Clear Sky Science · en
Experimentally calibrated multiscale model predicts schedule dependent drug combination effects
Why timing matters in cancer drug treatment
Cancer therapy is not just about choosing the right drugs; it is also about giving them in the right way and at the right time. Many promising drug pairs look powerful in simple lab dishes but lose their punch in real tumors, where drugs spread unevenly and cells respond in complex, coordinated ways. This study introduces a computer model that connects what happens inside single cancer cells to what happens in a whole mass of cells, and shows how the order and timing of two drugs can make the difference between modest slowing of growth and dramatic tumor collapse. 
Linking single cells to whole tumors
The researchers focused on a human gastric (stomach) cancer cell line called AGS and built a “multiscale” model that mirrors its behavior. At the most detailed level, each virtual cell carries a simplified wiring diagram of key signaling molecules that decide whether the cell grows or dies. These internal switches are represented as being either active or inactive, but combined in a way that still allows graded responses. At a higher level, each cell is an independent agent in a simulated tissue: cells push on each other, stop dividing when they feel crowded, and can die or keep growing depending on their internal signals. Above that, drugs and nutrients diffuse through a virtual environment, mimicking how substances move into and through a tumor-like cluster.
Teaching the model with single-drug data
To make the virtual system realistic, the team first trained it on real measurements of how AGS cells grow over time when they are untreated or exposed to single drugs. They used three targeted inhibitors that are relevant for cancer treatment: one blocking PI3K, one blocking MEK, and one blocking AKT, all proteins in pathways that promote cell survival and division. Using powerful optimization algorithms run on high-performance computers, they tuned parameters that control how fast cells divide, how sensitive they are to crowding, how easily drugs cross cell membranes, and how internal signals are converted into growth or death. With these settings, the model closely reproduced the observed growth curves: one drug mainly slowed growth, another caused cells to shrink back after a brief surge, and a third steadily reduced the population over time.
Predicting synergy without seeing it
Once the model could mimic single-drug behavior, the key test was whether it could predict what happens when drugs are combined, without being shown any combination data during training. The researchers constructed parameter ranges that were consistent with all three single-drug fits, then ran thousands of virtual experiments for two known synergistic pairs: PI3K plus MEK inhibition, and AKT plus MEK inhibition. Remarkably, the simulated combinations reproduced the stronger-than-expected growth suppression seen in real experiments. The model showed that each pair works by blending different strengths: one drug strongly blocks growth signals, the other pushes cells toward self-destruction. Together, they both shut down pro-survival cues and boost pro-death signals, driving a much larger fraction of cells to die than either could alone. 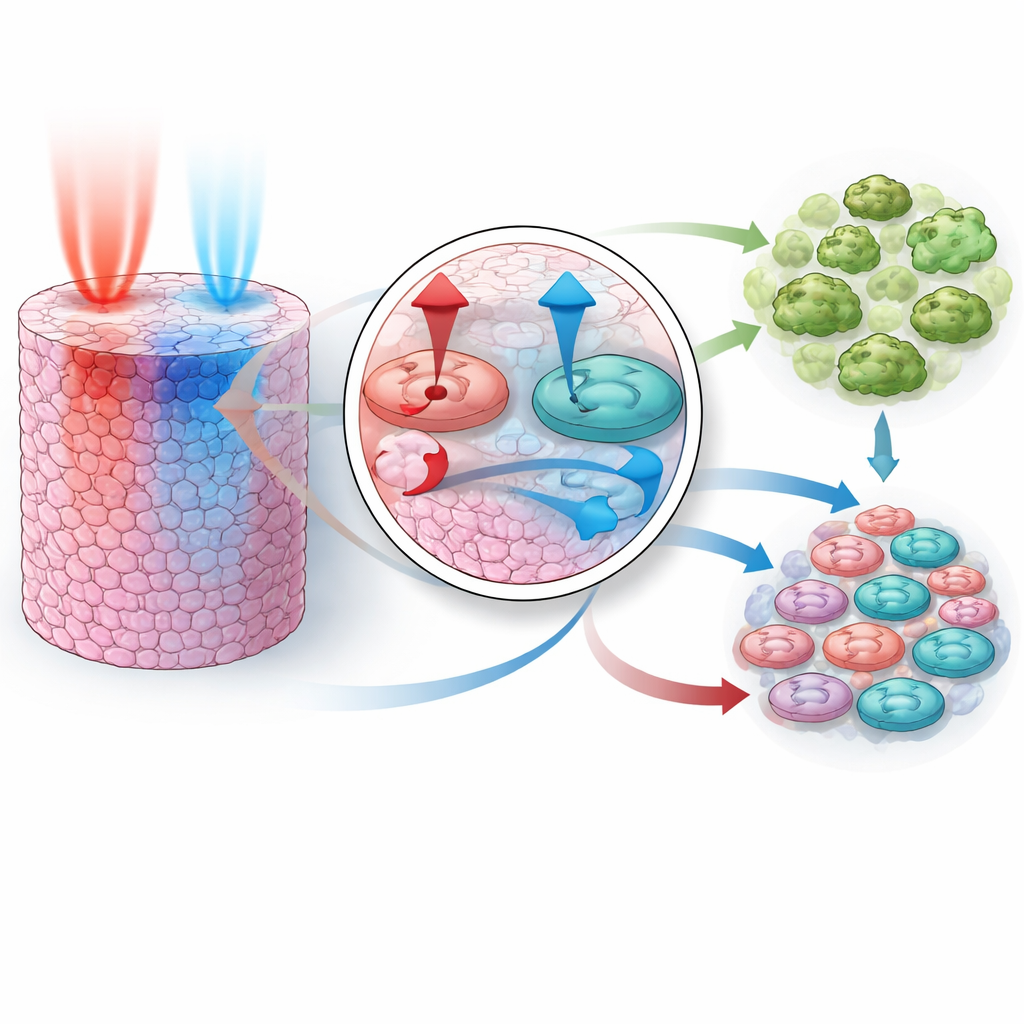
