Clear Sky Science · en
Biomimetic metal-drug coordination nanoplatform to counteract drug resistance in Pseudomonas aeruginosa via energy disruption
Why drug-resistant infections matter to all of us
Antibiotic-resistant infections are making once-treatable illnesses harder and more dangerous to cure. In hospitals, common bacteria are learning to evade our best medicines, turning routine pneumonia or wound infections into life-threatening crises. This study describes a new way to “reawaken” an old antibiotic against a particularly stubborn germ by cutting off the bacterium’s energy supply rather than inventing an entirely new drug.
A stubborn lung germ that shrugs off drugs
Pseudomonas aeruginosa is a hardy bacterium that often causes serious lung infections, especially in people with weakened defenses, such as those with cystic fibrosis or chemotherapy-induced immune suppression. It resists treatment by blocking drug entry through its outer membrane and by actively pumping antibiotics back out of the cell. At the same time, the global pipeline for brand-new antibiotics—especially those that work on this type of Gram-negative bacterium—has been thin for decades. The researchers therefore focused on a different goal: make existing antibiotics powerful again by disarming the bacterium’s built-in resistance systems.
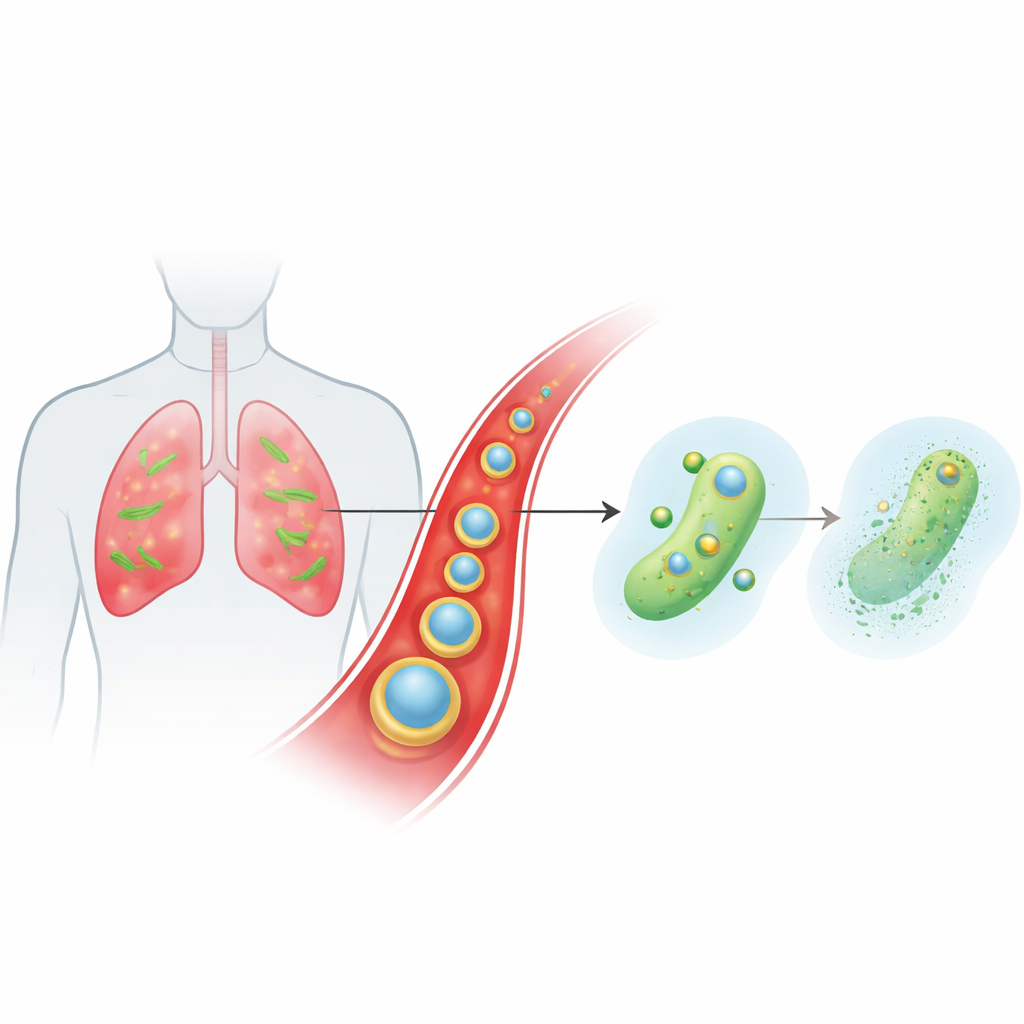
A smart carrier that rides the bloodstream to infection sites
The team designed a tiny, layered particle called Ce-Car@EV to deliver a classic beta-lactam antibiotic, carbenicillin, together with cerium, a metal ion with unusual chemistry. The inner core is a nanoscale coordination polymer in which carbenicillin molecules link up with cerium ions, creating a porous, high–drug-loading structure that falls apart in mildly acidic conditions. This core is then wrapped in natural extracellular vesicles harvested from ginger plants. These plant vesicles help the particles evade immune clearance, stay in the bloodstream longer, and preferentially accumulate in inflamed, infected lung tissue where blood vessels are leaky and immune cells are activated. Once the particles arrive in the acidic microenvironment of infection, their structure loosens and releases both carbenicillin and cerium ions on demand.
