Clear Sky Science · en
Deficiency of lysosomal TMEM175 in myeloid macrophages exerts anti-tumor immunity via inflammasome and cross-presentation pathway
Turning the Body’s Clean-Up Crew into Cancer Fighters
Cancer treatment increasingly relies on awakening the immune system, yet many tumors still slip past our defenses. This study explores an unexpected brake inside immune cells—an ion channel called TMEM175 that sits on tiny recycling compartments known as lysosomes. By removing TMEM175 specifically from macrophages, the body’s cellular clean‑up crew, the authors show in mice that tumors shrink, spread less, and respond better to existing immunotherapy. The work suggests a new way to rewire the tumor microenvironment so the immune system can recognize and destroy cancer cells more effectively.
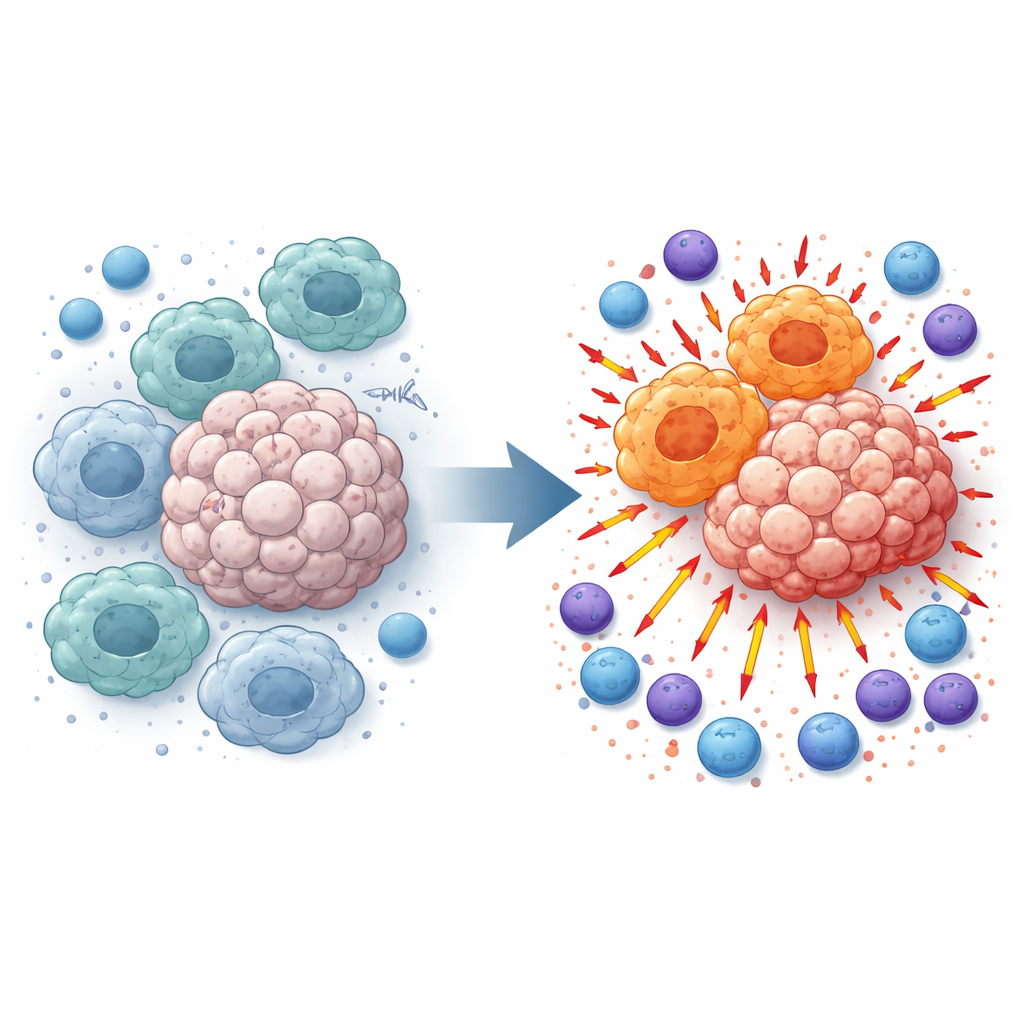
Why Certain Immune Cells Can Help Tumors Grow
Macrophages patrol tissues and gobble up debris, but inside tumors they often get reprogrammed into a nurturing, tumor‑friendly state. These “M2‑like” macrophages dampen inflammation and hinder T cells and natural killer (NK) cells that would otherwise attack cancer. Examining melanoma samples from patients and tumor‑bearing mice, the researchers found that TMEM175 levels were higher in tumors than in nearby normal tissue and were closely linked to this suppressive macrophage state. That observation led them to ask: what happens if TMEM175 is switched off only in myeloid cells such as macrophages?
