Clear Sky Science · en
Allosteric activation of the glutamate receptor mGlu2 by the serotonin receptor 5-HT2A
How Brain Cell Gatekeepers Team Up
Brain cells are covered with tiny “gatekeeper” proteins that sense chemical messengers like glutamate and serotonin. This study shows that two such gatekeepers, long linked to schizophrenia and Parkinson’s disease, do more than work side by side: they physically team up so that one quietly boosts the activity of the other. Understanding this partnership could help scientists design drugs that fine-tune brain signals with greater precision and fewer side effects.
Signals at the Cell Surface
Many commonly used medicines act on a huge family of cell-surface proteins called G protein–coupled receptors, or GPCRs. Each GPCR detects particular external signals and passes the message inside the cell. Because a single nerve cell can display dozens of GPCR types at once, researchers have long wondered how these receptors avoid sending mixed messages. One proposal is that different GPCRs physically join together, forming pairs or larger groups that share information. But whether such groupings really matter for how signals are processed has been hotly debated.
A Surprising Partnership in the Brain
The researchers focused on a specific duo found in brain regions involved in mood, perception and movement: the metabotropic glutamate receptor mGlu2 and the serotonin receptor 5-HT2A. Both are targets of antipsychotic drugs, and earlier work suggested that they form a functional pair in living brain tissue. Here, the team asked two key questions: Do these receptors truly sit in direct contact on the cell surface, and if so, does that physical contact change how mGlu2 behaves?
Proving the Receptors Touch
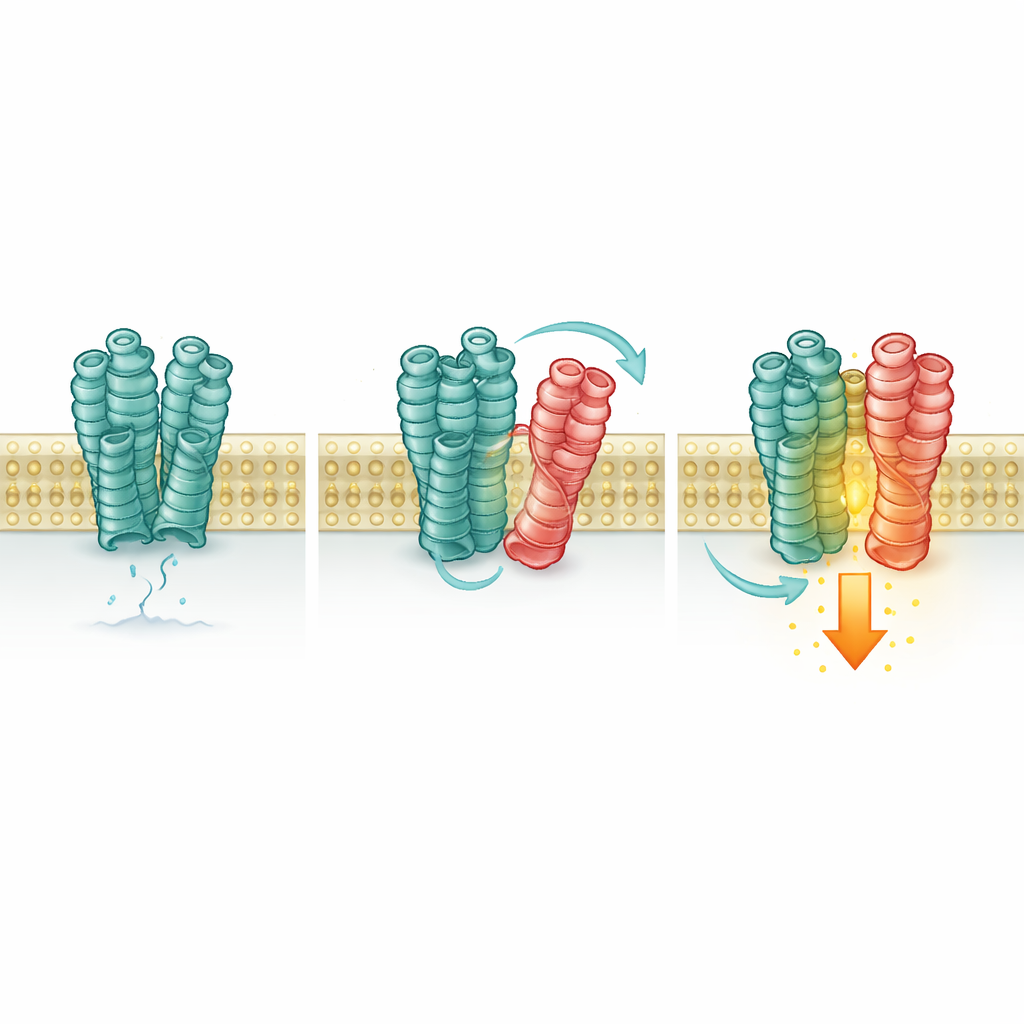
To see whether mGlu2 and 5-HT2A actually come together, the authors used human cells grown in the lab and tagged each receptor with tiny fluorescent markers. When the tagged proteins move close enough, energy can jump from one marker to the other, creating a measurable signal. These experiments showed a strong signal when both receptors were present, indicating that they cluster together on the cell surface. The team then used a clever chemical trick, introducing specially placed “hooks” into each receptor so that, when close, they could be locked together. The resulting crosslinked complexes confirmed that mGlu2 and 5-HT2A contact each other through specific sections buried in the cell membrane.
One Receptor Quietly Switches On Another
Next, the scientists tested whether this contact changes how mGlu2 responds. Using sensitive fluorescent sensors and biochemical readouts, they monitored the shape and activity state of mGlu2. When 5-HT2A was present, even without added glutamate, more mGlu2 receptors shifted into their “on” conformation and showed a small but significant rise in signaling inside the cell. Blocking mGlu2 itself removed this effect, but turning 5-HT2A on or off with its own drugs did not, and a mutant 5-HT2A that cannot activate its usual signaling pathway still boosted mGlu2. This means 5-HT2A acts more like a supportive brace than an active switch: its mere presence, and physical contact, stabilizes the active form of mGlu2.
Many Other Partners Join In
The story does not end with serotonin. The authors screened 44 other GPCRs to see whether they could similarly influence mGlu2. Several, including multiple dopamine receptors and another serotonin subtype, also nudged mGlu2 toward its active state, while others had little effect. One dopamine receptor, D1, clearly increased the baseline activity of mGlu2 in the absence of extra glutamate, again consistent with a physical partnership that favors the “on” position. In contrast, a muscarinic receptor used as a control neither interacted strongly with mGlu2 nor altered its activity.
Why This Matters for Brain Disorders
This work shows that certain brain receptors do more than sit independently waiting for their own chemical keys. By forming small assemblies, one receptor can allosterically, or indirectly, tune the behavior of another. In the case of mGlu2 and 5-HT2A, this means serotonin-linked receptors can shape how glutamate signals are processed, even when they are not actively signaling themselves. Because these receptor partnerships are tied to schizophrenia, Parkinson’s disease and possibly other conditions, they offer an appealing new angle for drug design: medicines that target one member of a pair might be crafted to modulate the other’s activity, providing more subtle control over complex brain circuits.
Citation: Gai, S., Lin, L., Meng, J. et al. Allosteric activation of the glutamate receptor mGlu2 by the serotonin receptor 5-HT2A. Nat Commun 17, 3303 (2026). https://doi.org/10.1038/s41467-026-69939-3
Keywords: GPCR heteromers, mGlu2 receptor, 5-HT2A receptor, allosteric modulation, schizophrenia