Clear Sky Science · en
Cryo-EM structure of TRPM1 reveals a non-canonical architecture with an inverted transmembrane domain
Seeing in the dark
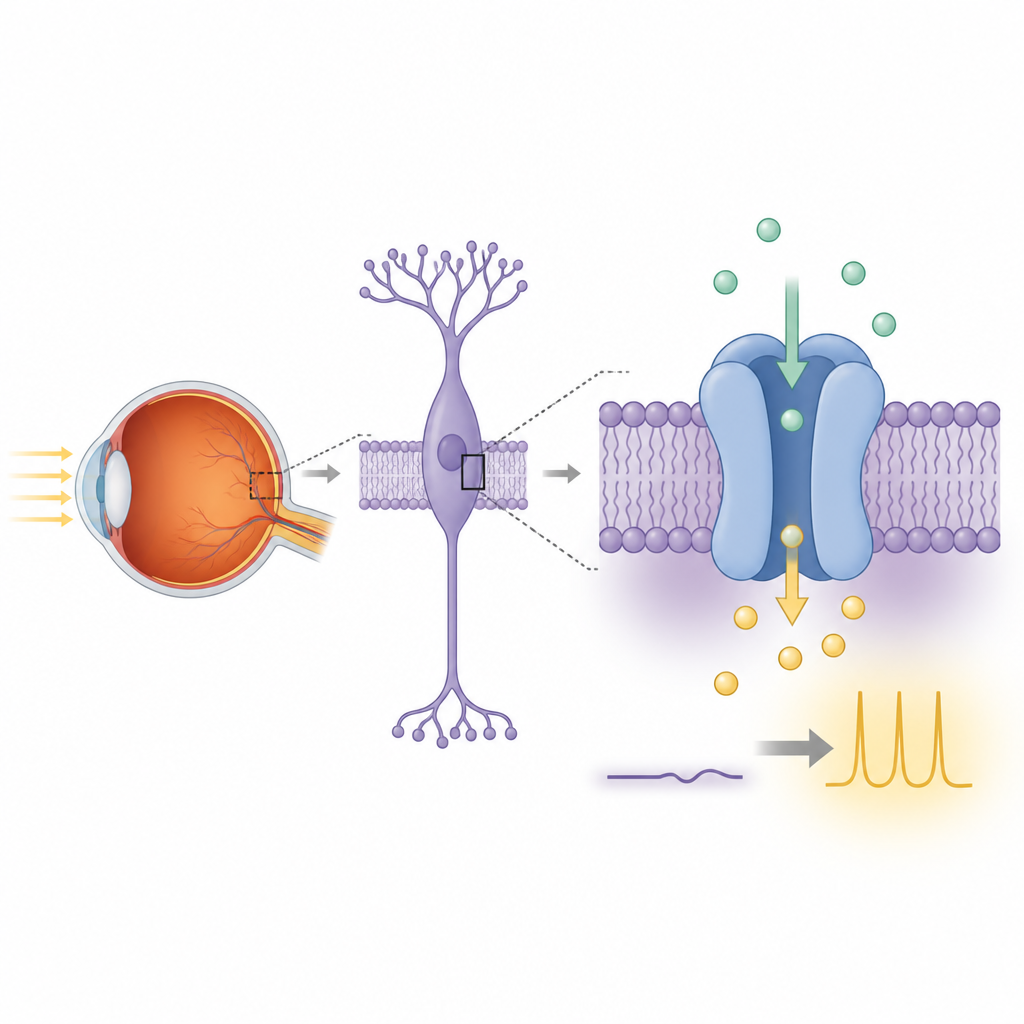
Anyone who has stumbled through a dark room has relied on a delicate set of molecules in the eye that make night vision possible. One of these, a protein called TRPM1, has long been suspected to act as a tiny gate in nerve cells of the retina, letting charged atoms flow and carrying signals about dim light. Yet scientists have struggled to show exactly how TRPM1 is built and whether it truly works as a stand‑alone ion channel. This study combines advanced imaging and cell experiments to reveal TRPM1’s unusual structure and how it likely supports night vision.
