Clear Sky Science · en
Montelukast attenuates diclofenac sodium-induced cardiotoxicity in male rats via targeting Wnt/β-catenin pathway
Why pain relievers and heart health matter
Many people rely on non‑steroidal anti‑inflammatory drugs (NSAIDs) like diclofenac to manage everyday aches, arthritis, and injuries. But growing evidence suggests that long‑term use of some of these medicines can quietly strain the heart. This study explores whether montelukast—an asthma drug that is already widely prescribed—can shield the heart from diclofenac‑related damage, and it uncovers a key cellular signaling route that may explain both the harm and the protection.
A closer look at a common painkiller
Diclofenac is one of the world’s most frequently used pain and anti‑inflammatory medicines. It works by blocking enzymes that make prostaglandins, substances involved in pain, fever, and inflammation. However, this same action can disturb delicate balances that protect the stomach, kidneys, liver, and heart. Earlier work in animals and humans has linked prolonged diclofenac use to heart problems, including tissue damage and impaired function. Scientists suspect that this damage is driven by a toxic combination of excess reactive oxygen molecules, runaway inflammation, scarring of heart tissue, and programmed cell death in heart muscle cells.
The signal switch inside heart cells
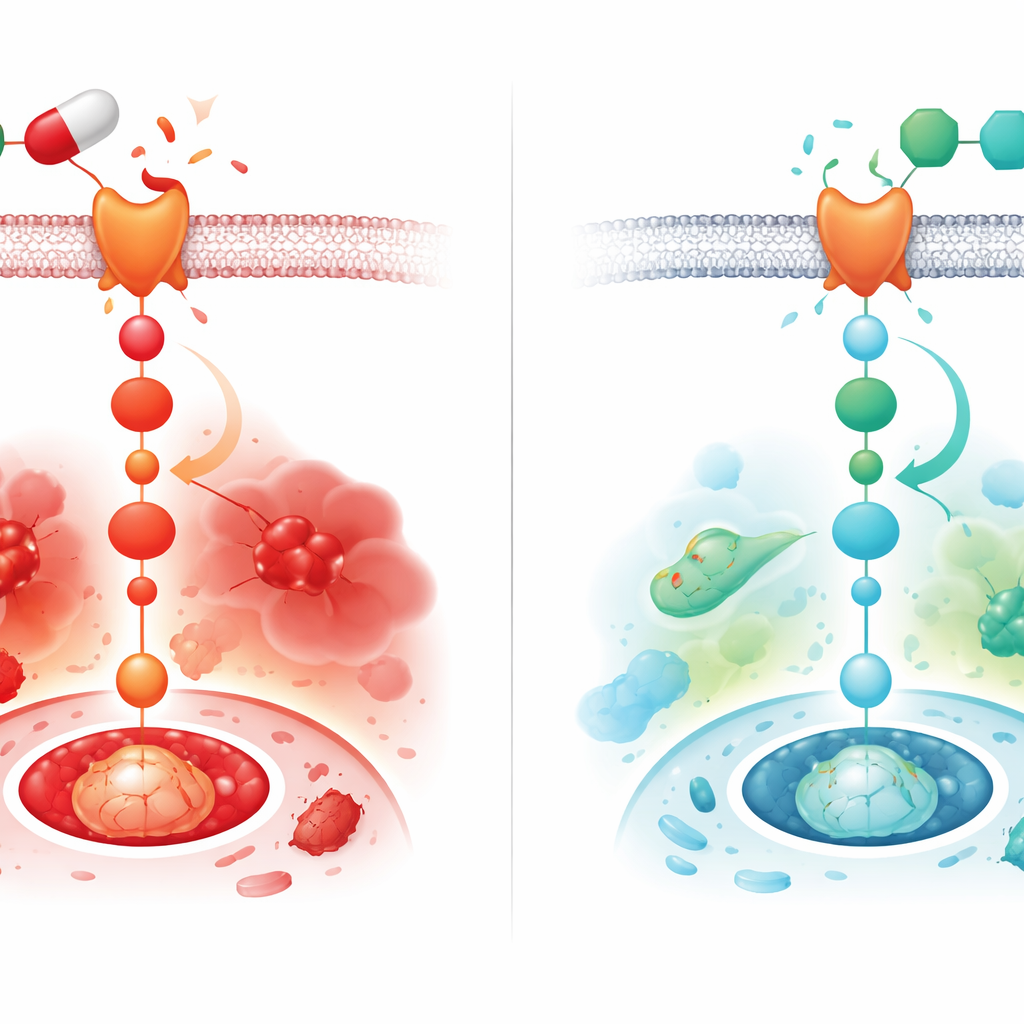
To understand how diclofenac injures the heart, the researchers focused on a cellular communication route known as the Wnt/β‑catenin pathway. This pathway helps control how cells grow, divide, and survive. In a healthy adult heart it is usually kept on a low setting, but in many forms of heart disease it becomes abnormally active and is linked to oxidative stress, inflammation, cell death, and fibrosis (the buildup of stiff scar tissue). In male rats given diclofenac for two weeks, the team found strong signs that this pathway was switched on: key proteins in the Wnt system were markedly increased in heart tissue. At the same time, blood tests and tissue samples showed clear evidence of heart injury, including leakage of heart enzymes into the bloodstream, swollen and degenerating heart muscle fibers, and increased heart size relative to body weight.
Testing a protector: the asthma drug montelukast
