Clear Sky Science · en
Molecular basis for inhibition of α-thrombin activity by bacterial lipopolysaccharides
Why Bacterial Toxins Matter for Blood Clots
Severe infections caused by Gram-negative bacteria can push the body into a dangerous spiral where the blood begins to clot uncontrollably, blocking small vessels and damaging organs. This study asks a deceptively simple question with big implications: when pieces of bacterial cell walls enter the bloodstream, do they physically tamper with thrombin, the key enzyme that makes blood clots? By uncovering how specific bacterial molecules interact with thrombin, the work sheds light on why sepsis-related clotting problems vary so much from patient to patient and may point toward new ways to predict or even control these life-threatening events.
Pieces of Bacteria That Talk to Blood
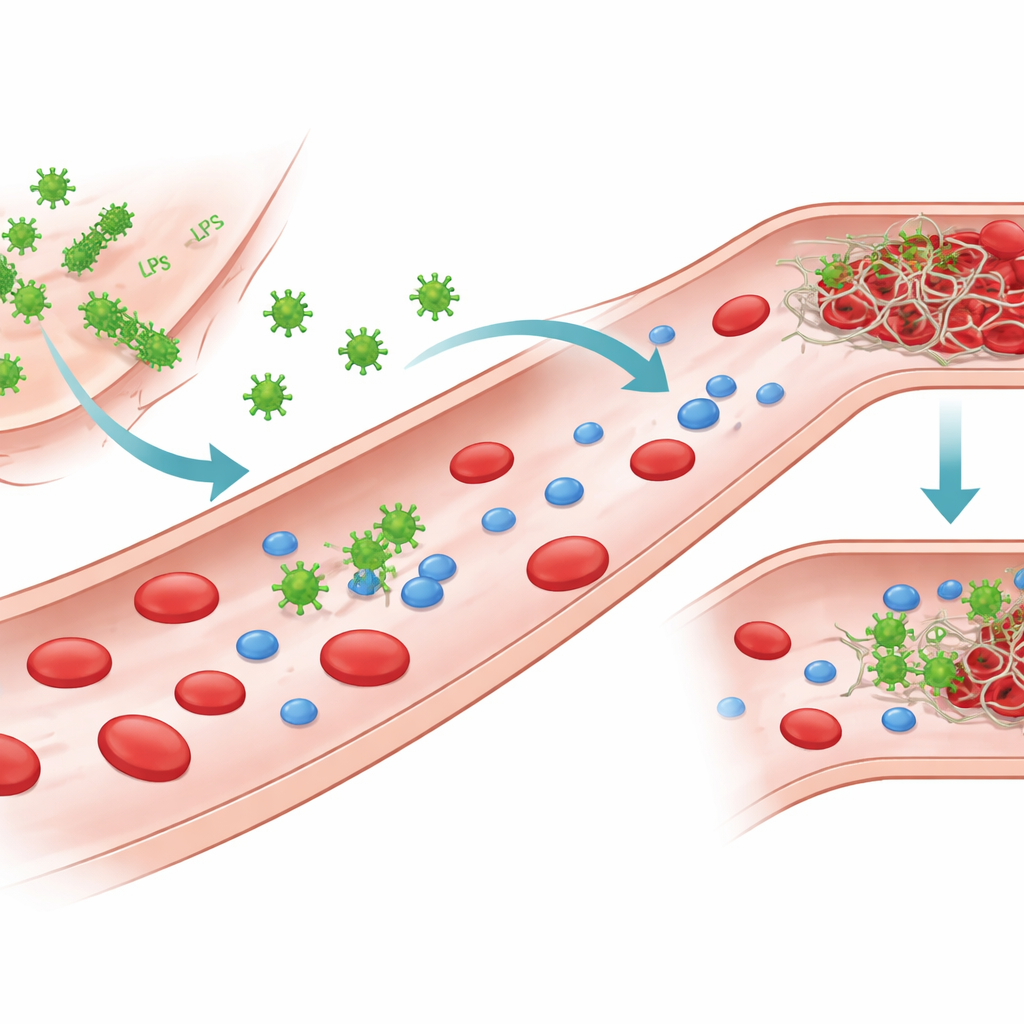
Gram-negative bacteria carry a potent molecule called lipopolysaccharide, or LPS, on their outer surface. When bacteria are damaged or destroyed, LPS can break free and circulate in the blood. The authors focused on LPS from several common pathogens, including two strains of Escherichia coli, as well as Klebsiella pneumoniae and Pseudomonas aeruginosa. Although all are built on the same basic scaffold, their sugar chains and charge patterns differ, creating distinct "chemotypes." These structural differences change how LPS packs together in water, forming single molecules, small clusters, or larger sheet-like assemblies, and they also alter how LPS interacts with proteins in blood.
How LPS Grabs Hold of the Clotting Enzyme
Thrombin carries two strongly positively charged patches on its surface, called exosites, which normally help it recognize and position natural partners like fibrinogen and regulatory cofactors. Because LPS is highly negatively charged, the team suspected that it might latch onto these regions. Using a combination of binding assays and sensitive fluorescence measurements, they showed that LPS from all four bacteria can indeed bind thrombin. However, only some chemotypes—most notably one strain of E. coli (O26:B6) and Klebsiella pneumoniae—induced clear structural shifts in thrombin. By blocking each exosite with short DNA-based probes, the researchers demonstrated that LPS must engage these charged patches to exert its effects, confirming that binding occurs at the same surfaces thrombin uses to do its normal job.
