extended, or “tertiary,” contact with SH3.
Two Tiny Spots That Decide Between Binding and Activation
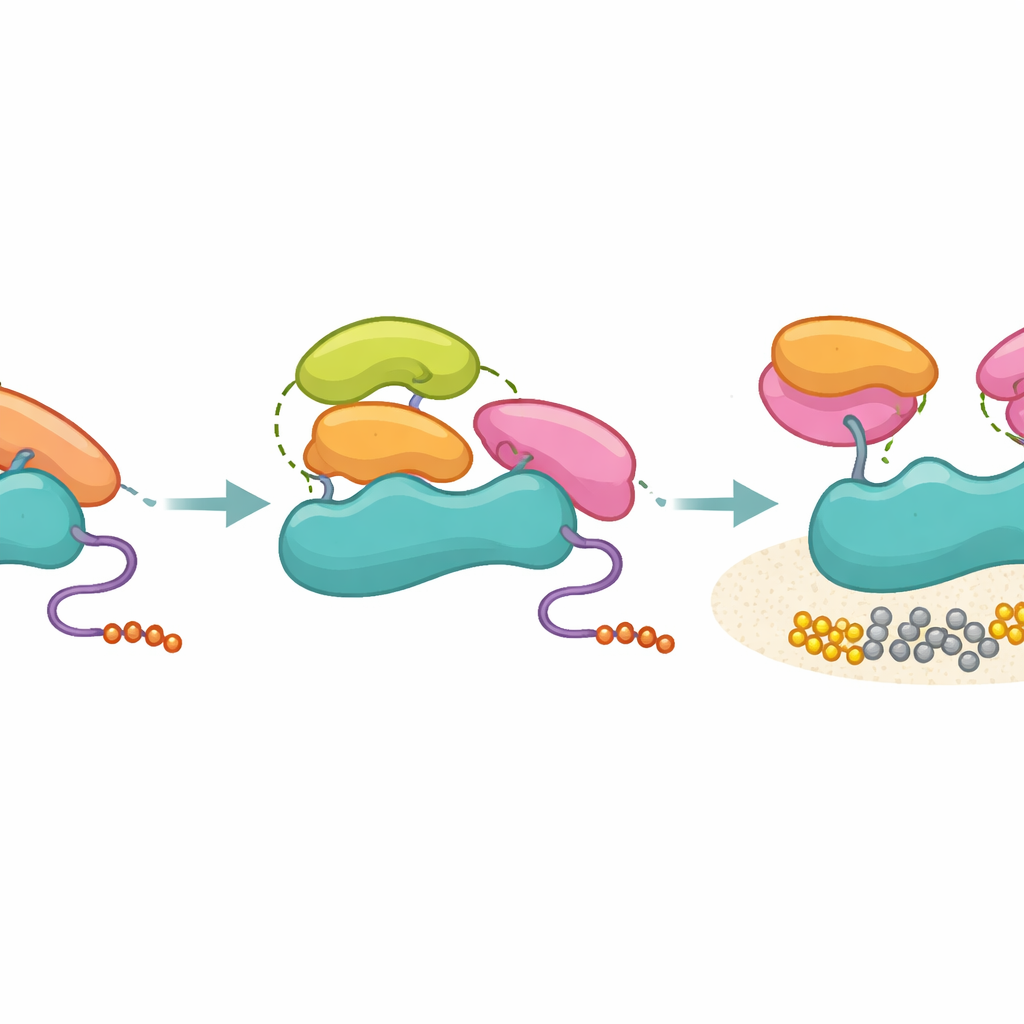
Nuclear magnetic resonance experiments mapped which parts of the Fyn SH3 domain are most disturbed when it binds full p85. Two positions stood out: one near the usual proline-binding groove (called the PR-proximal site) and another on the opposite face of the domain (a PR-distal site). Mutating these positions scarcely changed how well SH3 grabbed the simple proline-rich peptide, but dramatically altered how it engaged full-length p85 and, crucially, how much PI3K activity it triggered. In cell-based lipid kinase assays, some mutants with modest affinity actually activated PI3K more strongly than the wild type, while others with similar affinity lost most of their activating power. This showed that activation does not simply follow “how tight the grip is,” but depends on whether the SH3 domain can form the right tertiary contacts with both inhibitory SH2 regions of p85 and reposition them away from p110.
Separating Mere Contact from a True On-Switch
The researchers then tested SH3 domains from several other proteins, including Hck, Grb2, p85 itself, and Ubs3A, paying special attention to the structural neighborhood of the PR-distal site. All of these SH3 domains could bind p85 with broadly similar strength, yet only those with a compatible hydrophobic patch at the key distal position—like Hck and the C-terminal SH3 of Grb2—were able to turn on PI3K in biochemical assays. Non-activating SH3 domains, including p85’s own SH3, still latched onto p85’s proline-rich region but failed to generate the extra tertiary contact pattern needed to pull the inhibitory SH2 domains away. Crosslinking mass spectrometry supported this view: activating SH3 domains formed focused contacts with the SH2 regions, whereas non-activating ones sampled a wider set of positions, consistent with a looser, non-productive attachment.
What This Means for Disease and Drug Design
Taken together, the work proposes a model in which p85 acts as a molecular filter: it does not prevent many SH3 domains from binding, but it only allows those that can make the right three-dimensional contacts to actually unlock PI3K. In everyday terms, lots of keys fit into the keyhole, but only a few align the internal pins well enough to turn the lock. This separation of binding from activation helps maintain signaling accuracy in the crowded environment of the cell and explains how otherwise “promiscuous” SH3 modules can have specific functional outcomes. Understanding these tertiary contact rules may point the way to drugs that either mimic or block activating SH3 surfaces, offering new strategies to tune PI3K signaling in cancers and other PI3K-driven diseases.
Citation: Aljedani, S.S., Sandholu, A., Aldehaiman, A. et al. SH3 domains selectively activate the PI3 kinase through non-conventional tertiary contacts. Commun Biol 9, 515 (2026). https://doi.org/10.1038/s42003-026-09540-y
Keywords: PI3 kinase, protein signaling, SH3 domain, cancer biology, molecular interactions