Clear Sky Science · en
Structures of partially occupied hetero-tetramers provide insight into kainate receptor activation and desensitization
Why tiny brain switches matter
Kainate receptors are microscopic protein switches that help brain cells talk to each other. They respond to the neurotransmitter glutamate and are involved in processes ranging from learning and memory to pain and inflammation. When these switches malfunction, they have been linked to disorders such as epilepsy and psychiatric disease. This study explores, in unprecedented structural detail, how one major kainate receptor type turns on and then “shuts itself off” when only some of its subunits are bound by glutamate-like molecules—a situation that likely reflects what actually happens at many real synapses.
How this brain receptor is built
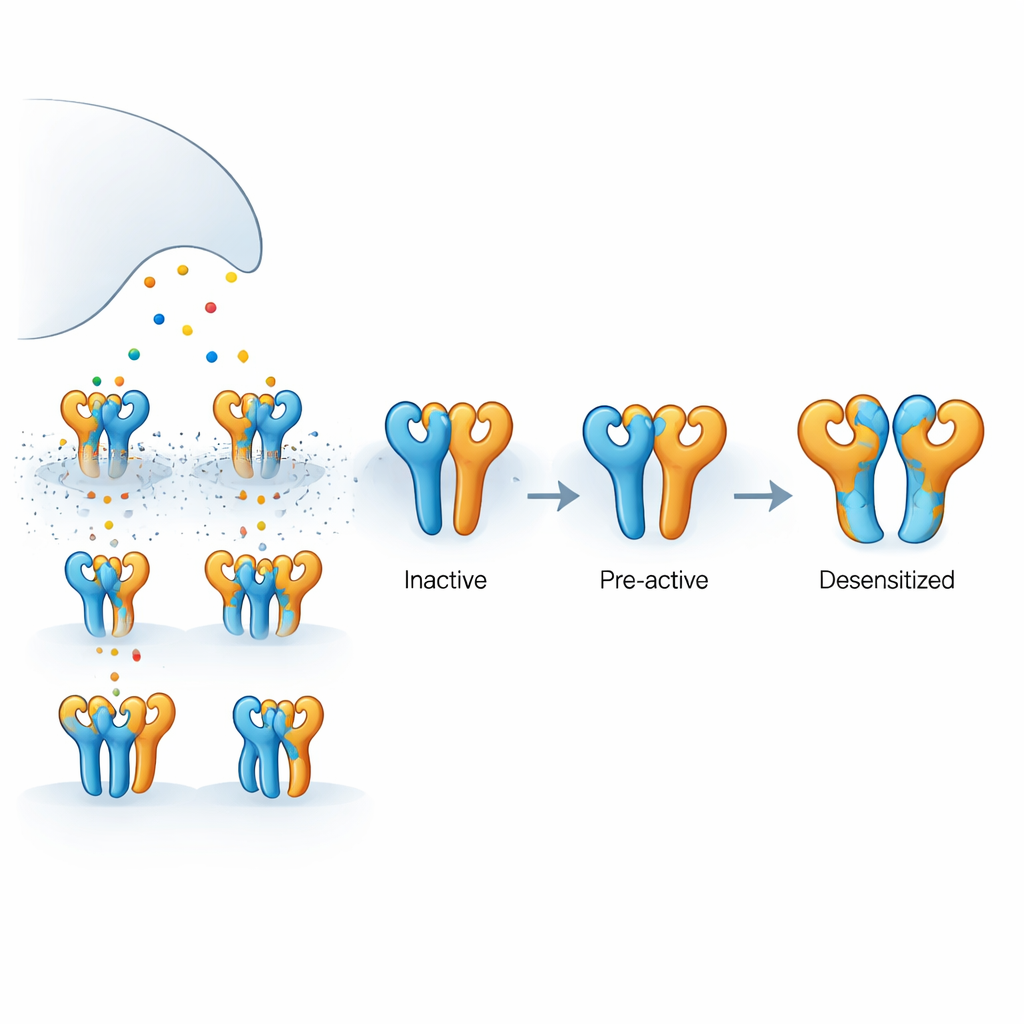
The receptor examined here is a kainate receptor made from two kinds of protein subunits, called GluK2 and GluK5, assembled into a four-part complex. GluK5 binds glutamate especially tightly and tends to occupy two specific positions in the tetramer, while GluK2 occupies the other two. Each subunit has an outer domain that catches glutamate and an inner pore-forming region that lets ions flow, generating electrical signals. Because glutamate is released briefly and is quickly taken up, not all four subunits are always occupied at once. Understanding what happens when only some of them are bound is key to explaining how real synapses modulate signal strength and timing.
Partially pressed triggers that do not jam
Using electrical recordings from human cells, the authors showed that a GluK5-selective drug, 5-iodowillardiine, drives a steady current through GluK2/GluK5 receptors without causing them to “desensitize”—the self-protective shutoff that usually follows activation. In contrast, the natural transmitter glutamate causes the same receptors to turn on and then very quickly fall silent. Another drug, AMPA, also activates these receptors but produces only partial desensitization. These behaviors suggest that when only the GluK5 subunits are occupied, the receptor can open or approach opening without falling into a long-lived inactive state, whereas more widespread binding pushes the receptor into a deeper shutoff configuration.
