Clear Sky Science · en
Bacterial immune activation via supramolecular assembly with phage triggers
How Bacteria Outsmart Their Viral Invaders
Bacteria are constantly hunted by viruses called phages that can turn a thriving microbial community into a graveyard within minutes. This study uncovers a surprising new way that some bacteria fight back: by building huge molecular rings that sense incoming phages and then deliberately shut down the infected cell from the inside. The work not only reveals a clever form of bacterial self-defense, but also echoes strategies used by our own innate immune system, hinting at deep common themes in how life detects and stops infections.
A Tiny Guardian with a Double Life
The paper focuses on a bacterial protein the authors rename RAZR, found in Escherichia coli and many other species. On its own, RAZR is harmless: cells can produce it at high levels without ill effects. Yet when certain phages infect, RAZR suddenly becomes deadly—to the host cell as well as to the virus. The system acts through “abortive infection”: once RAZR is switched on, the infected bacterium stops growing and effectively sacrifices itself, preventing the virus from making more copies and spreading through the population. Understanding how RAZR knows when to flip from quiet bystander to cellular executioner is the central question of the study. 
Viral Rings That Flip the Switch
The researchers discovered that RAZR is activated by specific proteins made by several unrelated phages. In one phage, a protein called Gp77—likely involved in repairing or circularizing the viral genome—served as the trigger. In other phages, the triggers were “portal” proteins that sit at a key gateway in the viral shell and help inject DNA into the host. Although these proteins look very different in sequence and shape as individual molecules, they all share one striking feature: they assemble into large rings of nearly identical diameter. Using cryo–electron microscopy, the team showed that Gp77 forms a 24-part ring, and that portal proteins form 12-part rings. RAZR, which normally forms loose linear chains, wraps itself around these viral rings to build a massive, multi-layered circular complex more than 25 nanometres across.
Building a Molecular Doomsday Device
Within this complex, RAZR brings together two functional regions: a zinc-finger “sensor” that grips the viral ring, and a HEPN “blade” that cuts RNA. Two zinc-finger segments from neighboring RAZR molecules bind each viral protein unit, ensuring that RAZR coats the ring in a precise and highly cooperative fashion. This ring-shaped scaffold forces RAZR’s RNA-cutting domains into a curved, tightly packed arrangement, allowing many cutting sites to line the outer edge like teeth on a circular saw. Mutations that disrupt the contact surfaces between RAZR and the phage proteins, or that weaken the way RAZR molecules stick to each other, cripple the defense. A small flexible linker that connects the sensing and cutting parts of RAZR also proves essential, helping the protein adopt the right geometry when it hugs the viral ring.
