Clear Sky Science · en
Design, synthesis, and antibacterial assessment of a new series of ciprofloxacin-based compounds as possible dual DNA gyrase/topoisomerase IV inhibitors
Why this matters for everyday infections
Antibiotic resistance is making once-routine infections harder and more expensive to treat. Ciprofloxacin, a workhorse antibiotic used for urinary, gut, and lung infections, is losing its punch against some bacteria. This study explores a clever way to upgrade ciprofloxacin’s design so that it can hit bacteria harder on two vital weak spots at once, potentially slowing the march of resistance and improving treatment of stubborn infections.
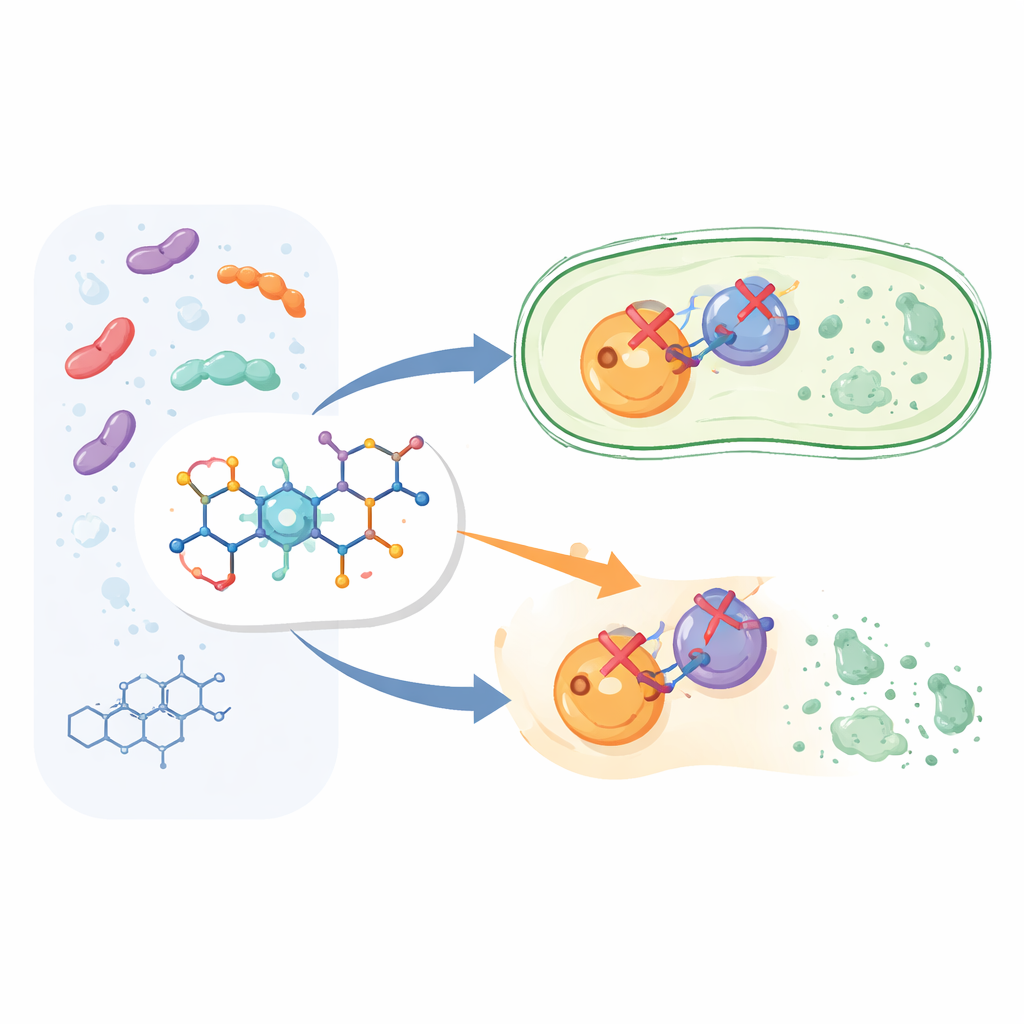
Building a smarter version of a familiar drug
Instead of starting a brand-new drug from scratch, the researchers modified the structure of ciprofloxacin, a member of the widely used fluoroquinolone family. They kept the core of ciprofloxacin that is known to kill bacteria, then attached an additional chemical fragment called an aryl pyridone at a specific position on the molecule. This produced twelve new compounds, labeled 6a through 6l. The idea was that this extra “handle” would help the drug grip its bacterial targets more strongly and in a more versatile way than standard ciprofloxacin.
