Clear Sky Science · en
CRISPR/Cas9-mediated targeted knock-in of human erythropoietin at the β-casein locus results in lactogenic hormone-responsive expression in HC11 mammary epithelial cells
Turning Milk Cells into Tiny Medicine Factories
Many modern medicines are proteins that our own bodies naturally make, but producing them at scale in factories is expensive and technically demanding. This study explores a clever alternative: rewiring milk-producing cells so they can switch on a human therapeutic protein only when triggered by hormones, much like they would naturally switch on milk proteins during lactation. The work offers a glimpse of how future farm animals might safely and efficiently make complex drugs in their milk.
Aiming for Safe, Precise Drug Production
For years, scientists have tried to use transgenic animals to produce medicines such as human erythropoietin (hEPO), a hormone that helps the body make red blood cells. Earlier attempts relied on randomly inserting the hEPO gene into the genome, which often led to several problems: the gene could switch on in the wrong tissues, be silenced, or cause illness in the animal. The authors of this paper set out to solve these issues by placing the hEPO gene into a very specific, well-understood spot in the genome: the region that normally controls a major milk protein called beta-casein in mammary (milk-producing) cells.
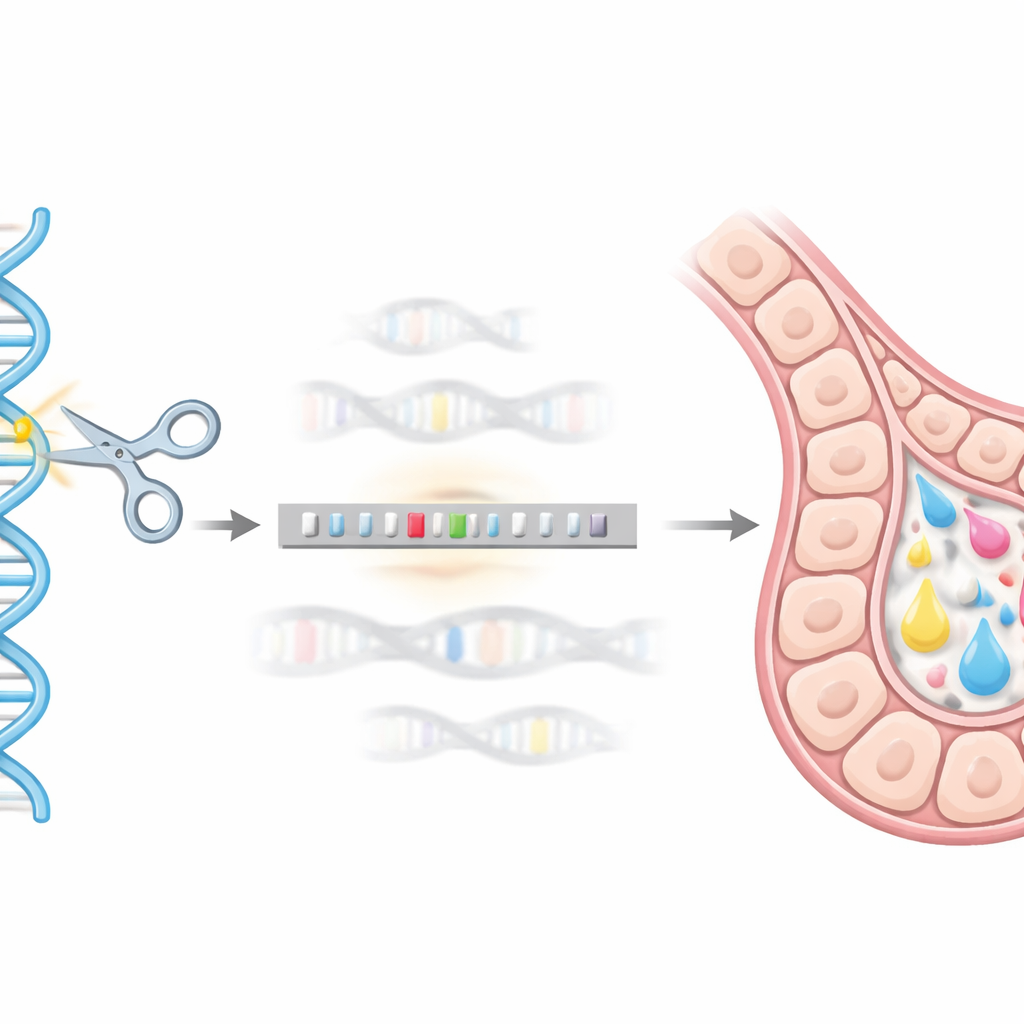
Using Gene Editing as a Genomic "GPS"
To reach this precise genomic address, the team used the CRISPR/Cas9 gene-editing system as a kind of molecular GPS and scissors. They designed a DNA donor construct carrying the hEPO gene fused to a helper tag (GST) and flanked it with short stretches of DNA matching the beta-casein region. When CRISPR/Cas9 cut the target site in mouse mammary HC11 cells, the cell’s natural repair machinery could use this donor construct as a template, neatly inserting the hEPO sequence into the beta-casein locus. Through a series of genetic tests (using PCR-based methods), they isolated single-cell clones in which one copy of the beta-casein gene had been successfully replaced by the hEPO construct, creating stable “knock-in” lines.