When Bacterial Fragments Slow the Clotting Machine
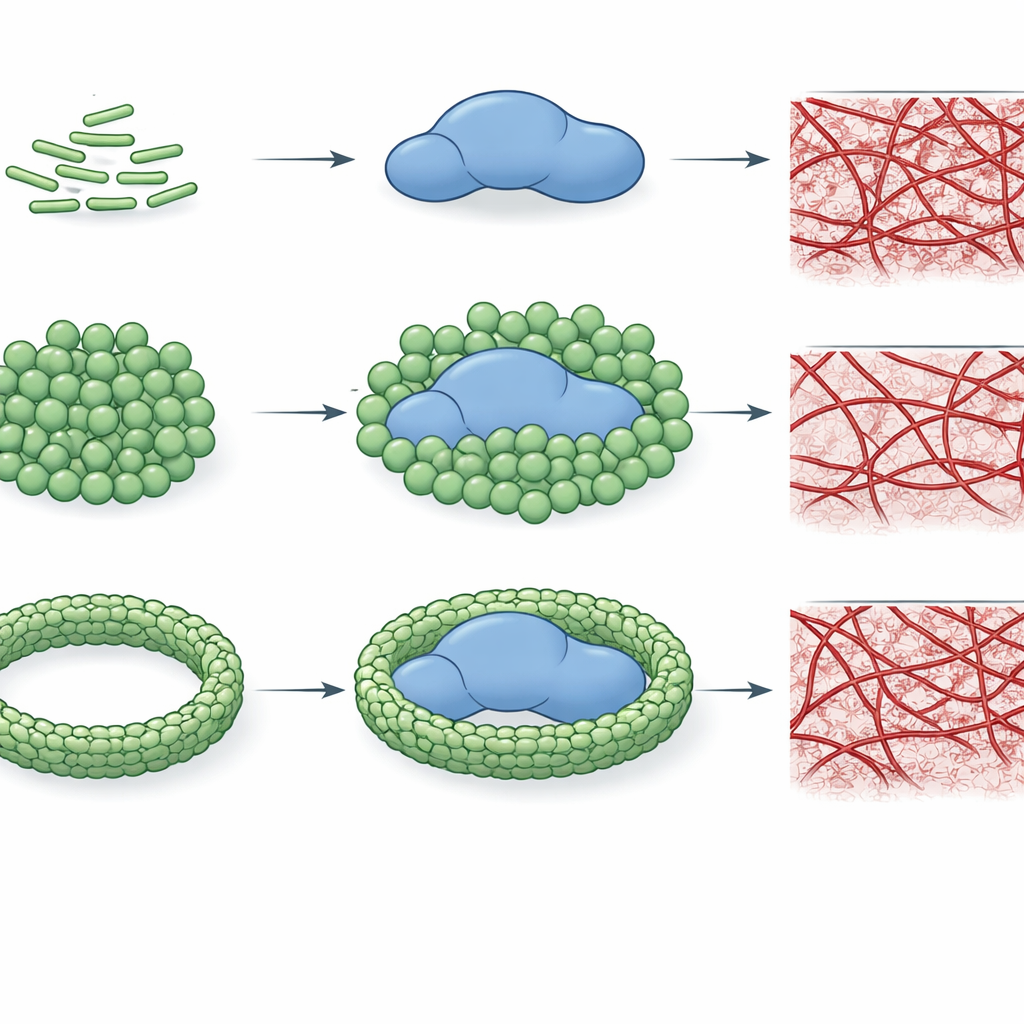
To test whether this binding actually changes thrombin’s performance, the authors tracked how quickly the enzyme cuts a small model substrate and how efficiently it converts fibrinogen into a fibrin network. They found that specific LPS chemotypes, especially E. coli O26:B6 and Klebsiella pneumoniae, can markedly reduce thrombin’s catalytic power, sometimes by more than a hundredfold. This suppression depends not only on the type of LPS but also on how the LPS molecules are organized. When LPS exists as smaller, more curved clusters, it interacts more intimately with thrombin and strongly inhibits it; when calcium ions drive LPS into larger, more ordered aggregates, inhibition is weakened or even lost for some chemotypes. Importantly, these same patterns carry over to clot formation: certain LPS forms can completely prevent fibrin network assembly in purified systems, while others merely slow or even speed up clot growth.
Different Bacteria, Different Clotting Outcomes
The story becomes even richer in plasma, where many other proteins compete for attention. In this more realistic environment, LPS chemotypes still show distinct “fingerprints.” At low concentrations, several types delay the onset of clotting, but at higher levels many switch to a pro-clotting behavior, shortening clotting times. One E. coli chemotype, O26:B6, stands out: when its LPS is assembled into calcium-stabilized aggregates, it continues to slow clotting even at high concentrations, hinting that this particular bacterial strain may promote a persistent tendency toward impaired clot formation. Overall, the work reveals that both the detailed chemistry of LPS and its larger-scale packing state determine whether thrombin is inhibited, left untouched, or nudged toward faster clot formation.
What This Means for Sepsis and Blood Health
The authors conclude that bacterial LPS is not just a distant trigger of inflammation but can directly tune thrombin’s activity through physical contact, with effects that vary from one bacterial strain to another and depend on how the molecules cluster in the bloodstream. In practical terms, this means that the same overall burden of infection may lead to very different clotting patterns depending on the mix and state of LPS present. By mapping this new layer of interaction, the study helps explain why sepsis can cause both excessive clotting and impaired clot formation, and it opens the door to therapies that target LPS–thrombin binding or LPS organization itself to restore balance to the body’s clotting system.
Citation: Lira, A.L., Drew, K.C., Dantas, R.L.M. et al. Molecular basis for inhibition of α-thrombin activity by bacterial lipopolysaccharides. Sci Rep 16, 10851 (2026). https://doi.org/10.1038/s41598-026-46276-5
Keywords: sepsis, thrombin, lipopolysaccharide, blood clotting, Gram-negative bacteria