Removing a Lysosomal Gatekeeper Tilts the Balance
The team engineered mice in which TMEM175 is deleted specifically in myeloid cells and then implanted melanoma or lung cancer cells under the skin, or into the bloodstream to provoke lung metastases. Across these models, tumors in TMEM175‑deficient mice grew more slowly and formed fewer metastatic nodules. Detailed cell analysis showed that macrophages inside these tumors shifted toward an “M1‑like” profile associated with strong immune activation, while the suppressive M2‑like population fell. At the same time, many more CD4 and CD8 T cells and NK cells accumulated in the tumor tissue and showed molecular signs of being switched on and ready to kill.
How Cell Debris Sparks a Fiery Immune Signal
To uncover the inner workings, the researchers studied bone‑marrow‑derived macrophages in culture. When these cells swallowed fragments of dead tumor cells, their lysosomes became destabilized, leaking enzymes such as cathepsin B and altering calcium levels in the surrounding fluid. In TMEM175‑deficient macrophages this disruption was stronger, leading to robust activation of a molecular alarm system called the NLRP3 inflammasome. Once active, this multi‑protein complex triggers the release of potent messenger molecules, including the cytokines IL‑1β and IL‑18. Blocking the enzyme caspase‑1, or neutralizing IL‑1β or IL‑18 with antibodies, largely erased the anti‑tumor benefit of TMEM175 loss in mice, showing that this inflammatory cascade is central to the effect.
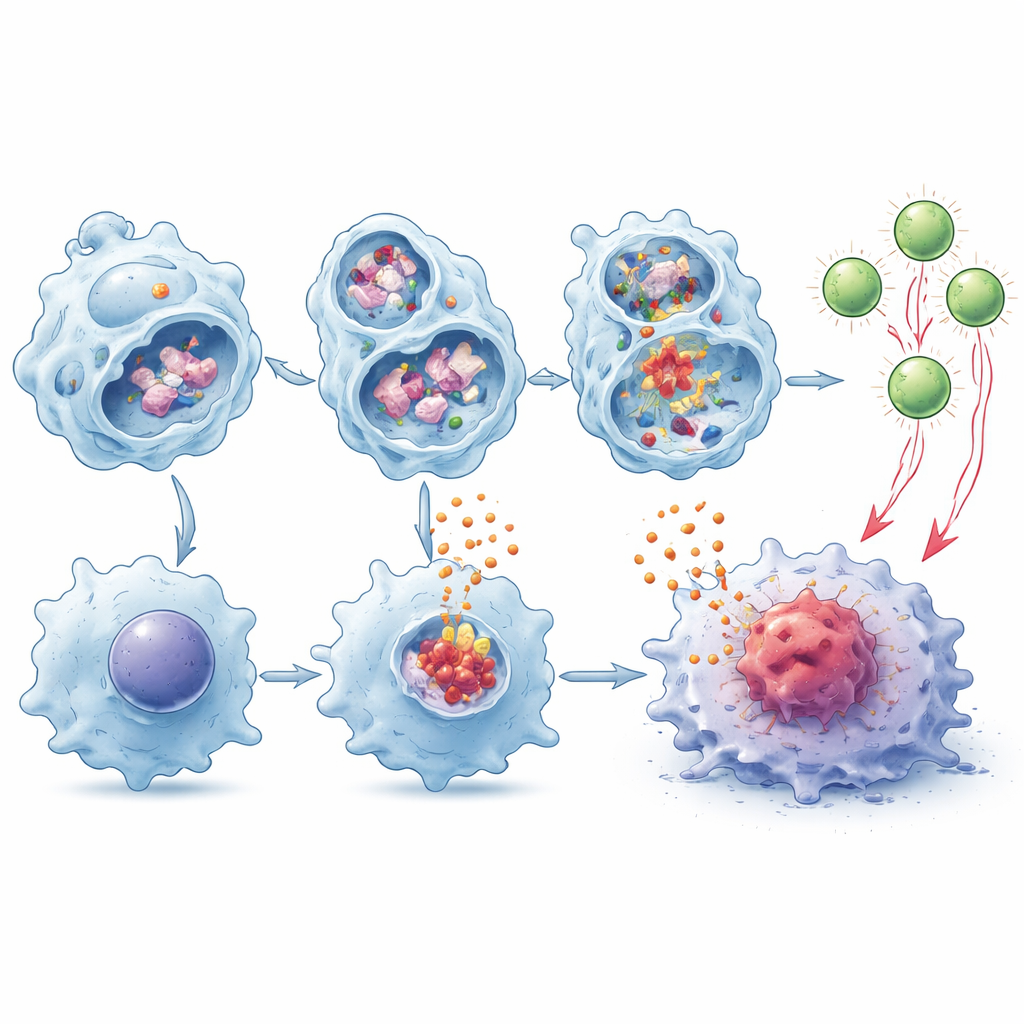
Helping T Cells See and Attack Tumors
Macrophages do more than secrete alarm signals—they can also chop up tumor proteins and display fragments on their surface to alert T cells, a process known as cross‑presentation. The authors found that TMEM175‑deficient macrophages broke down tumor material more slowly inside lysosomes, leaving more intact pieces available for display. In co‑culture experiments, these cells were much better at driving the expansion and activation of tumor‑specific CD8 T cells, and this effect grew even stronger in the presence of IL‑1β and IL‑18. When such primed T cells were transferred into tumor‑bearing mice that lack their own CD8 cells, they slowed tumor growth more effectively than T cells educated by normal macrophages.