Hormone Switch Mimics Natural Milk Production
The researchers then asked whether this new hEPO gene would follow the same on–off rules as the normal milk-protein gene. HC11 cells can be pushed into a milk-like state by adding “lactogenic” hormones, similar to those that act in a nursing animal. In the engineered clones, the team measured both RNA and protein levels before and after hormone treatment. They found that hEPO RNA levels rose modestly—about two to three times—when hormones were added, but protein levels in the culture medium climbed much more dramatically, by roughly 10- to 20-fold. This pattern shows that the inserted hEPO gene is indeed under the control of the mammary-specific regulatory elements and is secreted into the surrounding fluid in a hormone-dependent way, mimicking natural milk-protein production.
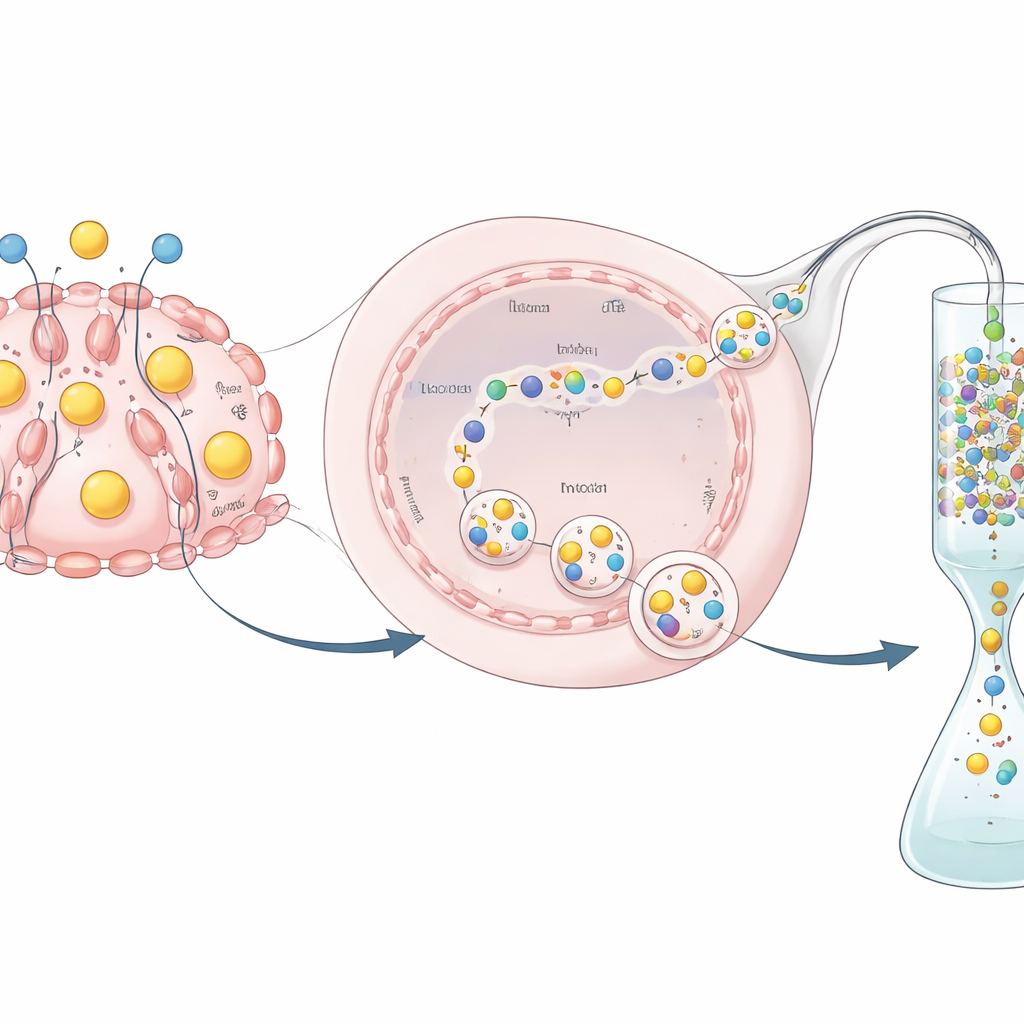
From Secreted Protein to Usable Medicine
Producing a protein is only half the challenge; it must also be purified and tested. Because the hEPO was fused to a GST tag, the researchers could capture it on a special column that binds GST, wash away impurities, and then use a specific enzyme to cut off the tag and release a cleaner hEPO product. They showed, using biochemical assays, that the purified protein retained measurable biological activity—indicating that the hormone made by these edited cells is not just present but functional. Although the overall yield and specific activity were lower than those achieved in industrial cell lines such as CHO cells, this experiment was designed as a proof of concept rather than a fully optimized production process.
What This Could Mean for Future Medicines
In simple terms, this study demonstrates that it is possible to park a human therapeutic gene directly under the control of a natural milk-protein switch in mammary cells, and to turn that gene on with the same hormones that drive lactation. The resulting hEPO can be secreted, partially purified, and shown to work, all from a single, well-defined genomic location. While further improvements are needed—such as generating animals with both gene copies modified, improving purification, and carefully analyzing the protein’s fine structure—this targeted strategy brings the idea of using the mammary gland as a safe, controllable bioreactor for complex medicines closer to reality.
Citation: You, HJ., Kim, GY. & Kang, MJ. CRISPR/Cas9-mediated targeted knock-in of human erythropoietin at the β-casein locus results in lactogenic hormone-responsive expression in HC11 mammary epithelial cells. Sci Rep 16, 14606 (2026). https://doi.org/10.1038/s41598-026-45234-5
Keywords: erythropoietin, CRISPR knock-in, mammary gland bioreactor, therapeutic protein production, beta-casein locus