Clear Sky Science · en
Dysferlin stabilizes membrane nanodomains of cardiomyocytes after myocardial infarction
Why Heart Cells Need Emergency Patching
When someone survives a heart attack, the real battle has only begun. The damaged region of the heart does not heal like a simple cut; instead, surviving heart muscle cells at the edge of the scar must keep contracting under intense mechanical stress. This study reveals how a little-known repair protein, dysferlin, acts as a kind of microscopic patch kit for these overstressed cells, helping their delicate membranes hold together and preserving the heart’s pumping power.
The Stressful Border Between Life and Scar
After a heart attack, blood flow is blocked to part of the heart, killing many cells outright and leaving a central scar. Surrounding this dead core is a narrow “border zone” of still-living heart muscle cells that endure low oxygen, inflammation, and powerful stretching with every beat. These conditions strain the cells’ outer membrane and its intricate folds, which house crucial structures for electrical signals and calcium handling. The authors suspected that if the membrane’s tiny architectural features could be protected in this zone, more heart function might be saved.
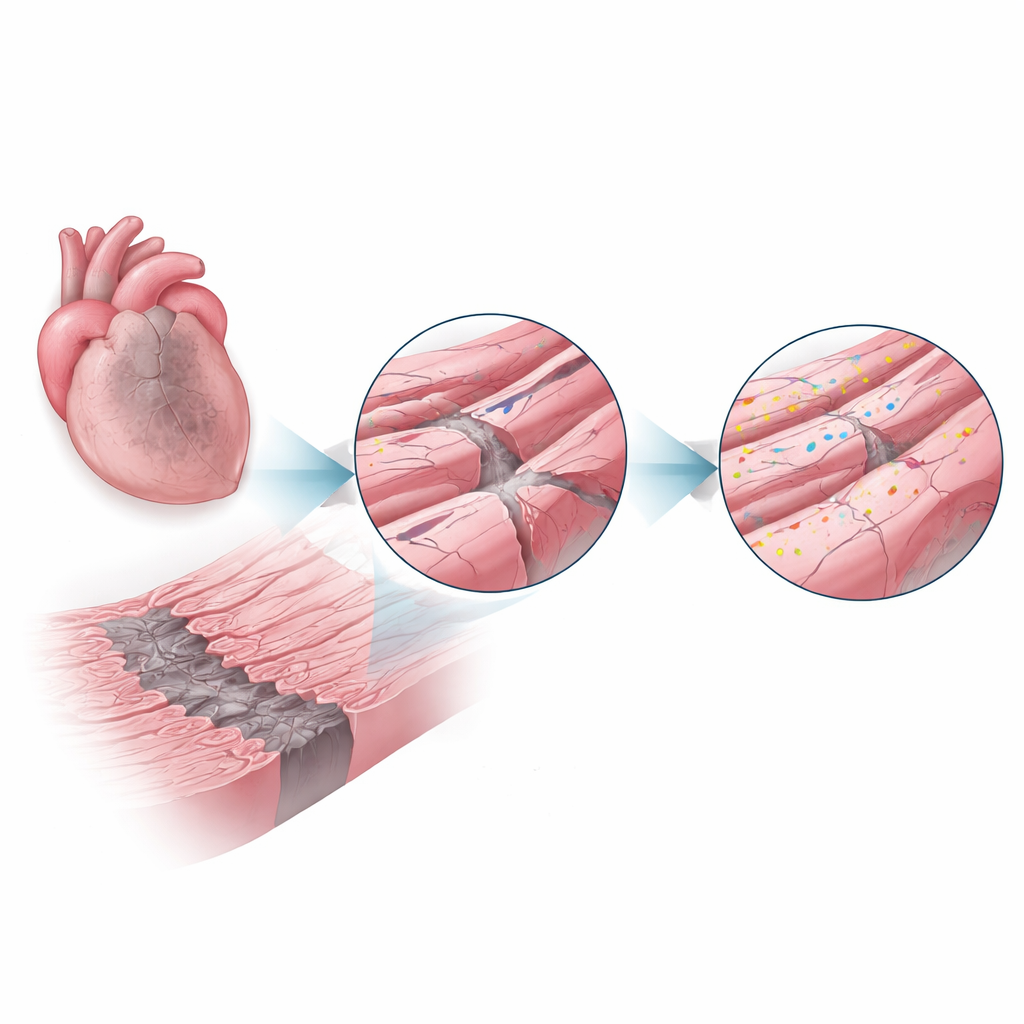
A Repair Protein Steps Into the Spotlight
Dysferlin is a large protein known from rare muscle diseases where its absence leads to weak skeletal muscles, partly because damaged fibers cannot repair their membranes efficiently. Here, researchers asked whether dysferlin plays a similar protective role in the heart. Using mice that either had normal dysferlin or completely lacked it, they induced heart attacks and examined the hearts one and four weeks later. In normal mice, dysferlin levels in the border zone surged to more than double those in healthy hearts, suggesting the cells were actively calling in this repair system where stress was highest.
What Happens When Dysferlin Is Missing
The team then compared outcomes in mice with and without dysferlin. Animals lacking the protein developed significantly larger scars and more dilated, weaker left ventricles. Their hearts pumped less blood with each beat, indicating that more of the surviving myocardium had failed. A powerful protein-survey technique allowed the researchers to catalog thousands of proteins in different heart regions. They found that dysferlin deficiency reshaped these protein profiles, especially in the infarct and border zones, altering pathways linked to cell death, inflammation, and calcium signaling—changes consistent with more fragile cells and progressive failure.
