Clear Sky Science · en
Engineering NK and T cells with metabolite-sensing receptors to target solid tumors
Turning Tumor Chemistry into a Homing Signal
Many of today’s most promising cancer treatments rely on unleashing the immune system, yet solid tumors often sit like walled-off fortresses: the very cells meant to destroy them never arrive in sufficient numbers. This study explores a new way to turn the tumor’s own chemical by-products into beacons that actively pull killer immune cells inside, potentially making next-generation cell therapies far more effective against hard-to-treat cancers such as breast and ovarian tumors.
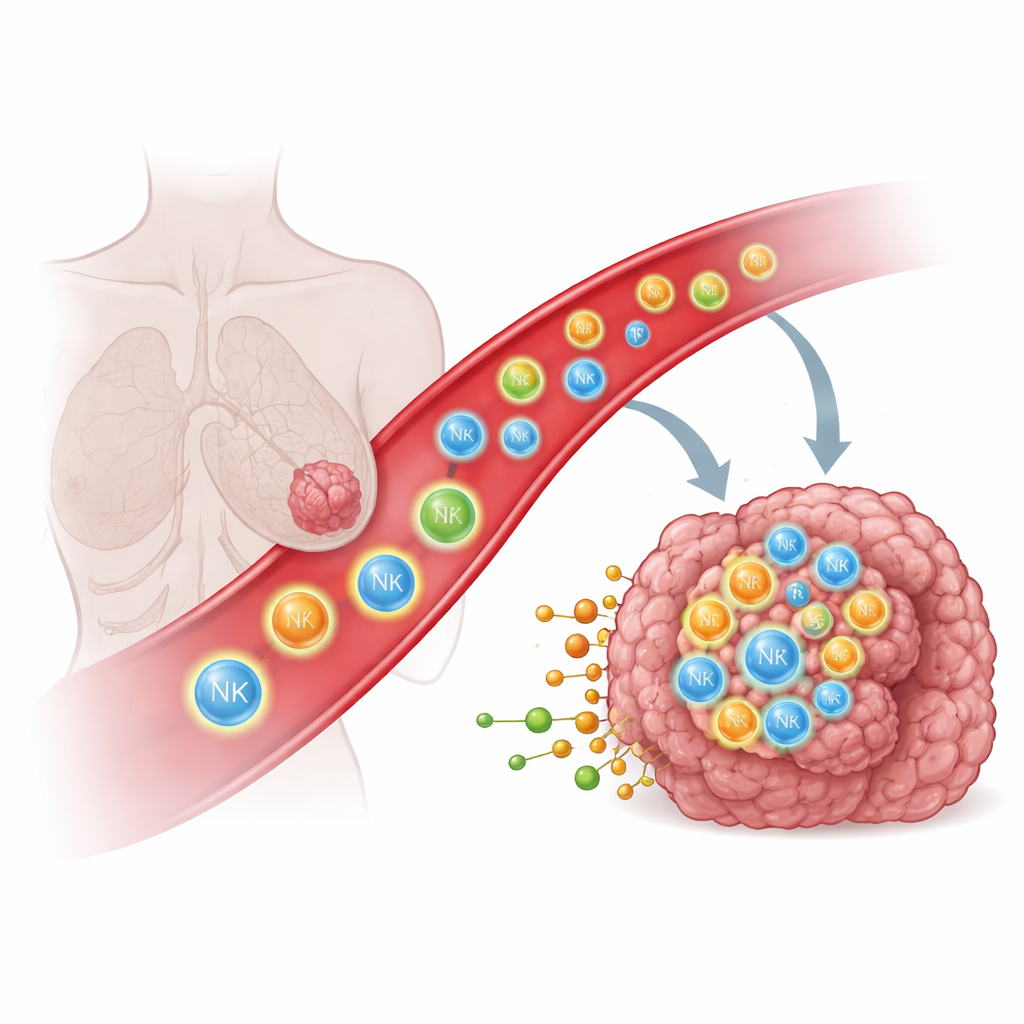
Why Immune Cells Struggle to Reach Solid Tumors
Immune cells like natural killer (NK) cells and T cells can recognize and destroy cancer cells, but only if they physically enter the tumor. In many patients, these cells either never reach the tumor or get stuck in nearby tissues such as the lungs, limiting the impact of powerful therapies, including engineered CAR T and CAR NK cells. Traditionally, scientists have tried to guide immune cells using protein-based cues, such as chemokines and adhesion molecules, that act like molecular street signs. Yet even with this knowledge, directing immune cells deep into solid tumors has remained surprisingly difficult, suggesting that other kinds of signals might be at play.
Discovering Hidden “Smell Receptors” for Tumor Metabolites
To uncover new guidance systems, the researchers used large-scale genetic screens in NK cells, switching on thousands of genes to see which ones helped these cells migrate into breast and ovarian tumors in mice. They repeatedly found a small group of receptors—members of the G-protein-coupled receptor (GPCR) family—that respond not to proteins but to small, bioactive molecules derived from fats and cholesterol. The standout receptor, called GPR183, along with several cousins, normally appears only in select immune cell types. When artificially added to NK or T cells, these receptors acted like new “smell receptors” that sensed metabolites released by cancer cells and drove the engineered cells toward the tumors.
