Clear Sky Science · en
CXCR5+ monocyte emigration impairs the radiation-induced antitumor immune response
When Cancer Treatment Fights Against Itself
Radiation therapy is a mainstay of cancer care, used in more than half of all patients. It is designed to damage tumor cells and rally the immune system to finish the job. Yet many tumors bounce back after an apparently successful course of treatment. This study uncovers a surprising reason why: certain immune cells are drawn into the tumor after radiation and end up shielding the cancer instead of helping to destroy it. 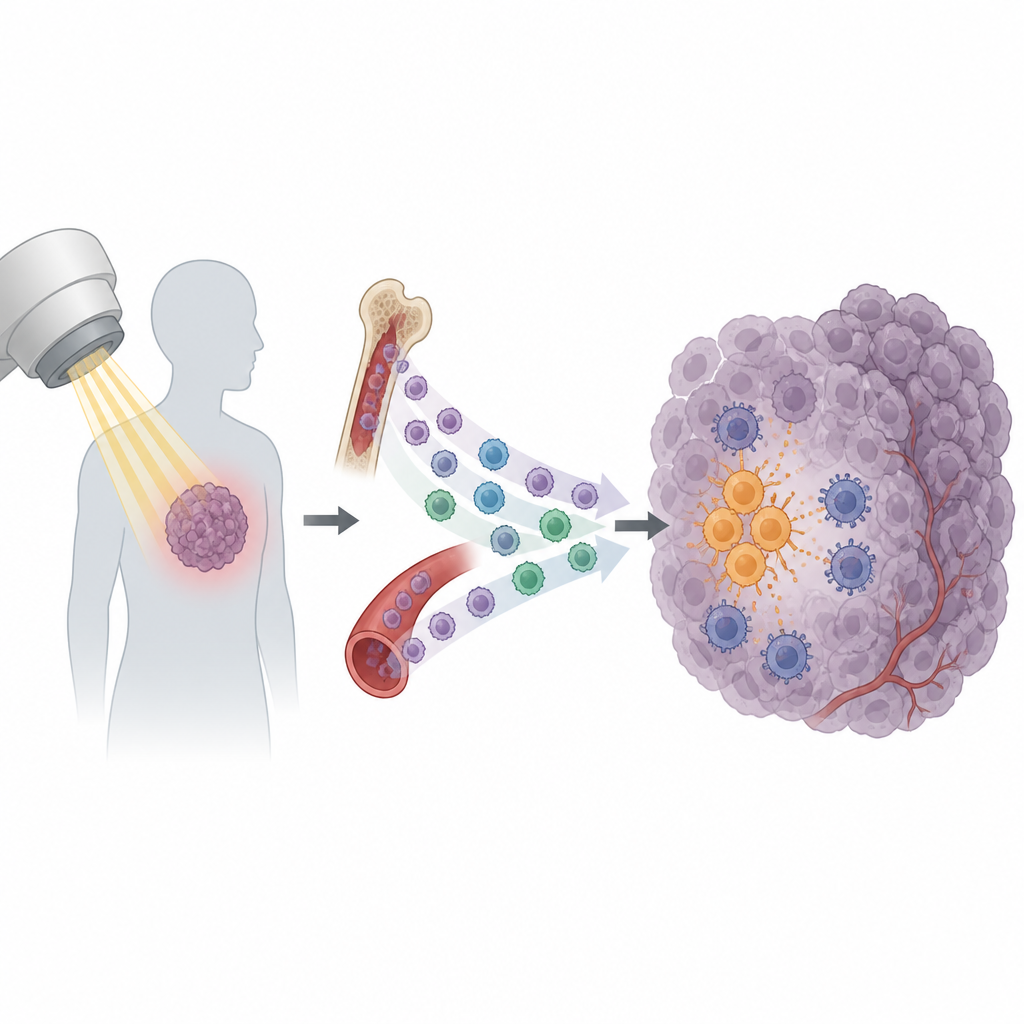
A Double-Edged Effect of Radiation
Radiation does more than just break DNA in cancer cells. It also causes tumors to release a burst of signals that call in immune cells. Some of these signals help activate killer T cells, which can recognize and attack tumor cells. But others appear to have the opposite effect. The researchers focused on a subset of circulating immune cells called monocytes. In several mouse tumor models, radiation caused a sharp rise in monocytes entering the tumor, while other white blood cell types changed little. This pattern suggested that monocytes might be important players in whether a tumor becomes sensitive or resistant to radiation.