Snapshots of moving parts in action
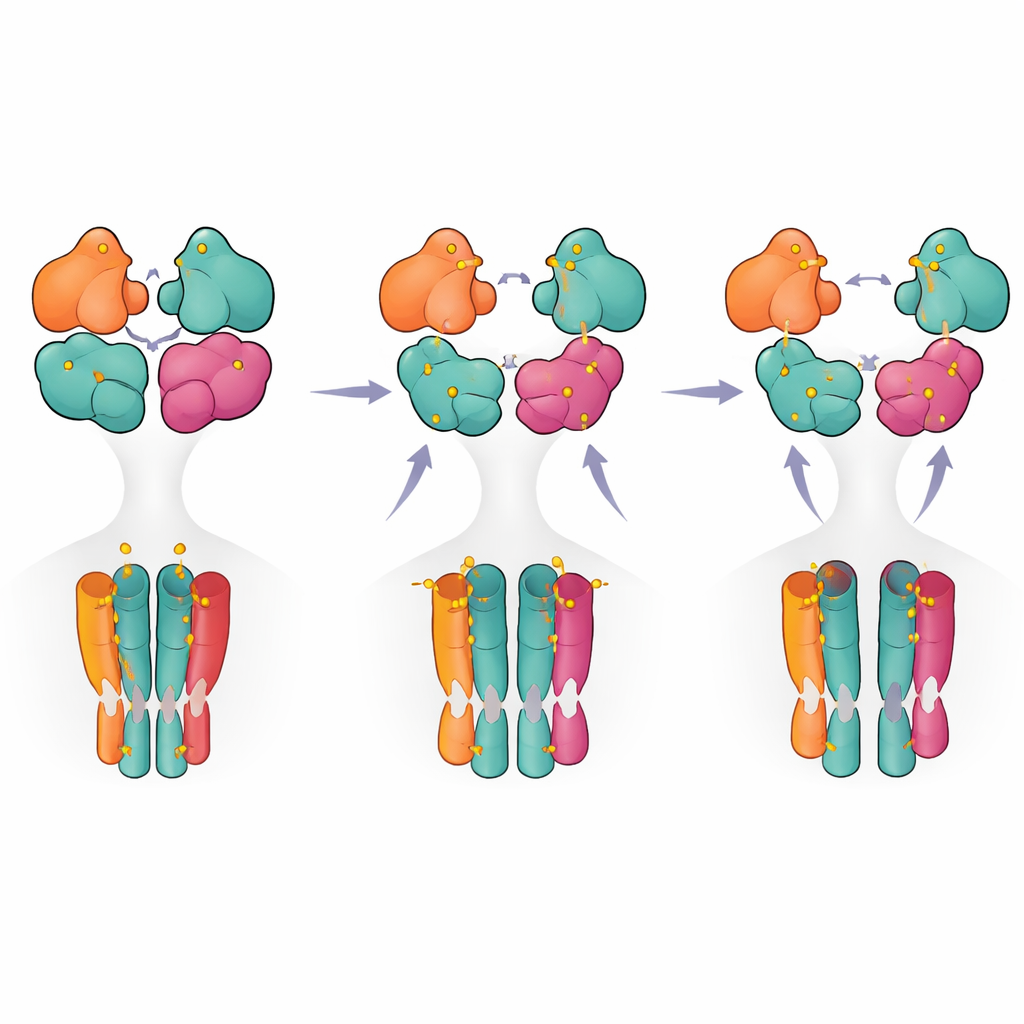
To see how these different functional states look in three dimensions, the team used cryogenic electron microscopy to capture structural snapshots under carefully controlled ligand conditions. With the GluK5-selective agonist present, they observed two main conformations. In one, all four ligand-binding domains formed intact pairs, with GluK5 units closed around ligand and GluK2 units remaining open. This “intact” state showed subtle loosening and rotation between the paired domains and a measurable expansion of the inner pore helices, consistent with a pre-active, ready-to-open configuration even though the ion channel was still closed. In the second, “partially ruptured” state, one pair of domains had split apart while the other remained intact, suggesting an intermediate on the path toward full desensitization but not yet locked in.
Hidden hinges that tune timing
By comparing these structures with previously solved fully active and fully desensitized kainate receptors, the authors traced how motions in the outer domain layer are transmitted to the pore. They identified key contact regions both within and between domain pairs that act as hinges and braces, especially around the two GluK5 subunits that sit opposite each other in the complex. Subtle shifts at these sites changed how strongly the domains hold together and how they tug on the linkers that pull the pore open. When the researchers swapped certain amino acids at these interfaces, they could make the receptor deactivate more quickly or more slowly and alter how readily it desensitized. This helps explain why GluK5-containing receptors, in particular, stay active unusually long after brief glutamate bursts.
A stepwise roadmap from activation to shutdown
Bringing together the structural snapshots and functional measurements, the authors propose a stepwise model in which the receptor moves from a resting state to a pre-active state when only two subunits (typically the GluK5 pair) are occupied. Additional ligand binding promotes partial rupture of one domain pair, and only when three or four subunits are occupied does the receptor reach a fully ruptured configuration associated with long-lasting desensitization. For non-specialists, this means that the receptor behaves less like a simple on–off switch and more like a multi-step dimmer, with different occupancy patterns and internal contacts tuning how strongly and how long it signals. These insights not only clarify the basic workings of a key brain receptor but also highlight structural hotspots that could be targeted by future drugs aimed at fine-tuning kainate receptor activity in disease.
Citation: Khanra, N.K., Strauss, A., Moreno Wasielewski, L. et al. Structures of partially occupied hetero-tetramers provide insight into kainate receptor activation and desensitization. Nat Commun 17, 3789 (2026). https://doi.org/10.1038/s41467-026-72226-w
Keywords: kainate receptors, glutamate signaling, synaptic transmission, cryo-EM structures, receptor desensitization