Clear Sky Science · en
Chronic stress drives oligoasthenozoospermia via gut microbiota-vitamin-Sting1/Slc9c2 axis
When Stress Reaches the Reproductive System
Most people know that chronic stress can sap energy, disturb sleep, and cloud mood, but few realize it may also quietly undermine male fertility. This study in rats uncovers how long‑lasting psychological stress can reduce sperm count and movement, not just through hormones, but by disturbing gut microbes and the vitamins they help supply. The work points to a hidden communication line between the brain, the gut, and the testes—and suggests that supporting beneficial bacteria and key nutrients might one day help protect fertility in stressed men.
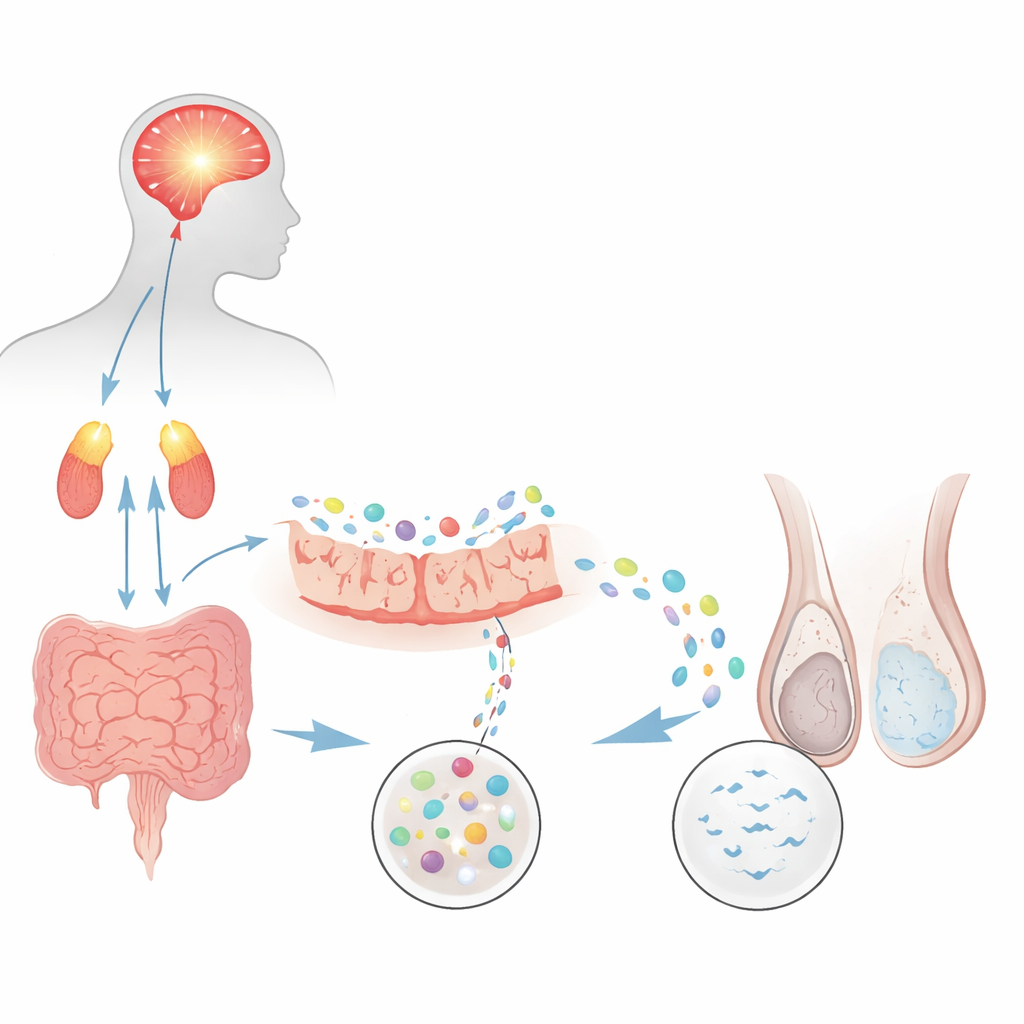
From Mental Strain to Fewer Sperm
The researchers first asked whether a well‑established rat model of chronic psychological stress could mimic a common form of male infertility called oligoasthenozoospermia, in which sperm are both too few and too sluggish. Rats repeatedly exposed to forced swimming developed strong signs of anxiety and depression, along with elevated stress hormones from the brain–adrenal system. When their semen was examined with automated tools, stressed animals had roughly half the sperm concentration and sharply lower motility compared with controls. Markers of sperm development inside the testes also dropped, and mating tests showed fewer pregnancies and smaller litters. Together, these findings showed that persistent stress alone can damage sperm production and performance.