Montelukast is best known as a pill that blocks leukotrienes—chemical messengers involved in asthma and allergies. Interestingly, previous animal studies suggested that montelukast also calms inflammation and oxidative stress in the heart. In this study, rats received montelukast alongside diclofenac. Compared with animals that got diclofenac alone, the co‑treated rats had far lower levels of heart injury markers in the blood and healthier‑looking heart tissue under the microscope. Their heart‑to‑body‑weight ratio was closer to normal, suggesting less swelling and thickening. Within the heart muscle, harmful oxidative changes were reduced: a marker of fat damage (malondialdehyde) fell, while natural antioxidant defenses (glutathione and superoxide dismutase) rebounded.
Dialing down inflammation, scarring, and cell death
Diclofenac alone triggered a surge of inflammatory signaling molecules in the heart and increased activity of caspase‑3, a protein that drives cells toward self‑destruction. It also reduced levels of Bcl‑2, a protein that normally helps cells survive. At the tissue level, collagen fibers accumulated between heart muscle cells, a hallmark of fibrosis that can stiffen the heart and impair pumping. Montelukast co‑treatment reversed many of these changes: inflammatory cytokines dropped, caspase‑3 levels declined, Bcl‑2 rose, and collagen buildup was markedly less pronounced. Importantly, the same treatment substantially blunted the diclofenac‑induced rise in Wnt‑1, active β‑catenin, and the receptor FZD‑1, tying the drug’s protective effect to a quieter Wnt/β‑catenin signal inside heart cells.
What this could mean for people using pain medicine
The findings suggest that diclofenac damages the heart in part by flipping on the Wnt/β‑catenin switch, which in turn fuels oxidative stress, inflammation, scarring, and cell death. In rats, montelukast turned this switch down and softened the damage on multiple fronts. Because montelukast is already widely used in humans and has a known safety profile, it emerges here as a promising candidate to protect the heart during long‑term NSAID therapy. Still, the work was done in male rats and focused on molecular and tissue changes rather than direct heart function. Before doctors could consider pairing these drugs in patients, future studies will need to confirm the pathway’s role using specific Wnt blockers, test females, measure heart performance, and determine whether similar benefits—and safe doses—translate to people.
Citation: Alsanea, S., Albuhayri, S., Alkharashi, L. et al. Montelukast attenuates diclofenac sodium-induced cardiotoxicity in male rats via targeting Wnt/β-catenin pathway. Sci Rep 16, 11717 (2026). https://doi.org/10.1038/s41598-026-46514-w
Keywords: diclofenac cardiotoxicity, montelukast, heart inflammation, oxidative stress, Wnt beta-catenin pathway