Clear Sky Science · en
Synthesis of 3-desoxycollinoketone B and its ability to reduce Alzheimer-associated misfolded proteins
A New Angle on Alzheimer’s
Alzheimer’s disease is driven in part by clumps of misfolded proteins that slowly damage the brain, but today’s treatments do little to remove these harmful deposits. This study introduces a lab-made cousin of a rare natural compound that can reach the brain more effectively and directly attack two major problem proteins at once. By combining advanced chemistry, artificial intelligence, and animal studies, the researchers show how this small molecule breaks down toxic protein aggregates and improves memory in mouse models of Alzheimer’s.
A Natural Molecule with Limitations
Scientists previously discovered a bacterial natural product called collinolactone that can dismantle protein clumps formed by amyloid-beta and tau—two hallmark proteins of Alzheimer’s disease. However, collinolactone is difficult to extract in useful quantities from its microbial source and has poor access to certain brain regions, especially the cortex, where much of the damage in Alzheimer’s occurs. These drawbacks make it hard to develop as a practical drug. The team set out to design and synthesize a related molecule that keeps the beneficial activity of collinolactone while improving its stability and its ability to cross into the brain.
Building a Smarter Molecule
Using modern organic synthesis, the researchers constructed a new compound called 3-desoxycollinoketone B. They assembled three simpler chemical building blocks and then triggered a rare ring-forming reaction that mimics a step believed to occur in nature. Detailed computer calculations explained why this reaction favors a specific three-ring shape that is more stable than alternative structures. The resulting molecule is slightly more fat-loving than the original natural product, a property expected to help it slip through the blood–brain barrier. Importantly, the synthetic route is scalable, meaning that gram-level quantities can be produced reliably in the lab for extensive biological testing and future optimization. 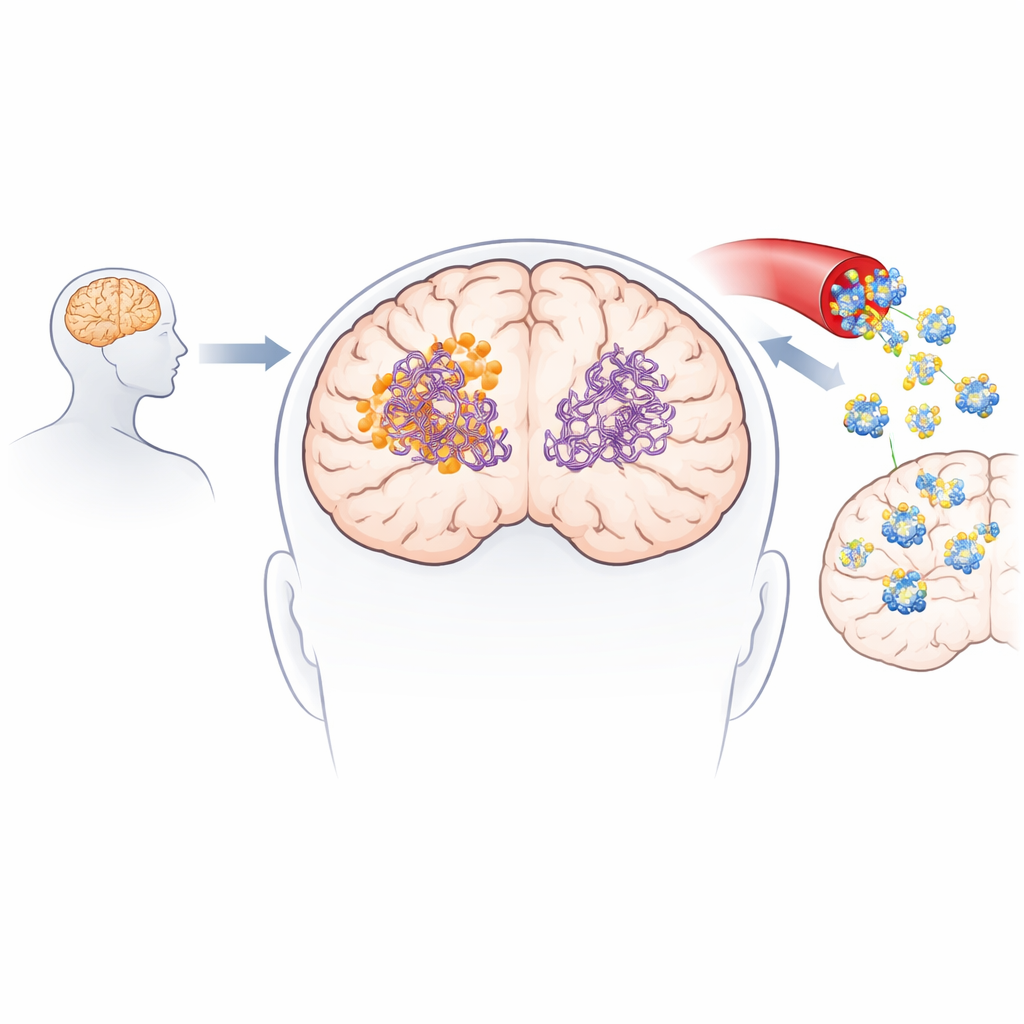
Simulations Reveal How It Grips Toxic Fibers
To understand how the new molecule might interact with disease-causing proteins, the team turned to artificial intelligence and molecular simulations. They used state-of-the-art docking models to explore thousands of ways the compound could attach to pre-formed amyloid-beta and tau fibers. The calculations consistently showed that 3-desoxycollinoketone B binds more tightly to both types of fibers than collinolactone does, nestling into hydrophobic (water-repelling) pockets and forming stabilizing contacts such as hydrogen bonds and salt bridges. Molecular dynamics simulations then followed the fibers over time and revealed that, once bound, the compound increases local flexibility and weakens critical contacts between protein strands, nudging the fibers toward structural breakdown.