How night vision signals travel
Night vision begins when rod photoreceptors in the retina detect single photons and pass that information to nearby bipolar cells. Inside these bipolar cells, a signaling chain links a glutamate receptor called mGluR6 to TRPM1. In the light, TRPM1 is associated with a steady inward current of positively charged ions that keeps these cells depolarized. In the dark, activation of mGluR6 releases G‑protein subunits that shut this current off, repolarizing the cell and ending the signal. When TRPM1 or its partners are damaged by genetic mutations, this light‑driven communication fails and people can develop complete congenital stationary night blindness, a condition where seeing in low light is severely impaired.
Testing whether TRPM1 is a real channel
Despite its close similarity to other TRPM family ion channels, TRPM1’s true function has been debated for years because its electrical activity is hard to reproduce in standard lab cell lines. The authors first created a rat TRPM1 construct suited for structural work, trimming a floppy tail region that is not needed for folding. They expressed either full‑length or truncated TRPM1 in human HEK293 cells engineered to glow when calcium enters. When calcium was added outside the cells, many more TRPM1‑expressing cells lit up than control cells, and their fluorescence increased far more strongly. These observations indicate that TRPM1 on its own can create a pathway for calcium ions to cross the membrane, supporting the idea that it is a bona fide ion channel.
A surprising channel shape
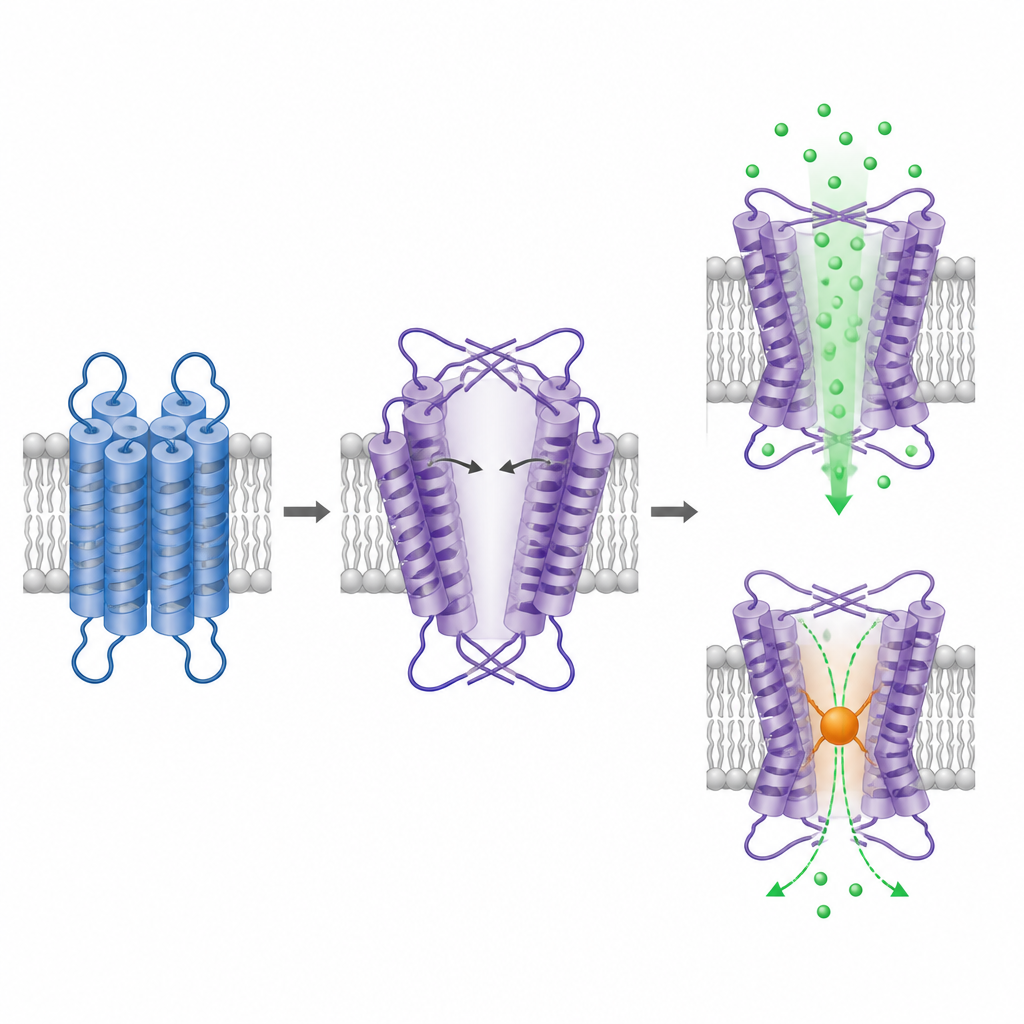
To see how TRPM1 is built, the team purified the truncated protein and imaged it with cryogenic electron microscopy, a technique that visualizes individual particles frozen in a thin layer of ice. The intracellular portion of TRPM1 forms a familiar fourfold symmetric arrangement similar to other TRPM channels, reinforcing its identity as a tetrameric channel. The surprise lies in the part that spans the membrane. Here, the voltage sensor‑like region and the pore region are still arranged in a domain‑swapped fashion, but with the opposite handedness compared to all related ion channels studied so far. This inverted layout forces two of the pore helices to bend and stretch at shallow angles, creating a large central cavity that resembles a wide, open pore.
Following ions through the pore