A Chemical Trail that Lures Suppressive Cells
Digging deeper, the team found that tumors exposed to radiation produced more of a chemical signal called CXCL13. Monocytes that carry a matching “receiver” molecule, CXCR5, were especially likely to flood into the tumor after treatment. Before any radiation was given, the cancer itself had already been priming these cells at a distance. Tumor cells secreted the growth factor VEGF into the bloodstream, which switched on a chain of signals inside monocytes known as the PI3K, Akt, mTOR, and HIF-1α pathway. This internal wiring change increased CXCR5 on their surface, effectively creating a circulating reserve of CXCR5-positive monocytes ready to follow the CXCL13 trail into irradiated tumors.
How Recruited Cells Silence Tumor Killers
Once inside the tumor, CXCR5-positive monocytes did not behave like helpful defenders. Instead, they strongly curbed the activity of CD8 T cells, the very cells needed for the immune system to clear cancer after radiation. In laboratory tests, these monocytes slowed T cell growth and reduced their production of key attack molecules. The effect depended in large part on PD-L1, a checkpoint protein on the monocytes that engages PD-1 on T cells and tells them to stand down. Mice lacking CXCR5, or tumors lacking CXCL13, showed better tumor control after radiation and more active CD8 T cells. Blocking CXCL13 or PD-L1 with antibodies further improved the impact of radiation in several models, pointing to a clear chain from monocyte recruitment to T cell suppression and treatment failure. 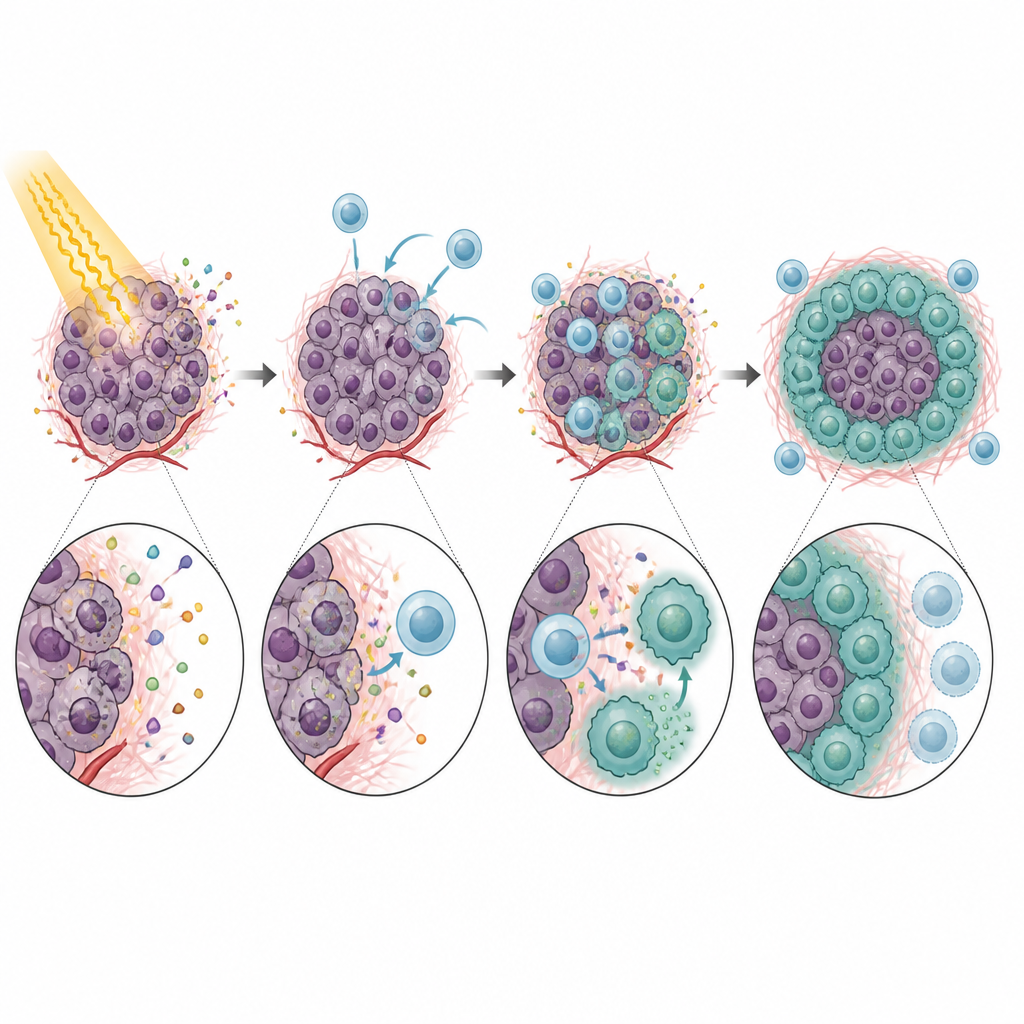
From Mobile Suppressors to Settled Shield Cells
The story did not end with monocytes. After entering the tumor, many CXCR5-positive monocytes matured into macrophages, another type of immune cell. Under the influence of a radiation-induced growth factor called GM-CSF, these descendants took on features of so-called M2-like macrophages, which are known to dampen immune responses and support tissue repair. These CXCR5-positive macrophages were rich in PD-L1 and other genes linked to immune suppression, and they were particularly effective at blocking T cell activity. Reducing GM-CSF levels or disrupting the CXCL13–CXCR5 axis decreased the number of these M2-like macrophages and improved tumor responses to radiation in mice.
