Clear Sky Science · en
Perivascular spread of CSF-derived α-synuclein oligomers drives region-specific Parkinson’s-like pathology
Why this matters for Parkinson’s disease
People often think of Parkinson’s disease as a movement disorder, marked by tremor and stiffness. But years before these motor problems appear, many patients lose their sense of smell. This study asks a simple but powerful question: could toxic proteins floating in the brain’s clear bathing fluid help start the disease in smell-related brain regions, long before movement centers are hit? By tracing how these proteins travel and what damage they cause in mice, the researchers uncover a hidden plumbing system that may drive the stepwise progression of Parkinson’s—and point to an unexpected new way to slow it down.
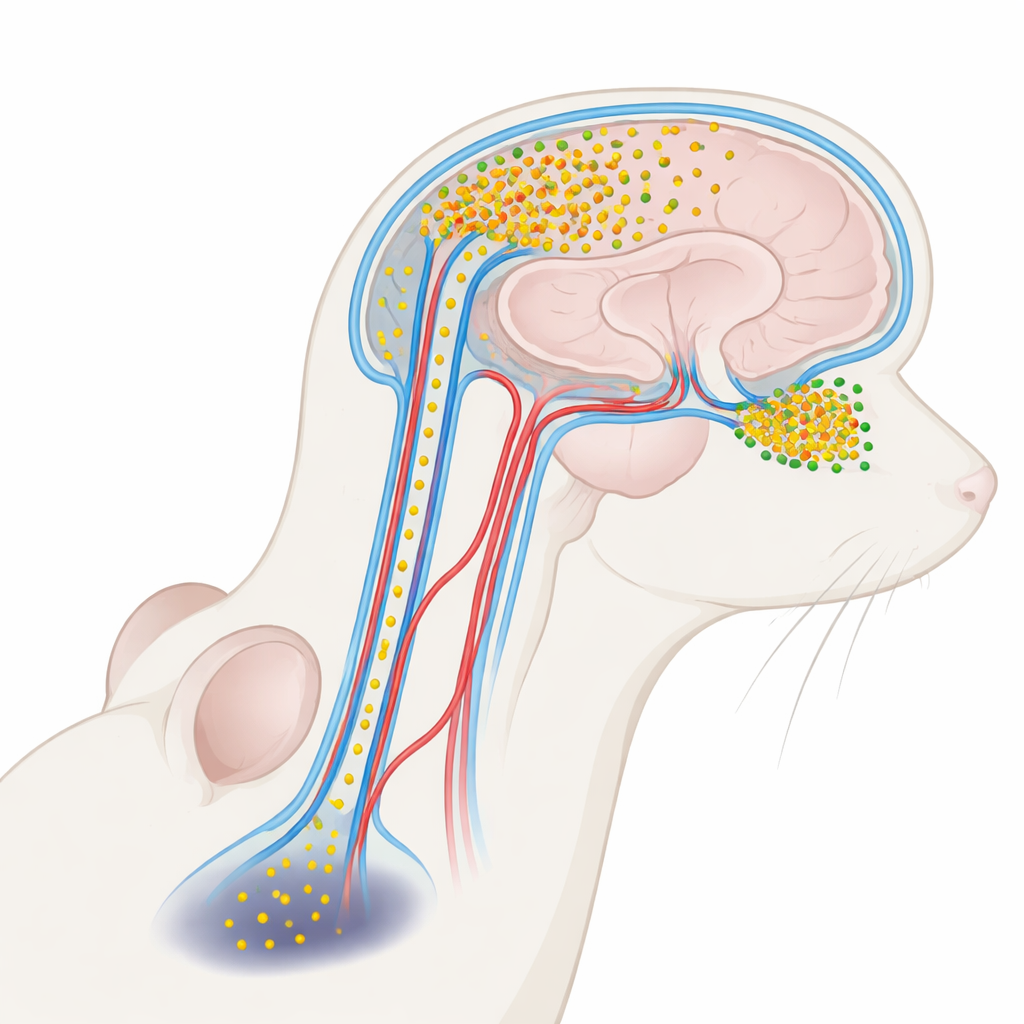
A harmful protein riding in brain fluid
Parkinson’s disease is closely linked to a protein called alpha‑synuclein. In healthy brains, it helps nerve cells communicate. Under disease conditions, however, alpha‑synuclein can clump into small soluble clusters called oligomers that are especially toxic to neurons. These oligomers show up at higher levels in the clear liquid that bathes the brain and spinal cord—the cerebrospinal fluid, or CSF—of people with Parkinson’s. Until now, most work treated CSF alpha‑synuclein as a useful marker of disease, not as an active player. The authors of this paper asked whether these CSF‑borne oligomers might actually seep back into the brain and injure specific regions, helping explain why smell problems come early while movement problems emerge later.
The brain’s plumbing and a vulnerable smell center
CSF does not just cushion the brain; it circulates through a network of channels alongside blood vessels known as perivascular spaces, part of a broader “glymphatic” clearance system. Using glowing tags and advanced three‑dimensional microscopes, the researchers injected labeled alpha‑synuclein into the CSF of mice and watched where it went. They found that the protein entered the brain along these perivascular routes and accumulated very unevenly. The olfactory bulb—the small structure at the front of the brain that processes odors—soaked up far more of the incoming protein than deeper regions such as the substantia nigra, which controls movement. Even within the olfactory bulb itself, some subregions showed heavier influx than others, suggesting a fine‑grained vulnerability shaped by local fluid flow and vessel layout.