The enlarged cavity in TRPM1’s membrane domain is estimated to be at least about one nanometer across at its narrowest point, comparable to the openings of other TRPM channels in their active state. This supports previous suggestions that TRPM1 tends to be constitutively open. To test whether the putative pore really carries calcium, the researchers engineered a cysteine at the narrowest point and applied a chemical reagent that attaches to exposed cysteines and adds bulk. This treatment strongly reduced calcium‑dependent fluorescence only in the mutant channel, indicating that blocking this position interferes with ion permeation. Together, the structure and the blocking experiment argue that the observed cavity is the functional ion pathway in TRPM1.
Clues to drug responses and disease
Because TRPM1 is closely related to TRPM3, which has been structurally characterized with various small‑molecule ligands, the authors compared known binding pockets between the two channels. Sites in the voltage sensor‑like region that bind activators and inhibitors in TRPM3 are well conserved in TRPM1, and the inhibitor primidone indeed dampened TRPM1‑dependent calcium entry. In contrast, a steroid that activates TRPM3 at a pocket between the sensor and pore could not be accommodated in TRPM1’s inverted architecture and failed to boost TRPM1 activity. The team also mapped human night blindness mutations onto the structure; most lie in regions that appear to stabilize the protein fold or control its trafficking to the right place in retinal cells.
What this means for night vision
In plain terms, this work shows that TRPM1 really is an ion channel, but with a membrane‑spanning region arranged in an unexpected, inverted way that still allows a large pore for ions to pass. This unusual shape may explain why TRPM1 behaves differently from its relatives and responds to some drugs but not others. By tying the three‑dimensional architecture of TRPM1 to its ability to carry calcium, the study offers a structural foundation for understanding inherited night blindness and for exploring how this special channel helps our eyes see in the dimmest light.
Citation: Fabrizio, M., Brewer, M., Bogdanović, N. et al. Cryo-EM structure of TRPM1 reveals a non-canonical architecture with an inverted transmembrane domain. Nat Commun 17, 4260 (2026). https://doi.org/10.1038/s41467-026-70722-7
Keywords: TRPM1, ion channel, night vision, retina, cryo-EM