Shredding RNA to Halt Viral Spread
Once assembled on its phage-made scaffold, RAZR turns into a powerful RNA-destroying machine. The authors show that activated RAZR leaves DNA untouched but slices many types of single-stranded RNA: transfer RNAs, ribosomal RNAs, and messenger RNAs from both the bacterium and the infecting phage. In living cells, this widespread RNA cleavage rapidly blocks protein production while leaving DNA replication and transcription largely intact. In test-tube experiments, mixing purified RAZR with its viral triggers is enough to stop protein synthesis cold. By halting translation in this blunt, non-specific way, the system dooms the infected cell but also starves the phage of the molecular tools it needs to reproduce. 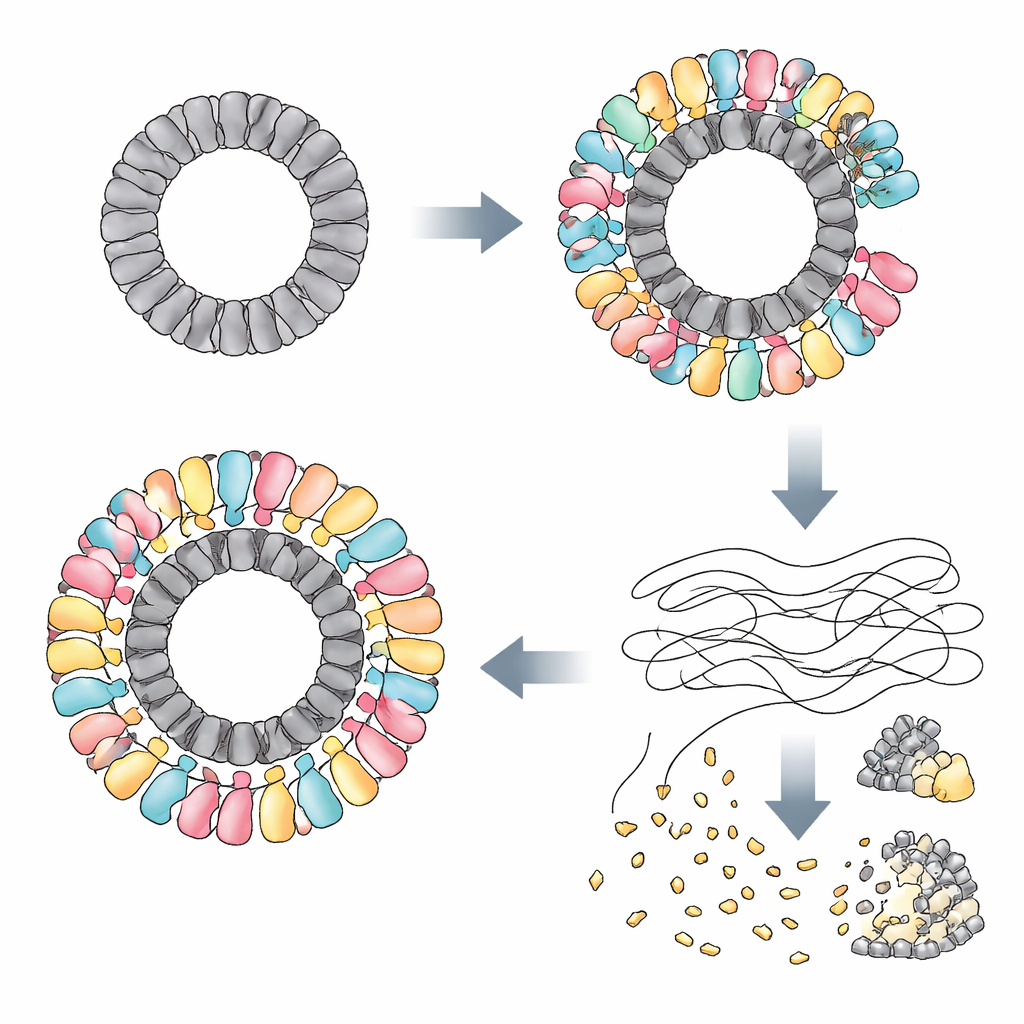
Tailoring Defense to Different Viruses
Although RAZR proteins from different bacteria share a similar cutting domain, the study finds that their zinc-finger sensors vary widely. Swapping these sensor segments between two RAZR versions, from E. coli and Klebsiella variicola, largely swaps which phages they can stop. Even when two RAZR variants target the same phage, they rely on slightly different contact points on the viral portal protein, as shown by distinct viral escape mutations. This suggests that bacteria can fine-tune their antiviral range by evolving the sensor portion of RAZR, while keeping the powerful RNA-cutting machinery intact.
Shared Ideas Across the Tree of Life
In the end, the authors propose that RAZR senses not a specific viral “password” but a structural signature: large protein rings of a particular size that appear only during phage infection and are absent from the host’s own machinery. By wrapping around these rings and using them as templates for its own assembly, RAZR amplifies a faint viral signal into an all-or-none immune response. This strategy closely mirrors how many innate immune complexes in animals, including humans, form giant filamentous or ring-shaped assemblies to detect pathogens and activate destructive enzymes. The work thus reveals a striking convergence: from bacteria to mammals, life has repeatedly evolved ways to build huge protein structures that turn the geometry of invading viruses against them.
Citation: Zhang, T., Lyu, Y., Beck, C.R. et al. Bacterial immune activation via supramolecular assembly with phage triggers. Nature 651, 1051–1059 (2026). https://doi.org/10.1038/s41586-025-10060-8
Keywords: bacteriophage immunity, bacterial innate defense, supramolecular protein complex, RNA cleavage, RAZR system