The Gut as Middleman
Because the intestinal tract is highly sensitive to stress hormones, the team next looked at the gut. In stressed rats, the protein seals that normally tighten the intestinal lining were weakened, suggesting a “leaky” barrier. Blocking the glucocorticoid receptor—the main docking point for stress hormones—with a drug largely restored both gut integrity and sperm quality, tying the damage to hormone signaling. To test whether gut microbes themselves were part of the problem, the scientists transplanted fecal bacteria from stressed or control donors into new rats whose own microbiota had been wiped out by antibiotics. Animals receiving microbes from stressed donors developed the same poor sperm counts, low motility, and reduced fertility, even though they had never been stressed, revealing that disturbed gut communities can drive reproductive problems at a distance.
A Helpful Bacterium and Missing Vitamins
Microbial DNA profiling showed that chronic stress reduced overall diversity in the small intestine and, in particular, depleted Lactobacillus, a group of bacteria often associated with gut and reproductive health. The abundance of these microbes strongly tracked with sperm counts and motility. When stressed rats were given oral Lactobacillus throughout the stress period, their semen quality and testis markers improved, and fertility outcomes moved back toward normal. At the same time, chemical analyses of gut contents, blood, and testes revealed that stress disrupted bile acids and fat‑soluble vitamins, especially vitamins A and E, which are important for germ‑cell development and protection against oxidative damage. Levels of several bile acids and these vitamins fell in the circulation and in testicular tissue, and these declines were closely linked both to the loss of Lactobacillus and to worsening sperm parameters.
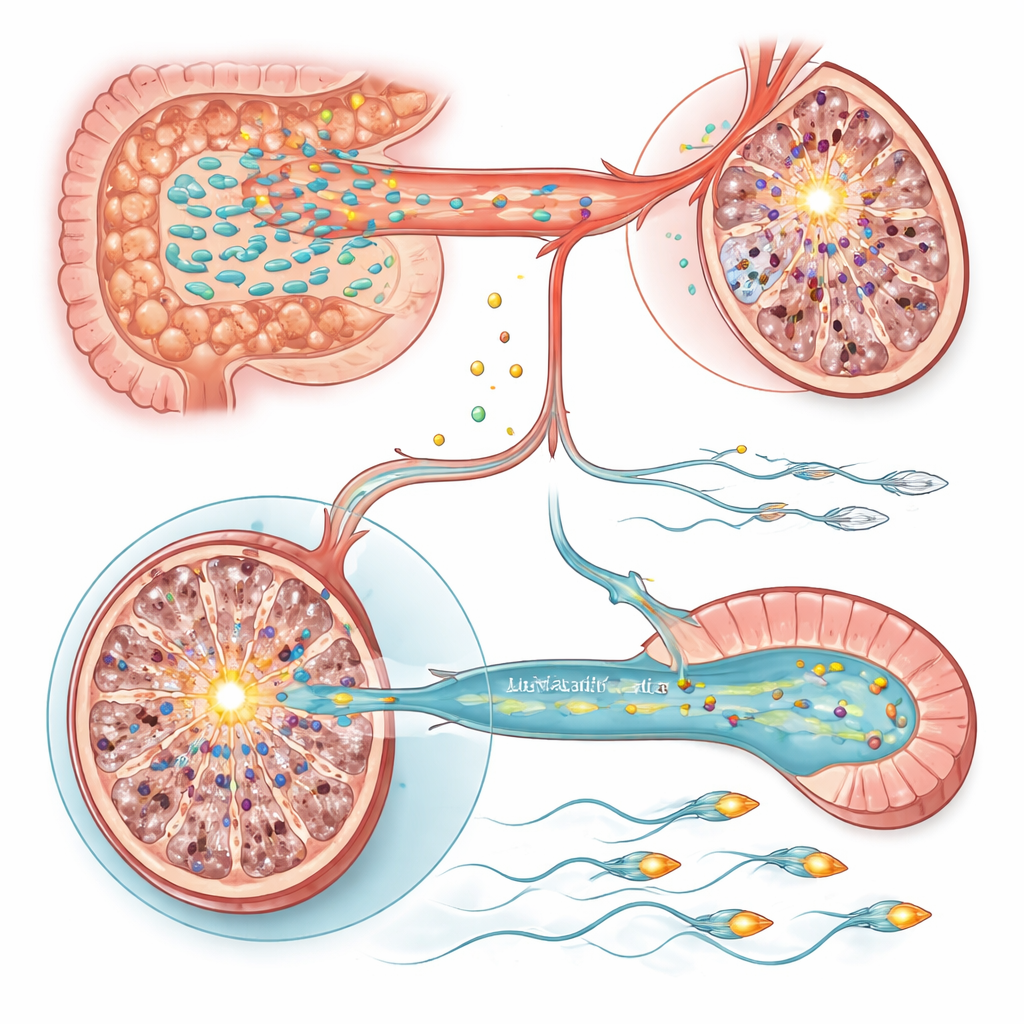
Two Molecular Switches in Testis and Sperm