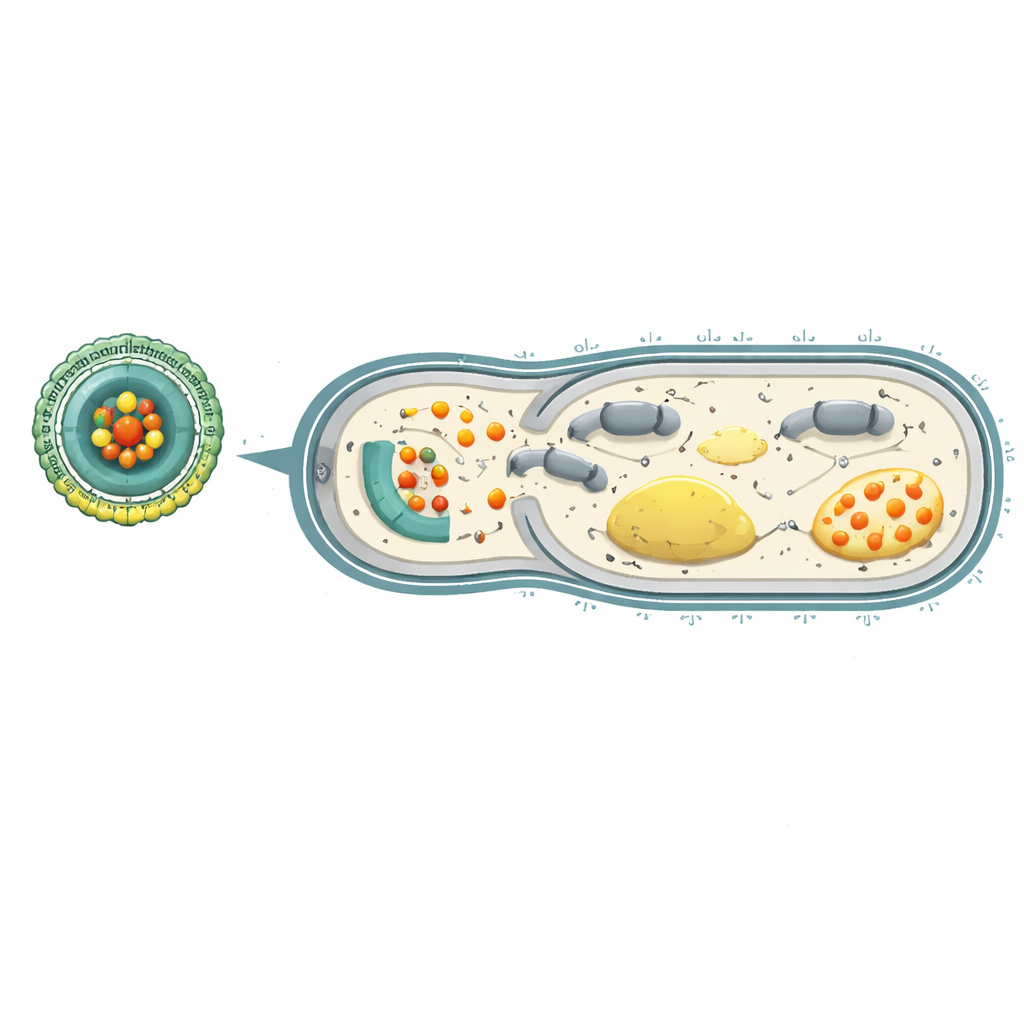
Starving bacteria of energy to stop drug resistance
Inside bacterial cells, adenosine triphosphate (ATP) acts as the universal energy currency, powering everything from cell wall building to the efflux pumps that spit out antibiotics. Cerium ions are able to break down ATP and disturb the chain of reactions that normally generate it. Laboratory tests showed that when resistant P. aeruginosa encountered Ce-Car@EV in an acidic environment similar to infection sites, the nanoparticles released cerium and carbenicillin, sharply cutting bacterial survival. Cerium not only helped carbenicillin bind more tightly to its protein target that builds the cell wall, but also depleted ATP and impaired key steps in bacterial respiration. As a result, the proton-driven “battery” across the bacterial membrane collapsed and the efflux pumps that normally remove drugs were left without power.
Reversing resistance and protecting the lungs
To see whether this strategy truly turned back resistance, the researchers grew P. aeruginosa for many generations in the presence of either free carbenicillin or the Ce-Car@EV platform. Bacteria exposed only to the antibiotic rapidly became highly resistant, needing 256 times more drug to be controlled. In contrast, those repeatedly treated with Ce-Car@EV developed far less resistance. Genetic analyses revealed that genes involved in energy production, respiration, and multidrug efflux pumps were dialed down in bacteria treated with the nanoplatform. In mice with severe, drug-resistant P. aeruginosa pneumonia, intravenous Ce-Car@EV accumulated strongly in infected lungs, dramatically lowered bacterial counts there and in other organs, reduced lung swelling and tissue damage, and improved survival, all while showing minimal toxicity in blood tests and organ examinations.
What this could mean for future treatments
By combining a familiar antibiotic with a metal ion inside a nature-inspired delivery shell, this work offers a blueprint for outsmarting resistant bacteria without relying solely on new drugs. The Ce-Car@EV particles home in on inflamed lungs, fall apart in the acidic infection niche, and then shut down the bacterium’s energy and drug-pumping systems so that carbenicillin can work again. If similar approaches prove safe and effective in humans, they could extend the useful life of existing antibiotics and provide new options against multidrug-resistant infections that currently leave doctors with few choices.
Citation: Ye, Y., Zhang, K., Wang, Y. et al. Biomimetic metal-drug coordination nanoplatform to counteract drug resistance in Pseudomonas aeruginosa via energy disruption. Nat Commun 17, 2808 (2026). https://doi.org/10.1038/s41467-026-69712-6
Keywords: antimicrobial resistance, Pseudomonas aeruginosa, nanoparticle therapy, energy metabolism, beta-lactam antibiotics