Targeting two bacterial lifelines at once
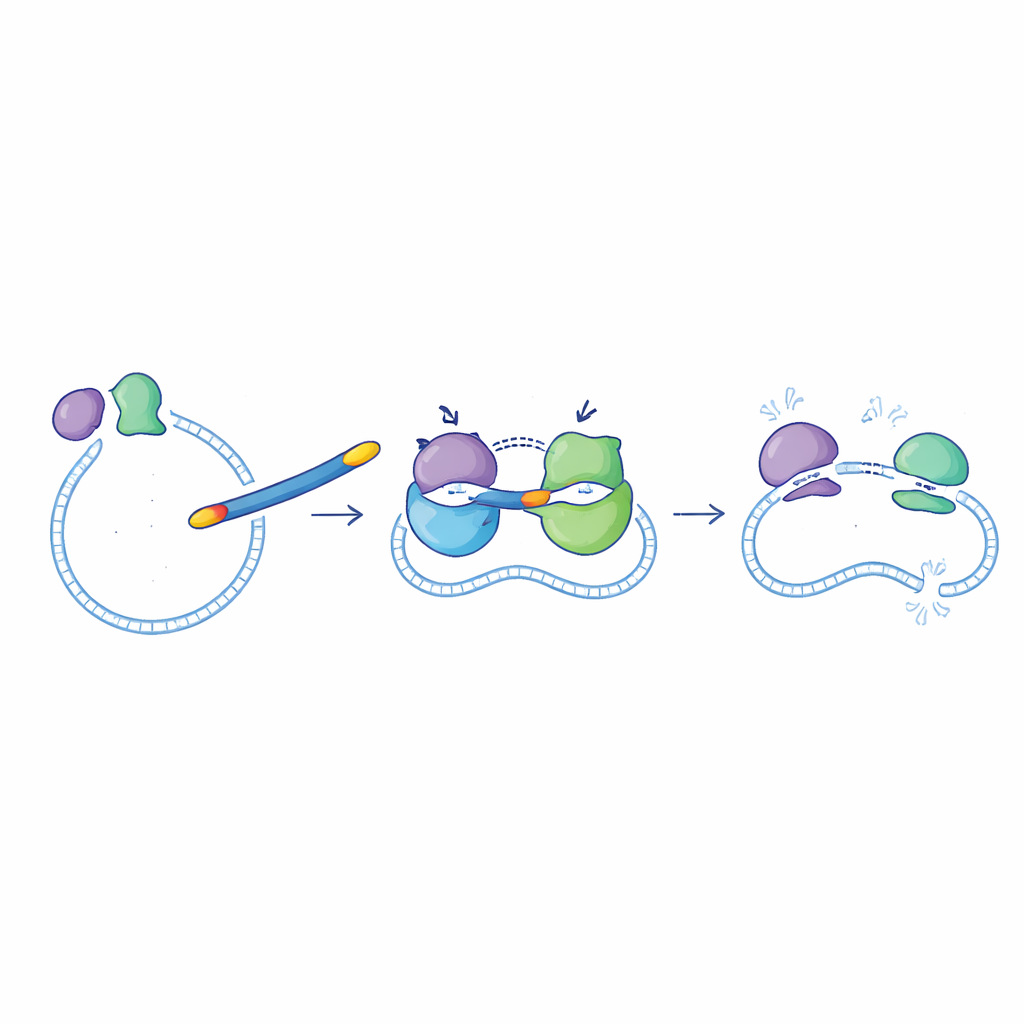
Bacteria rely on two closely related enzymes, DNA gyrase and topoisomerase IV, to manage their DNA as they grow and divide. Ciprofloxacin already interferes with these enzymes, but not equally well. The new compounds were designed to be strong blockers of both targets at the same time, which should make it harder for bacteria to escape by mutating just one enzyme. In enzyme tests using Escherichia coli proteins, most of the new molecules inhibited DNA gyrase and topoisomerase IV at useful levels. One compound, called 6g, stood out: it blocked DNA gyrase slightly better than ciprofloxacin and inhibited topoisomerase IV about seven times more strongly, making it the most powerful “dual-action” molecule in the set.