To understand how these metabolic changes alter cells, the team examined gene activity in the testes and epididymis, where sperm mature. In the testes of stressed rats, one gene stood out: Sting1, a master switch in innate immune and cell‑death pathways, was strongly activated. Artificially boosting Sting1 in otherwise healthy animals was enough to reduce sperm production, while silencing it in stressed rats partially rescued sperm numbers. Both Lactobacillus treatment and vitamins A and E supplementation dialed Sting1 back down, linking microbe‑driven vitamin balance to this inflammatory switch. In contrast, reduced sperm motility traced to the epididymis, where a gene called Slc9c2, encoding the sperm‑specific ion exchanger NHE11 that helps control internal pH and tail function, was suppressed by stress. Lower NHE11 was associated with weaker sperm movement and impaired ability to undergo activation steps needed for fertilization. Restoring Slc9c2 expression, or supplementing Lactobacillus or vitamins A/E, improved motility, and experimentally knocking down Slc9c2 erased these gains.
What This Means for Men Under Pressure
Taken together, the study outlines a chain of events that begins with chronic psychological strain and ends with fewer, less active sperm. Stress hormones weaken the gut barrier and shrink beneficial Lactobacillus populations, which in turn disrupt bile acids and vitamins A and E. These nutrient shifts switch on a cell‑death pathway in the testes via Sting1, cutting sperm production, and dim the activity of NHE11 in the epididymis, slowing sperm tails. Although the work was done in rats and more research in humans is needed, it reframes certain cases of male infertility as a whole‑body condition involving the brain, gut microbes, nutrients, and reproductive organs. It also raises the possibility that carefully designed probiotic and vitamin strategies, alongside stress management, could one day help protect or restore fertility in men living with chronic psychological stress.
Citation: Zheng, JC., Jin, ZR., Liu, BH. et al. Chronic stress drives oligoasthenozoospermia via gut microbiota-vitamin-Sting1/Slc9c2 axis. Nat Commun 17, 4108 (2026). https://doi.org/10.1038/s41467-026-72450-4
Keywords: male infertility, chronic stress, gut microbiota, vitamin A and E, sperm motility