Clear Sky Science · en
A complex interplay of various intracellular motifs determines G protein binding and activation of muscarinic receptors
Why tiny cell switches matter for medicine
Every second, our cells rely on tiny molecular switches called receptors to sense hormones, neurotransmitters, and drugs. A major class of these switches, known as G protein–coupled receptors, sits in the cell membrane and passes messages inside via partners called G proteins. Some receptors talk to just one type of G protein, while others broadcast to several, shaping everything from heart rate to mood. This study asks a deceptively simple question with big implications for drug design: what features inside a receptor decide which G proteins it talks to—and how strongly?
Cell phone towers on the membrane
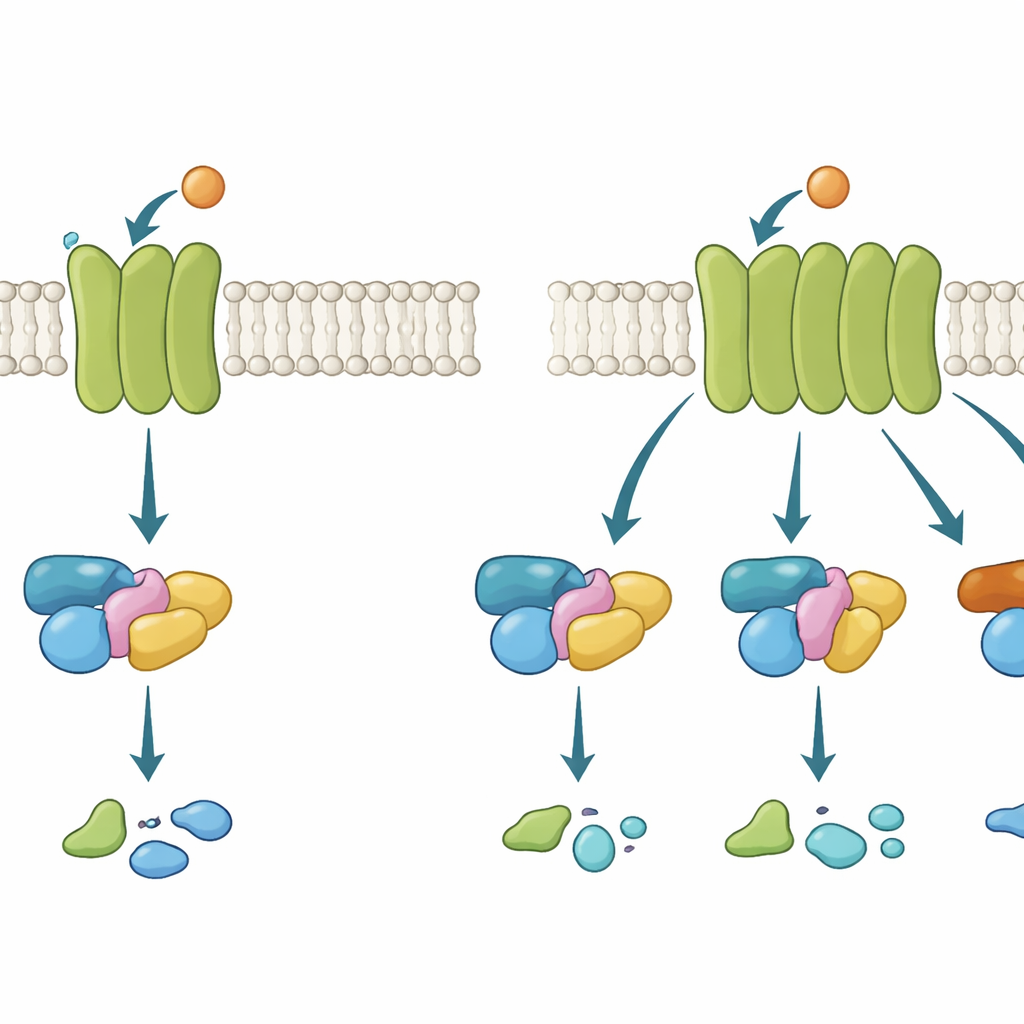
G protein–coupled receptors (GPCRs) act a bit like cellphone towers embedded in the cell’s outer shell. When a signal molecule, such as the neurotransmitter acetylcholine, binds on the outside, the receptor changes shape on the inside and recruits a G protein. That G protein then switches from an inactive to an active state and passes the message along. There are several G protein families, each triggering different cellular responses. Some GPCRs are highly picky, using just one family; others are more promiscuous, engaging several. Understanding what makes a receptor selective or flexible is crucial, because many blockbuster drugs work by nudging these pathways.
Swapping parts to rewrite the signal
The researchers focused on two closely related acetylcholine receptors in the brain and body: the M2 receptor, which normally signals mainly through a Gi/o-type G protein, and the M3 receptor, which prefers a Gq-type G protein but can also weakly engage Gi/o. Rather than guessing at single “magic” amino acids, the team took a modular approach. They built chimeric receptors—hybrids in which they swapped entire internal segments between M2 and M3, such as the short loops inside the cell, the inner ends of two key helices (called 5 and 6), a short tail helix, and stretches of the tail rich in positively charged residues. They then asked two separate questions for each chimera: does it bind to a given G protein, and if so, does it successfully activate it?
Watching binding and activation separately
To track these microscopic interactions, the team used light-based methods in living cells. In a first set of experiments, they used a technique called FRET to detect how stably a receptor holds onto a G protein in a controlled setting where the usual energy-carrying nucleotide GTP is absent. This reveals how likely and how long a receptor–G protein pair stays together. In a second set of experiments, they used a related method, BRET, with specialized sensor constructs to measure whether the bound G proteins actually flip into their active state. By comparing how wild-type and chimeric receptors handled two G protein families—Gi/o and Gq—the researchers could tease apart which structural segments favored which partner and at which step.
Many small changes, big shifts in behavior
The results showed that no single internal feature acted as an on–off switch for G protein choice. Instead, combinations of motifs acted together, and their effects depended on the receptor background. For example, transplanting the inner ends of helices 5 and 6 from the more flexible M3 receptor into the selective M2 receptor shifted it to signal mainly through Gq rather than Gi/o, in some cases turning M2 into a predominantly Gq-coupled receptor. Adding further loop segments could restore the ability to bind and activate both G protein families. Conversely, inserting M2-like segments into M3 could make it favor Gi/o more strongly or, with changes in its tail, push it toward signaling almost exclusively through Gq. Strikingly, some chimeras could bind a G protein without activating it, while others could still trigger activation even when binding was too fleeting to detect with the binding assay.
What this means for future drugs
In everyday terms, this work shows that GPCRs choose their signaling partners not through a single keyhole but through a whole combination lock of internal parts. The inner loops, the inner ends of certain helices, and the tail all contribute in a coordinated way to whether a receptor prefers one G protein family, several, or can bind without truly turning them on. By artfully reshuffling these segments, the authors could reprogram receptors from selective to broadly signaling and back again. For drug discovery, this suggests that tuning how a receptor uses its various inner “dials” could let future medicines steer signals toward helpful pathways and away from side effects, without needing to completely redesign the receptor itself.
Citation: Kirchhofer, S.B., Jelinek, V., Klingelhöfer, K. et al. A complex interplay of various intracellular motifs determines G protein binding and activation of muscarinic receptors. Sci Rep 16, 12370 (2026). https://doi.org/10.1038/s41598-026-48667-0
Keywords: G protein coupled receptors, muscarinic receptors, signal transduction, receptor selectivity, drug targeting