Clear Sky Science · en
Quinoline carboxylic acid derivatives as potent ectonucleotidase inhibitors
Why calming cancer’s “danger signal” matters
Cancer does not only grow; it also learns to hide. One of its favorite tricks is to turn a natural danger signal released by stressed cells into a soothing chemical that lulls the immune system to sleep. This paper explores a new family of small molecules that aim to disrupt that escape route, with the long-term goal of helping the body’s own defenses better recognize and attack tumors.
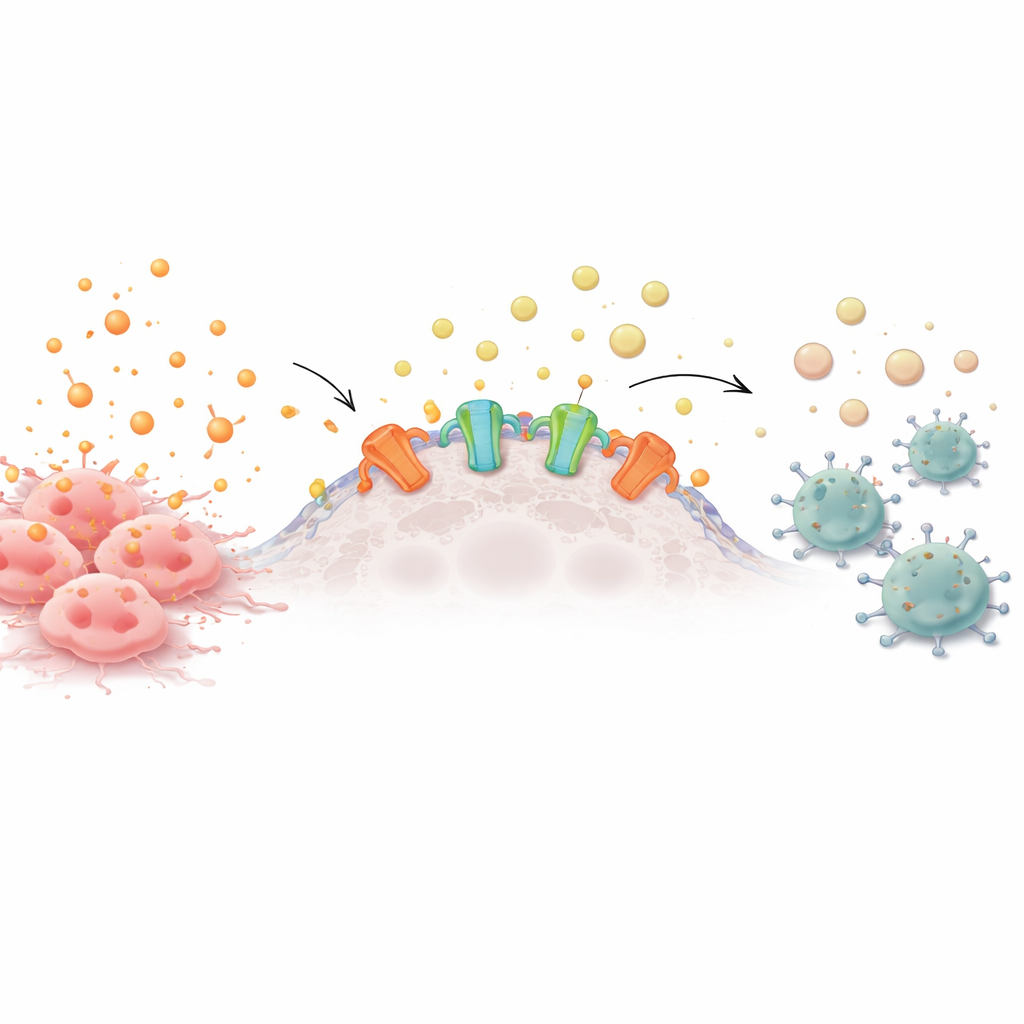
From alarm bells to chemical silence
Healthy and cancerous cells constantly release energy-carrying molecules into their surroundings. When these molecules, such as ATP, build up outside cells, they act like alarm bells, drawing in immune cells to investigate trouble. On the surface of many tumor cells, however, sit enzymes called ectonucleotidases that steadily chop ATP down into adenosine, a calming signal that tells immune cells to stand down. Many aggressive cancers overproduce these enzymes, creating a protective cloud of adenosine that weakens nearby T cells, natural killer cells, and other defenders.
Designing new chemical blockers
The researchers set out to design and synthesize a set of twelve related compounds built on a quinoline “core,” a flat, ring-shaped structure already known to interact with biological targets and to have anticancer potential. By attaching different chemical groups to this core, they created a small library of quinoline-6-carboxamide derivatives. These molecules were carefully characterized using standard chemistry tools to confirm their structures and purity, then tested against several key ectonucleotidases, including four members that process ATP and related substances, as well as two enzymes that complete the final step of making adenosine.
Finding the most effective enzyme stoppers
When the new molecules were mixed with purified human enzymes in the lab, several stood out as strong blockers. One compound, labeled 4d, potently shut down two ATP-processing enzymes (h-NTPDase1 and h-NTPDase2) at very low concentrations, marking it as a promising broad-acting inhibitor. Other derivatives showed more selective behavior: compound 4a strongly blocked ecto-5′-nucleotidase, which performs the last cut that produces adenosine; compound 4k was the best at blocking another enzyme, ENPP1; and compounds 4b and 4g were particularly effective against different NTPDase subtypes. By comparing which chemical decorations improved or weakened activity, the team mapped how small changes in structure can dramatically tune enzyme blocking strength and selectivity.