From Test Tubes to Mouse Brains
Laboratory experiments confirmed that 3-desoxycollinoketone B not only slows the formation of new amyloid-beta and tau fibers, but also actively breaks apart existing fibers and smaller, highly toxic oligomers. In cell-free assays, it reduced amyloid-beta fibers and oligomers by significant amounts, and showed even stronger effects on tau fragments central to tangle formation. The molecule crossed artificial and biological models of the blood–brain barrier and was detectable in the brains of mice after both intravenous and oral dosing. In two different Alzheimer-like mouse models, short- and longer-term treatment lowered levels of amyloid plaques and soluble amyloid species, reduced markers of brain inflammation, partially restored synaptic proteins, and improved performance on memory and learning tasks such as Y-maze and novel object recognition. 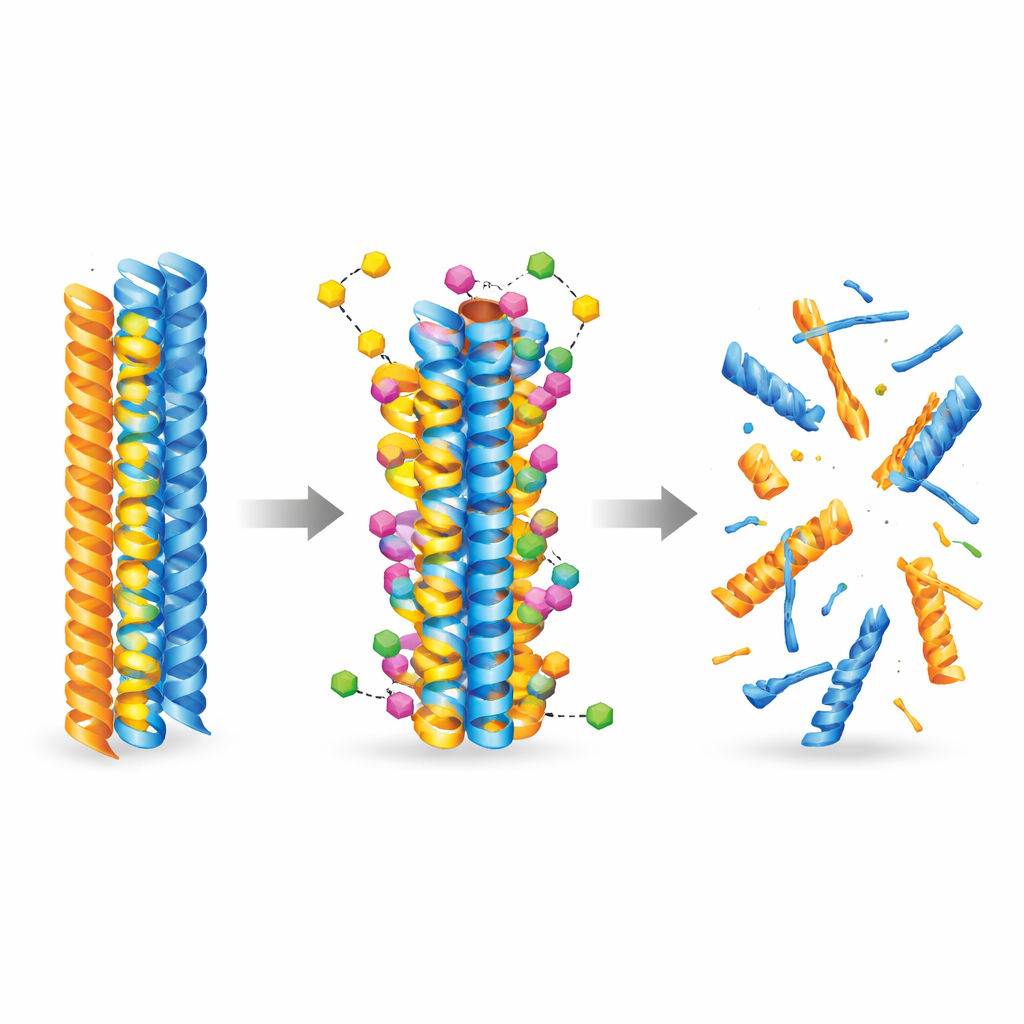
Promise and Next Steps
Overall, the study demonstrates that 3-desoxycollinoketone B is a dual-acting small molecule that can enter the brain, bind strongly to both amyloid-beta and tau aggregates, and promote their breakup, leading to measurable cognitive benefits in mice. For a layperson, this means that the compound acts a bit like a molecular solvent for two key types of Alzheimer’s-related protein clumps, rather than merely masking symptoms. While the molecule is cleared relatively quickly from the body and will need further tweaking to last longer in the bloodstream, its scalable synthesis, brain penetration, and broad activity against misfolded proteins make it a promising starting point for developing more accessible and cost-effective treatments that tackle Alzheimer’s disease at its root.
Citation: Heo, S., Cha, M., Zhung, W. et al. Synthesis of 3-desoxycollinoketone B and its ability to reduce Alzheimer-associated misfolded proteins. Nat Commun 17, 3288 (2026). https://doi.org/10.1038/s41467-026-69662-z
Keywords: Alzheimer’s disease, amyloid-beta, tau protein, small-molecule therapy, protein aggregates