Peering inside the mechanism of synergy
Because the model tracks events from molecular switches to population outcomes, it can offer hypotheses about how the synergy arises. For PI3K and MEK inhibitors, the simulations suggested that cells become globally “primed” for death: their threshold for triggering apoptosis drops, and the internal machinery that executes cell death becomes more active, especially through pathways involving the protein Caspase-9. For the AKT and MEK pair, the model pointed to a different route: survival signals linked to a growth-promoting factor are dampened, while a counteracting factor that encourages cell death gains influence. In this case, the synergy seems to depend more heavily on an alternative death pathway driven by Caspase-8. These distinct mechanisms highlight how the same overall outcome—stronger killing—can be achieved through different internal wiring.
Timing and drug movement shape success
The team then extended the model into three dimensions to mimic a small solid tumor, where drugs enter from one side and must diffuse inward through layers of cells. In this more realistic setting, they explored what happens if the two drugs are given together, or if one precedes the other by various amounts of time, and how changes in how fast each drug moves through tissue affect the outcome. The simulations revealed a clear rule: the pathway controlled by PI3K and AKT must be shut down before or at the same time as the pathway controlled by MEK for the combination to be highly effective. If MEK is blocked first, feedback within the network allows PI3K/AKT activity to rebound, temporarily rescuing cell survival and blunting the overall effect. Differences in how quickly each drug diffuses amplify this effect, meaning that even a “simultaneous” dose at the patient level may arrive in a particular order at the cells themselves.
What this means for future cancer treatment
This work shows that a carefully calibrated computer model, grounded in single-drug experiments, can successfully anticipate how drug pairs will behave over time and suggest why the order of dosing matters. By connecting molecular logic inside cells to the behavior of an entire cell population and to the physical movement of drugs, the framework can generate concrete, testable ideas about which schedules should work best in the lab and, eventually, in patients. While the current results apply specifically to one gastric cancer cell line, the same approach could be adapted to other cancers, helping researchers design smarter combinations and timing strategies rather than relying solely on trial-and-error testing.
Citation: Hayoun-Mya, O., Montagud, A., Valencia, A. et al. Experimentally calibrated multiscale model predicts schedule dependent drug combination effects. npj Syst Biol Appl 12, 52 (2026). https://doi.org/10.1038/s41540-026-00669-4
Keywords: cancer drug combinations, multiscale modeling, drug scheduling, gastric cancer, computational oncology