Boosting the Power of Checkpoint Therapy
Because TMEM175‑deficient tumors were rich in active T cells, the researchers tested whether this setting would respond differently to anti‑PD‑1, a widely used checkpoint inhibitor. In standard mice, anti‑PD‑1 barely affected melanoma growth, mirroring resistance often seen in the clinic. In contrast, mice lacking TMEM175 in macrophages showed markedly smaller tumors and more tumor cell death when given the same drug. Their tumors contained more activated macrophages, T cells, and NK cells, indicating that reshaping the tumor environment through TMEM175 makes established immunotherapy work better.
A New Handle on the Tumor Microenvironment
Overall, the study reveals TMEM175 as a molecular switch that controls how macrophages respond to dying tumor cells. When this lysosomal ion channel is absent, tumor debris more readily destabilizes lysosomes, calcium levels rise, and the inflammasome fires, unleashing IL‑1β and IL‑18. At the same time, slowed degradation of tumor proteins improves their presentation to CD8 T cells. Together, these changes transform macrophages from tumor accomplices into powerful allies that rally T cells and NK cells against cancer and sensitize tumors to anti‑PD‑1 therapy. While drugs that selectively block TMEM175 and reach tumor‑associated macrophages still need to be developed, the work points to a promising new avenue for combination cancer immunotherapy.
Citation: Zhang, Z., li, X., Lu, T. et al. Deficiency of lysosomal TMEM175 in myeloid macrophages exerts anti-tumor immunity via inflammasome and cross-presentation pathway. Nat Commun 17, 2770 (2026). https://doi.org/10.1038/s41467-026-69546-2
Keywords: tumor microenvironment, macrophages, inflammasome, cancer immunotherapy, antigen cross-presentation