Clear Sky Science · en
In vivo site-specific engineering to reprogram T cells
Turning the Body into Its Own Cell Factory
Doctors can now cure some blood cancers by giving patients custom-made immune cells called CAR T cells, but these powerful treatments are slow and extremely expensive to manufacture. This study explores a different idea: instead of building these cellular medicines in a factory and infusing them back, what if we could reprogram a person’s own T cells directly inside the body? The researchers describe a way to do exactly that in mice with human immune systems, pointing toward cancer therapies that could someday be faster, cheaper and more widely available.
Why Reprogramming T Cells Is So Hard
T cells are white blood cells that can hunt down infected or cancerous cells. In today’s approved CAR T therapies, T cells are removed from the patient, genetically altered in a lab to carry a tumor-targeting receptor, grown to large numbers, and then reinfused. This ex vivo process can take weeks, costs hundreds of thousands of dollars, and produces variable cell products. Earlier efforts to program T cells directly in the body have relied on viral vectors that insert DNA at random positions in the genome, or on messenger RNA that disappears after a short time. Random insertion raises safety concerns, and short-lived expression may not hold cancers in check. The team behind this paper aimed to solve both problems at once by inserting the CAR precisely into a natural T‑cell control switch, so that only T cells would be altered and the new receptor would be expressed in a more regulated, durable way.
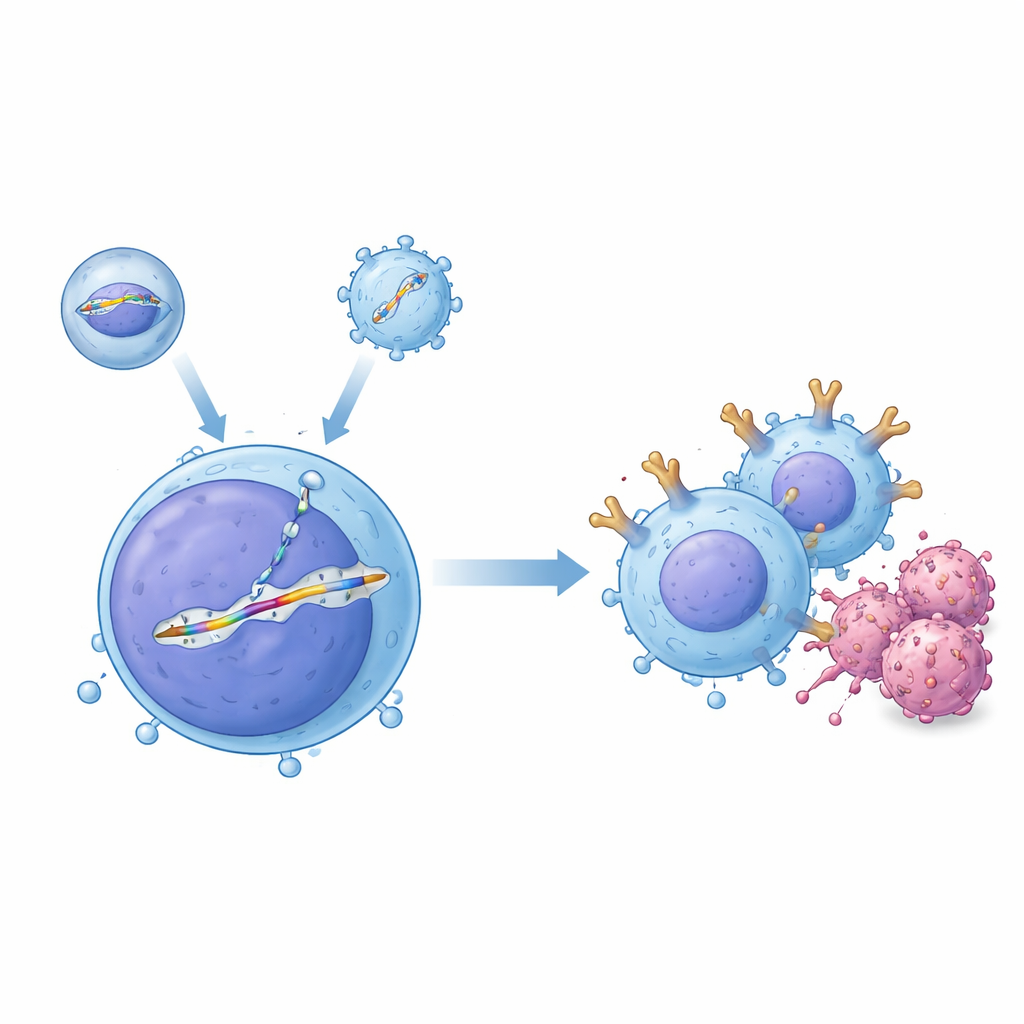
A Two-Tool System for Precision Editing in the Bloodstream
The researchers created a “two-vector” delivery system that works together inside the body. One tool, called an enveloped delivery vehicle, brings in the CRISPR–Cas9 gene-editing protein programmed to cut a specific site in the T cell’s DNA. The second tool, built from an adeno-associated virus, carries a matching DNA template that contains the CAR blueprint flanked by sequences that guide it into the chosen slot, a region of the T cell receptor gene known as TRAC. When both arrive in the same cell, the cut is repaired by copying from the template, neatly inserting a promoterless CAR cassette under the control of TRAC’s natural on/off switch. In test-tube experiments with human T cells, this pairing generated large fractions of precisely edited cells, and the investigators showed that the edited T cells could efficiently kill B‑cell leukemia cells.
Engineering Delivery Vehicles to Hit T Cells—and Miss the Wrong Targets