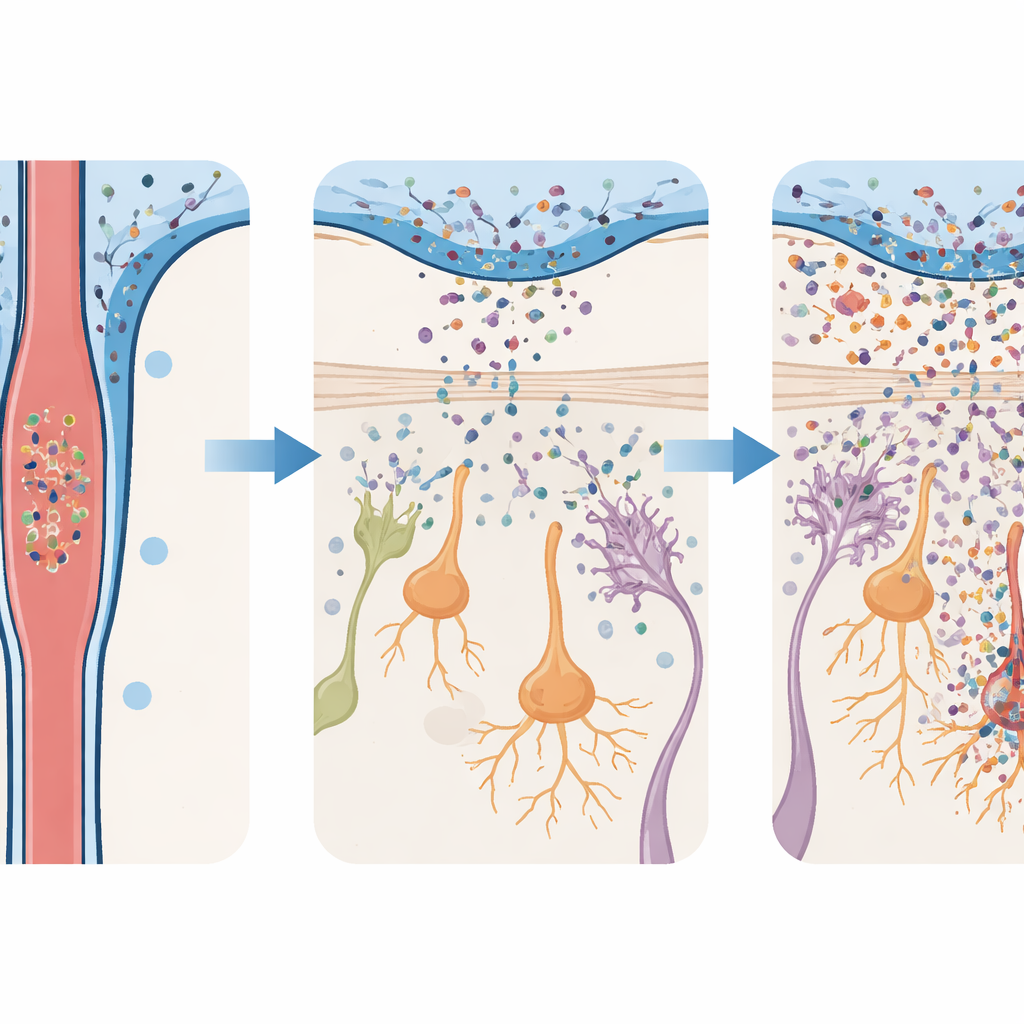
From silent influx to inflammation and neuron loss
To test what this influx does, the team infused mice with toxic alpha‑synuclein oligomers and examined their brains over time. In both the olfactory bulb and the substantia nigra, immune support cells called microglia and astrocytes became activated, and inflammatory molecules rose. But these reactions were much stronger in the olfactory bulb. There, dopamine‑producing neurons—cells that use the same chemical messenger implicated in Parkinson’s—began to die off early and continued to decline for weeks. In contrast, dopamine neurons in the substantia nigra remained largely intact until much later, when losses finally appeared. This staged damage mirrors the clinical picture: early problems in smell circuitry, followed by later injury to movement‑controlling cells.
Early smell problems and late movement issues
The behavioral changes in the mice followed the same timeline as the cellular damage. Animals given alpha‑synuclein oligomers quickly developed trouble finding hidden food by smell, showed less interest in attractive odors such as female mouse urine, and were less inclined to avoid unpleasant smells. These smell‑based deficits appeared within days and persisted for almost two months. Yet the same mice walked, balanced, and coordinated normally on standard movement tests until much later, when clear motor difficulties finally emerged. Importantly, when the researchers lowered the dose of injected oligomers, the olfactory bulb showed far less inflammation and neuron loss, and the animals largely retained normal smell abilities. This dose‑sensitivity suggests that the burden of toxic protein in CSF directly shapes how much early damage occurs.
What this means for future treatments
Taken together, the findings support a new view of Parkinson’s disease in which toxic alpha‑synuclein oligomers in CSF do more than signal ongoing damage—they actively spread it by flowing back into the brain along perivascular channels. Because the olfactory bulb receives a stronger share of this influx, it suffers earlier and more severe injury, explaining why loss of smell can precede tremor and stiffness by years. Crucially, the work also hints at an intervention: if we can safely reduce or remove these oligomers from CSF—through filtration devices, targeted antibodies, or other “CSF‑clearing” strategies—we might protect vulnerable regions like the olfactory bulb and slow the entire cascade of Parkinson’s‑like pathology.
Citation: Zhu, WX., He, XZ., Meng, JC. et al. Perivascular spread of CSF-derived α-synuclein oligomers drives region-specific Parkinson’s-like pathology. npj Parkinsons Dis. 12, 84 (2026). https://doi.org/10.1038/s41531-026-01300-3
Keywords: Parkinson’s disease, alpha-synuclein, cerebrospinal fluid, glymphatic system, olfactory dysfunction