How Tumor By-Products Become a Chemical Trail
The team then asked what exactly the tumors were producing that attracted GPR183-equipped cells. They focused on oxysterols—oxidized forms of cholesterol—made by enzymes that are active in invasive breast cancer cells, especially when those cells are exposed to inflammatory signals. By knocking out key enzymes in cancer cells, the researchers showed that removing the ability to make specific oxysterols sharply reduced the migration of GPR183-expressing NK cells toward tumor-conditioned media. Additional experiments revealed that these ligands not only created a chemical trail but also reshaped the gene activity of NK cells in a ligand-dependent manner, boosting programs linked to movement and membrane organization while dialing down certain metabolic pathways.
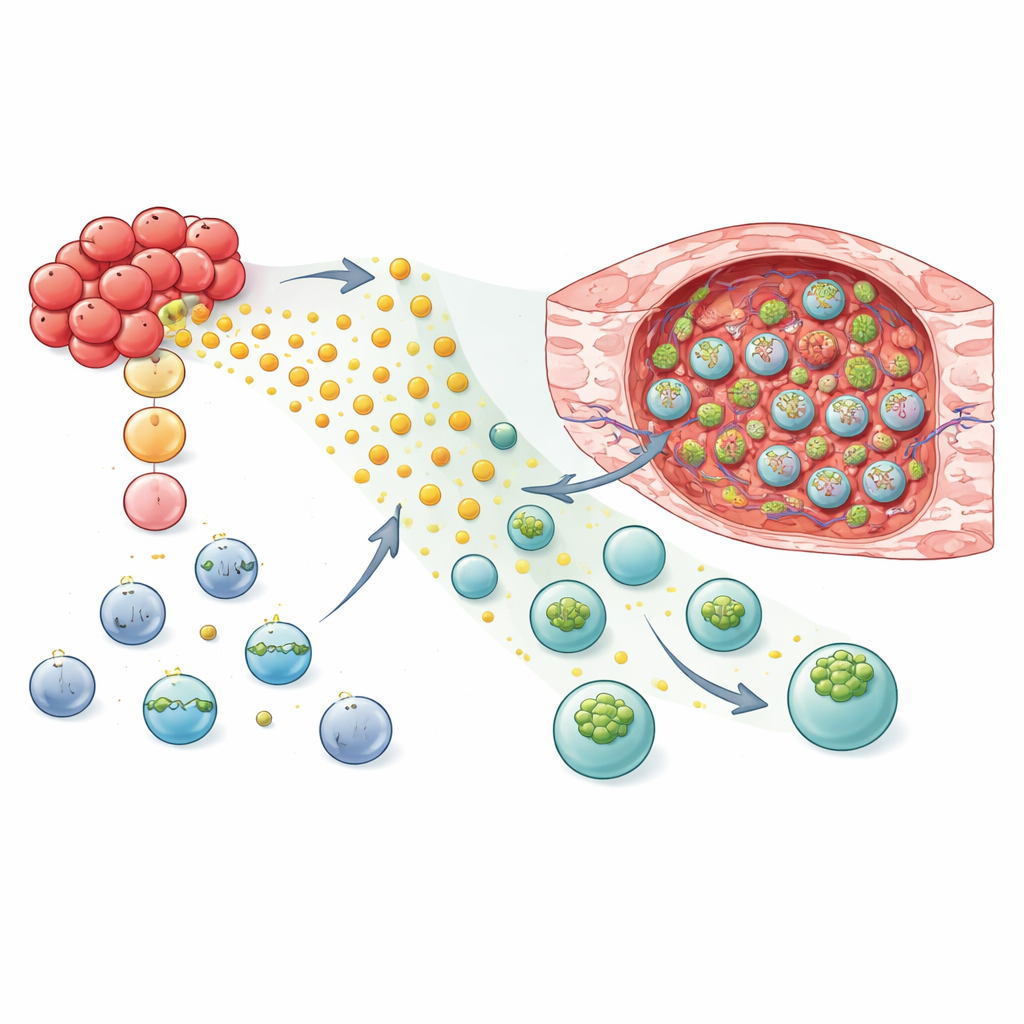
Reprogramming NK and T Cells to Home In and Attack
Armed with this mechanistic insight, the researchers tested whether adding GPR183 could make living therapies better tumor hunters. In mouse models of breast cancer, NK-92 cells and primary human NK cells engineered with GPR183 showed strong, preferential migration to tumor tissue over healthy lung tissue. When these edited NK cells—or CAR NK cells designed to recognize a tumor surface marker—were transferred into mice, tumors grew more slowly than in animals receiving standard cells. The strategy also worked for T cells: both conventional CD8 T cells and tumor-targeting CAR T cells with GPR183 accumulated at higher levels inside tumors while remaining similar in the lung and spleen. In an immunocompetent mouse model, T cells bearing the mouse version of GPR183 controlled breast tumors more effectively and increased the fraction of animals with complete tumor clearance.
What This Could Mean for Future Cancer Treatments
To a non-specialist, the core message is that the authors have found a way to rewire immune cells so they follow the chemical “smell” of a tumor’s own metabolism. By equipping NK and T cells with metabolite-sensing receptors like GPR183, they turned scattered immune forces into targeted seekers that accumulate where they are most needed. The work suggests that, rather than fighting tumor chemistry, we can hijack it—using the molecules cancer cells naturally produce as homing cues for engineered cell therapies. If translated safely into the clinic, this approach could help overcome one of the biggest obstacles in treating solid tumors: getting enough potent immune cells into the right place at the right time.
Citation: Kim, YM., Tsai, M.K., Sun, C. et al. Engineering NK and T cells with metabolite-sensing receptors to target solid tumors. Nat Immunol 27, 1039–1052 (2026). https://doi.org/10.1038/s41590-026-02473-y
Keywords: immunotherapy, cell engineering, solid tumors, chemotaxis, GPCRs