Microscopic Architecture Under Repair
High-resolution imaging zoomed in on the heart cells’ fine structure. In healthy tissue, the cell membrane dips inward in a regular grid of tunnels called the transverse–axial tubule network, which helps electrical signals trigger calcium release and contraction. After a heart attack, this network in the border zone became disorganized and partly lost, particularly its transverse elements. Yet the remaining tubules were coated with bright clusters of dysferlin and surrounded by small internal bubbles of membrane, hinting at active repair and remodeling. At the cell–cell junctions, where heart cells exchange electrical impulses and cling to each other, dysferlin also accumulated alongside key junction proteins. Biochemical experiments showed that dysferlin physically associates with these partners in large complexes, supporting the idea that it helps maintain and rebuild these contact sites under stress.
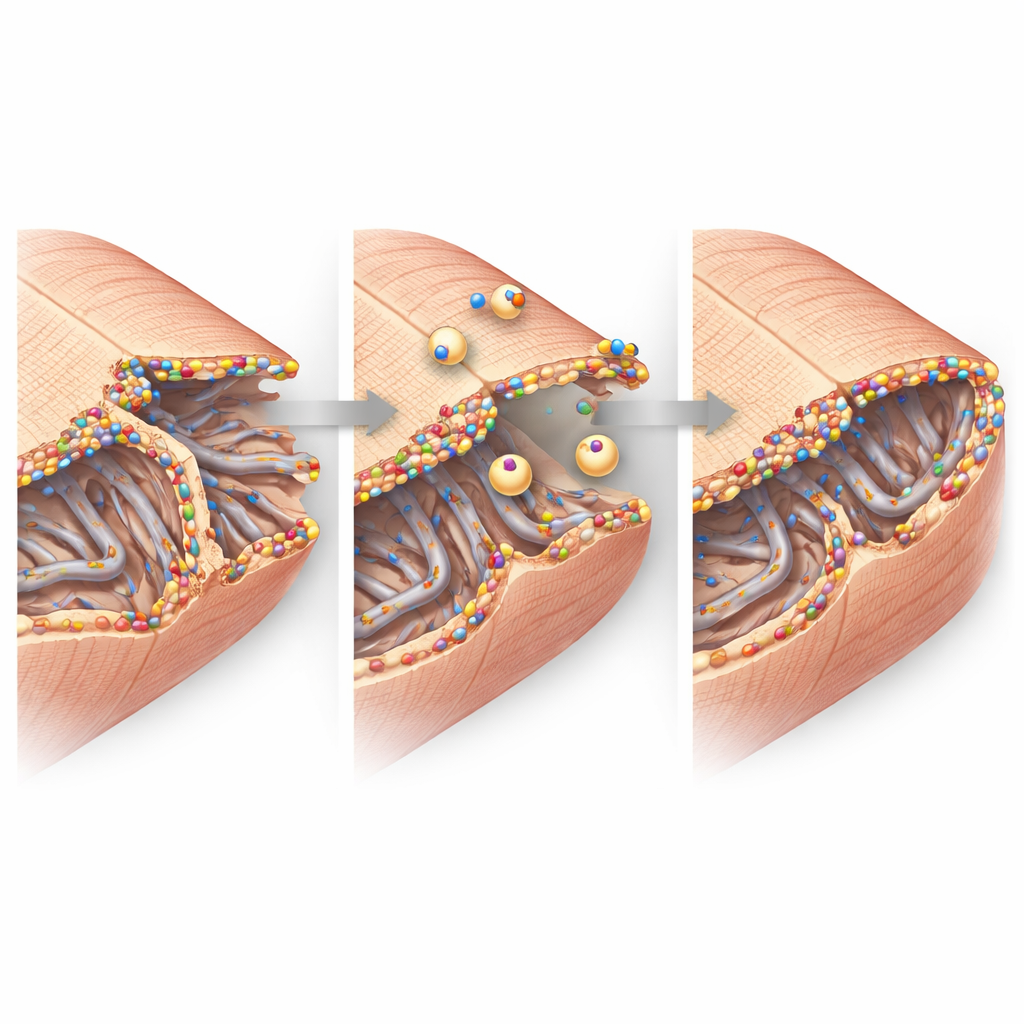
From Molecules to Heart Function
Putting these findings together, the study paints a picture of dysferlin as a guardian of the heart cell membrane in the vulnerable border zone after a heart attack. When present and upregulated, dysferlin clusters at damaged tubular networks and cell–cell junction folds, drawing in repair vesicles and stabilizing these tiny but critical structures. When absent, those nanoscopic supports crumble more readily, more cells are lost or malfunction, and the heart’s pumping power declines. For patients, this work suggests that boosting dysferlin activity or mimicking its repair function could one day become a strategy to limit heart failure after a heart attack by protecting the very membranes that allow heart cells to beat in unison.
Citation: Wegener, J.B., Zühlke, Y., Fleischhacker, C. et al. Dysferlin stabilizes membrane nanodomains of cardiomyocytes after myocardial infarction. Sci Rep 16, 10488 (2026). https://doi.org/10.1038/s41598-026-42800-9
Keywords: myocardial infarction, cardiomyocyte membrane repair, dysferlin, heart failure prevention, nanodomain remodeling