Hints from Patient Samples and Possible New Strategies
To test whether similar processes occur in people, the researchers examined publicly available cancer datasets and samples from patients receiving radiotherapy. Tumors from patients who had undergone radiation showed higher levels of CXCR5, CD14 (a human monocyte marker), and PD-L1. In blood samples, monocyte numbers rose after treatment in patients whose disease later progressed, but not in those whose tumors shrank or remained stable. In rectal cancer specimens, tumors after radiotherapy contained more CXCL13 and CXCR5-positive monocytes and macrophages. In cell culture, human monocytes exposed to tumor fluids behaved much like those in mice, gaining CXCR5 through VEGF-driven signaling and expressing high PD-L1.
Turning a Weakness into an Advantage
Overall, this work shows that radiation can unintentionally call in and reshape a population of immune cells that protect tumors from the very treatment meant to destroy them. By boosting CXCL13 in tumors and VEGF-driven CXCR5 in monocytes, radiation creates a pipeline that delivers suppressive monocytes and macrophages, which then switch off killer T cells through PD-L1. In mouse models, interrupting this pipeline at several points, such as blocking VEGF, CXCL13, GM-CSF, or PD-L1, allowed radiation to work more effectively. For patients, these findings suggest that pairing radiotherapy with carefully chosen immune-targeted drugs may tip the balance back toward a true antitumor response and improve long-term control of cancer.
Citation: Lei, Y., Jia, R., Chen, C. et al. CXCR5+ monocyte emigration impairs the radiation-induced antitumor immune response. Nat Commun 17, 4258 (2026). https://doi.org/10.1038/s41467-026-70858-6
Keywords: radiotherapy, tumor immunity, monocytes, immune suppression, PD-L1