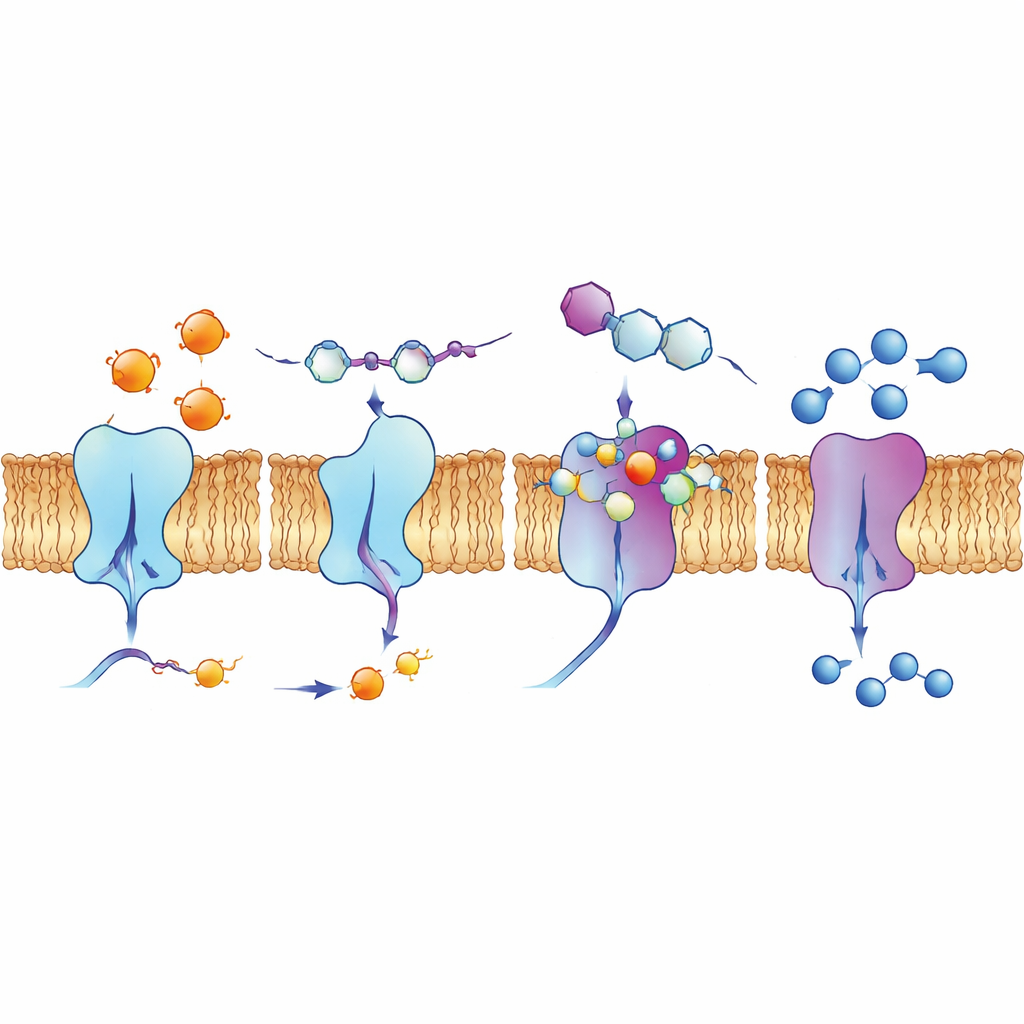
Peering into binding pockets and living cells
To understand why certain compounds worked so well, the authors used computer-based docking simulations and quantum chemistry calculations. These models showed how the quinoline-based molecules nestle into grooves on the enzyme surfaces, forming networks of attractive forces, including hydrogen bonds, stacking between flat rings, and interactions involving halogens such as fluorine, chlorine, or iodine. Calculations of electron distribution supported the idea that the most active compounds have the right balance of stability and reactivity to engage charged sites in the enzymes. The team also examined how one lead compound, 4d, behaves in human lung cancer cells using fluorescence methods. It naturally glows under specific light, and imaging revealed that it concentrates along the cell membrane, the very location of ectonucleotidase enzymes, suggesting that it can reach its intended targets in a realistic biological setting.
Early steps toward anticancer action
Because a useful drug must not only hit its target but also affect cancer cell growth, the researchers assessed how the new molecules influence breast cancer cells grown in culture. Several derivatives, especially 4j and 4k, reduced cell survival and slowed cell division over time, though not as strongly as a standard chemotherapy drug. Computational checks of absorption, distribution, and basic drug-likeness suggested that most of the compounds have properties compatible with oral use and can potentially reach the brain, raising the possibility of future applications in hard-to-treat tumors.
What this means for future cancer therapies
Overall, the study introduces a new family of quinoline-based molecules that efficiently block enzymes responsible for turning immune-stimulating signals into immune-dampening adenosine. While these compounds are at an early stage and show only moderate direct cancer-killing effects, they are promising tools for reshaping the tumor environment so that the body’s own immune cells can work more effectively. With further refinement to boost potency and selectivity, such ectonucleotidase inhibitors could one day complement existing immunotherapies, helping to lift the chemical “fog” that shields tumors from attack.
Citation: Ishaq, A., Nawaz, I., Qadir, J. et al. Quinoline carboxylic acid derivatives as potent ectonucleotidase inhibitors. Sci Rep 16, 10127 (2026). https://doi.org/10.1038/s41598-026-36994-1
Keywords: cancer immunotherapy, adenosine signaling, enzyme inhibitors, quinoline compounds, tumor microenvironment