Delivering these genetic tools safely in a living body is far more challenging than in a dish, because blood contains antibodies and many other cell types that must be avoided. To overcome this, the team evolved an AAV capsid variant, dubbed AAV-hT7, that remains active even in the presence of human serum and prefers human T cells over stem cells and cancer cells. Using CRISPR screens, they found that a surface protein called CD7 strongly influences how well this vector enters T cells, helping explain its specificity. In parallel, they redesigned their enveloped delivery vehicle by mutating its normal viral coat so it could no longer bind its usual receptors, then decorating it with an antibody fragment that recognizes CD3, a hallmark of T cells. This dual-targeting strategy not only steered particles toward T cells but also briefly activated them, nudging more of them into a cell cycle state that favors precise DNA repair and stable insertion of the CAR cassette.
In-Body CAR T Cells that Control Leukemia, Myeloma and Solid Tumors
Armed with their optimized pair—CD3-targeted particles plus the AAV-hT7 DNA carrier—the scientists tested the approach in several humanized mouse models. In mice engrafted with human blood cells, a single intravenous dose of the two vectors converted up to roughly one in five T cells into CAR T cells at the desired TRAC site and wiped out normal B cells, a hallmark of active anti-CD19 CAR function. In aggressive leukemia models, this in vivo–generated CAR T population expanded quickly, cleared tumors, and often protected against a later tumor rechallenge. Compared head-to-head with in vivo lentiviral CAR delivery, the TRAC-targeted method produced more uniform CAR levels on each cell, faster T‑cell expansion, and better overall tumor control. The team then swapped in CAR designs targeting BCMA, a myeloma marker, or B7-H3, a protein found on many solid tumors, and showed strong responses in models of multiple myeloma and sarcoma, including durable control after rechallenge in many animals.
What This Could Mean for Future Cancer Care
For non-specialists, the key message is that the authors have turned the body itself into a programmable factory for designer immune cells. By delivering CRISPR machinery and a carefully crafted DNA template together, and by steering both only into T cells, they achieved precise, stable “rewiring” of human T cells inside living animals. These newly made CAR T cells behaved much like or better than current lab-grown versions in several cancer models. Although much work remains to prove safety, control dosing and translate to humans, the study outlines a realistic path toward off-the-shelf injections that instruct a patient’s own immune system to fight blood cancers—and potentially some solid tumors—without ever having to ship their cells to a factory.
Citation: Nyberg, W.A., Bernard, PL., Ngo, W. et al. In vivo site-specific engineering to reprogram T cells. Nature 652, 712–721 (2026). https://doi.org/10.1038/s41586-026-10235-x
Keywords: CAR T cells, in vivo gene editing, CRISPR, cancer immunotherapy, viral vectors