Putting the new molecules to the test against bacteria
The team then checked whether the enzyme improvements translated into better performance against real bacteria grown in the lab. Selected top performers, including 6d, 6f, 6g, 6i, and 6l, were tested against two Gram-negative species (E. coli and Pseudomonas aeruginosa) and two Gram-positive species (Staphylococcus aureus and Bacillus subtilis). Overall, these new compounds worked best against Gram-negative bacteria, where their ability to cross the outer membrane is especially important. Compound 6g again emerged as the front-runner: its minimal effective doses against E. coli and P. aeruginosa were in the same range as ciprofloxacin, and it remained reasonably active against S. aureus, though it was less potent there than the original drug.
Fighting bacterial communities and probing safety
Beyond free-floating cells, bacteria often hide in slimy communities called biofilms, which make infections more persistent and harder to eradicate. The researchers found that 6g strongly suppressed E. coli biofilms, cutting them by 96 percent at the same concentration needed to halt growth, and still showing notable activity at lower doses. Early safety checks in a normal human breast cell line suggested that 6g is not toxic at levels much higher than those needed to affect bacteria, an encouraging sign for future development, although far from a full safety assessment.
Looking inside the molecular handshake
To understand why 6g performs so well, the team used computer modeling to examine how the molecule fits into the pockets of DNA gyrase and topoisomerase IV. Simulations showed that 6g forms a dense network of interactions—hydrogen bonds, contact with charged amino acids, and snug packing against the protein surface—that are stronger and more persistent than those of ciprofloxacin. Additional calculations of how the protein–drug complexes move over time suggested that 6g helps hold the enzyme structures in a stable, locked state, consistent with its potent enzyme-blocking behavior. Other analyses of absorption and metabolism pointed to acceptable fat–water balance and limited interference with liver enzymes, but also hinted that the compound may not be ideal for simple oral dosing without further optimization.
What this could mean for future antibiotics
Taken together, the results highlight compound 6g as a promising starting point for next-generation antibiotics. It is engineered to shut down two essential bacterial enzymes at once, shows strong activity—especially against tough Gram-negative species—and disrupts protective biofilms, all while appearing non-toxic in an initial human cell test. At the same time, its large size and modest predicted absorption mean that 6g is better viewed as a lead scaffold than a finished medicine. With further fine-tuning to improve how it enters the body and bacteria, this dual-action approach may help extend the useful life of ciprofloxacin-like drugs and offer new options against resistant infections.
Citation: Al-Wahaibi, L.H., Alzahrani, H.A., Bräse, S. et al. Design, synthesis, and antibacterial assessment of a new series of ciprofloxacin-based compounds as possible dual DNA gyrase/topoisomerase IV inhibitors. Sci Rep 16, 13911 (2026). https://doi.org/10.1038/s41598-026-50106-z
Keywords: antibiotic resistance, ciprofloxacin derivatives, DNA gyrase inhibitors, topoisomerase IV, Gram-negative bacteria